DEF 14AFALSE0001326110iso4217:USD00013261102025-01-012025-12-31000132611012025-01-012025-12-31000132611022025-01-012025-12-31000132611032025-01-012025-12-3100013261102024-01-012024-12-3100013261102023-01-012023-12-3100013261102022-01-012022-12-3100013261102021-01-012021-12-310001326110ibrx:SummaryCompensationTableValueOfStockAwardsMemberecd:PeoMember2025-01-012025-12-310001326110ibrx:SummaryCompensationTableValueOfOptionAwardsMemberecd:PeoMember2025-01-012025-12-310001326110ecd:YrEndFrValOfEqtyAwrdsGrntdInCvrdYrOutsdngAndUnvstdMemberecd:PeoMember2025-01-012025-12-310001326110ecd:ChngInFrValOfOutsdngAndUnvstdEqtyAwrdsGrntdInPrrYrsMemberecd:PeoMember2025-01-012025-12-310001326110ecd:ChngInFrValAsOfVstngDtOfPrrYrEqtyAwrdsVstdInCvrdYrMemberecd:PeoMember2025-01-012025-12-310001326110ibrx:SummaryCompensationTableValueOfStockAwardsMemberecd:NonPeoNeoMember2025-01-012025-12-310001326110ibrx:SummaryCompensationTableValueOfOptionAwardsMemberecd:NonPeoNeoMember2025-01-012025-12-310001326110ecd:YrEndFrValOfEqtyAwrdsGrntdInCvrdYrOutsdngAndUnvstdMemberecd:NonPeoNeoMember2025-01-012025-12-310001326110ecd:ChngInFrValOfOutsdngAndUnvstdEqtyAwrdsGrntdInPrrYrsMemberecd:NonPeoNeoMember2025-01-012025-12-310001326110ecd:ChngInFrValAsOfVstngDtOfPrrYrEqtyAwrdsVstdInCvrdYrMemberecd:NonPeoNeoMember2025-01-012025-12-31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14A INFORMATION
Proxy Statement Pursuant to Section 14(a)
of the Securities Exchange Act of 1934
(Amendment No. )
| | | | | | | | | | | | | | |
| Filed by the Registrant ☒ | | Filed by a Party other than the Registrant ☐ |
| | |
| Check the Appropriate Box: |
☐ Preliminary Proxy Statement |
☐ Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) |
☒ Definitive Proxy Statement |
☐ Definitive Additional Materials |
☐ Soliciting Material Under §240.14a-12 |
ImmunityBio, Inc.
(Name of Registrant as Specified in its Charter)
(Name of Person(s) Filing Proxy Statement, if other than the Registrant)
| | |
| Payment of Filing Fee (Check all boxes that apply): |
☒ No fee required. |
☐ Fee paid previously with preliminary materials. |
☐ Fee computed on table in exhibit required by Item 25(b) per Exchange Act Rules 14a-6(i)(1) and 0-11. |
Letter to Our Stockholders
Dear Fellow Stockholders,
This past year and the early part of 2026 have been transformational for ImmunityBio. We achieved commercial scale, secured multiple international approvals, and validated our vision of Immunotherapy 2.0 with clinical results that continue to exceed expectations. We are proud to share these achievements with you.
From Approval to Commercial Success
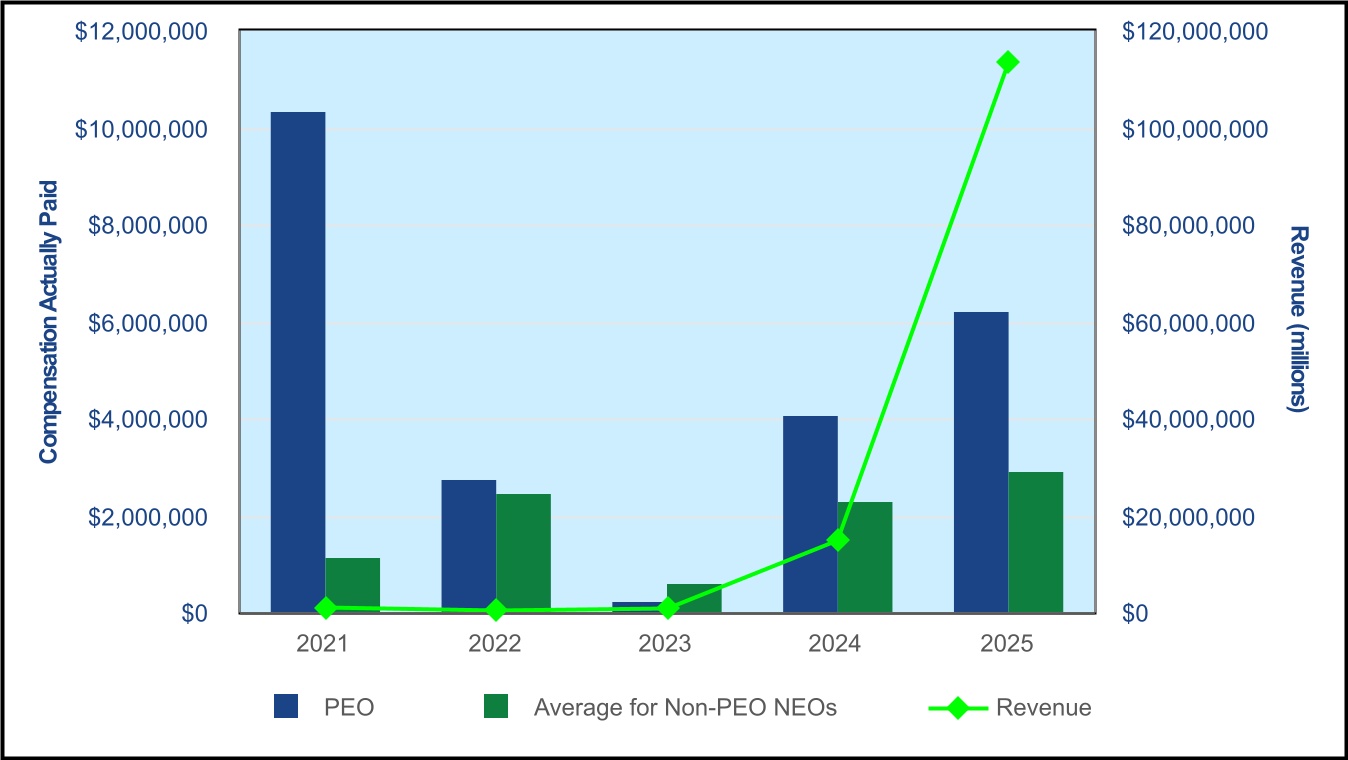
When ANKTIVA® received FDA approval in April 2024, we set an ambitious goal to demonstrate that our first-in-class IL-15 immunotherapy could become a new standard-of-care for non-muscle invasive bladder cancer patients with CIS with or without papillary disease (NMIBC). The results speak for themselves. Full-year 2025 net product revenue reached approximately $113 million, a 700% increase over the prior year. Unit sales grew 750%. Today, commercial and governmental insurance programs covering more than 240 million lives provide access to ANKTIVA.
The implementation of our permanent J-code (J9028) in January 2025 simplified reimbursement and accelerated adoption. But the true driver of our commercial success has been clinical outcomes. NMIBC CIS patients are achieving durable complete responses measured in years, not months. ANKTIVA is changing the treatment paradigm.
Global Expansion
Our regulatory momentum continued this period. In NMIBC, the UK’s MHRA approved ANKTIVA in July 2025, and in February 2026 the EC issued a conditional marketing authorization covering the EU as well as Iceland, Liechtenstein, and Norway. A significant milestone, however, came from the SFDA, which granted accelerated approval for ANKTIVA in metastatic non-small cell lung cancer in combination with checkpoint inhibitors for adult patients whose disease has progressed after standard therapy. This is the first regulatory approval for this indication anywhere in the world.
We view this lung cancer approval as validation of the core thesis behind our QUILT clinical trials: that ANKTIVA restores immune competence. In checkpoint-refractory patients who had exhausted standard options, ANKTIVA rescued lymphocyte populations and extended median overall survival to 21 months in patients who achieved higher immune competence, nearly three times historical outcomes.
Advancing the Science
The scientific rationale underlying our approach has never been stronger. Every cancer patient receives a complete blood count, yet the absolute lymphocyte count, a powerful predictor of outcomes, has been largely overlooked by oncology. Standard treatments including chemotherapy, radiation, and even some immunotherapies deplete the very immune cells needed to fight cancer. ANKTIVA is designed to help address this fundamental problem. Our RMAT designation for the reversal of lymphopenia in pancreatic cancer, our FDA Expanded Access authorization for solid tumors, and our Phase 3 NSCLC trial all advance a single hypothesis: restore the immune system, and patients survive longer.
Our off-the-shelf CAR-NK cell therapy platform reached another milestone this year with durable complete responses in Waldenström lymphoma. It is the first chemotherapy-free, lymphodepletion-free CAR cell therapy to demonstrate 100% disease control in this population, and it is administered entirely in the outpatient setting.
Helping Solve the BCG Shortage
For 14 years, a BCG shortage has left tens of thousands of bladder cancer patients without adequate treatment. Through our partnership with Serum Institute of India, we secured FDA authorization for an Expanded Access Program providing recombinant BCG to urologists nationwide. Shipments began in the first quarter of 2025. We continue to work with the FDA on pathways to make BCG permanently available.
Looking Ahead
As we enter 2026, our pipeline continues to advance toward registration across multiple indications. Our BCG-naïve trial has fully enrolled with interim results showing statistically significant improvements. Our glioblastoma registration trial is underway. Our Phase 3 NSCLC trial with BeiGene is enrolling patients. We remain committed to our mission: activating the patient’s natural immune defenses to achieve durable, lasting responses against cancer and infectious diseases. We call this Immunotherapy 2.0, and the results are validating our vision. Thank you for your continued support and confidence in ImmunityBio.
Sincerely,
Dr. Patrick Soon-Shiong, Executive Chairman and Global Chief Scientific and Medical Officer
Richard Adcock, President and Chief Executive Officer
Notice of Annual Meeting of Stockholders
| | | | | |
| Date and Time: | Tuesday, June 9, 2026 at 10:30 a.m. Pacific Time. |
| |
Internet Availability of
Proxy Materials: | Under rules adopted by the SEC, we may furnish proxy materials, including this proxy statement and our Annual Report by providing access to such documents on the Internet instead of mailing printed copies. Most of you will not receive printed copies of the proxy materials unless you request them. Instead, the Notice of Internet Availability of Proxy Materials, which is being mailed to stockholders on or about April 30, 2026, will instruct you as to how you may access and review all of the proxy materials on the Internet. The Notice of Internet Availability of Proxy Materials also instructs you as to how you may submit your proxy on the Internet. If you would like to receive printed copies of our proxy materials by mail, please follow the instructions in the Notice of Internet Availability of Proxy Materials for requesting such materials. If you request printed copies of the proxy materials by mail, the materials you receive will include the proxy card or voting instruction form for the Annual Meeting. A copy of our proxy statement and our Annual Report are also posted on proxyvote.com, and are available from the SEC on its website at sec.gov. |
| |
| Webcast Address: | virtualshareholdermeeting.com/IBRX2026 |
| |
| Items of Business: | 1.To elect nine directors from the nominees named in the accompanying proxy statement to serve until our 2027 annual meeting of stockholders and until their respective successors are duly elected and qualified or until their earlier resignation or removal. 2.To ratify the selection of Deloitte & Touche LLP as ImmunityBio’s independent registered public accounting firm for the fiscal year ending December 31, 2026. 3.To transact other business that may properly come before the Annual Meeting or any continuation, postponement, or adjournment thereof. |
| |
| Record Date: | Only stockholders of record of our common stock as of the close of business on Monday, April 13, 2026, are entitled to receive notice of, and vote at, the Annual Meeting and any continuation, postponement, or adjournment thereof. |
| |
| Meeting Admission: | You are invited to attend the Annual Meeting via Internet Webcast if you are a stockholder of record or a beneficial owner of shares of our common stock, in each case, as of April 13, 2026. You will be able to attend the Annual Meeting via Internet Webcast at virtualshareholdermeeting.com/IBRX2026 by entering the 16-digit Control Number included in our Notice of Internet Availability of Proxy Materials, on your proxy card, or in the instructions that accompanied your proxy materials. |
| | |
| By Order of the Board of Directors, |
|
|
|
| Philip LoScalzo |
| Corporate Secretary |
San Diego, California
April 29, 2026
3530 John Hopkins Court
San Diego, California 92121
PROXY STATEMENT
The information provided in the “Questions and Answers” format below is for your convenience only and is merely a summary of the information contained in this proxy statement. You should read the entire proxy statement carefully.
Questions and Answers about the Proxy Materials and Annual Meeting
Why am I receiving these materials?
This proxy statement and the enclosed form of proxy are posted in connection with the solicitation of proxies by the company’s board of directors (the Board) for use at the 2026 Annual Meeting of Stockholders of ImmunityBio, Inc. (the Annual Meeting), a Delaware corporation, and any postponements or adjournments thereof. The Annual Meeting will be held on Tuesday, June 9, 2026 at 10:30 a.m. Pacific Time, via Internet Webcast.
Stockholders are invited to attend the Annual Meeting via Internet Webcast and are requested to vote on the items of business described in this proxy statement. A copy of this proxy statement and our 2025 Annual Report are posted on proxyvote.com, and are also available from the SEC on its website at sec.gov. The Notice of Internet Availability of Proxy Materials, which is being mailed to stockholders on or about April 30, 2026, will instruct you as to how you may access and review all of the proxy materials on the Internet.
What is a proxy?
A proxy is your legal designation of another person to vote the stock you own. The person you designate is your “proxy,” and you give the proxy authority to vote your shares by submitting your proxy card or, if available, voting by telephone or the Internet. We have designated each of Richard Adcock, our President and Chief Executive Officer (CEO), Philip LoScalzo, our General Counsel and Corporate Secretary, and David Sachs, our Chief Financial Officer (CFO), to serve as proxies for the Annual Meeting.
What am I voting on?
You are being asked to vote on two proposals:
•The election of nine directors from the nominees named in this proxy statement to serve until our 2027 annual meeting of stockholders and until their respective successors are duly elected and qualified or until their earlier resignation or removal; and
•The ratification of the selection of Deloitte & Touche LLP as ImmunityBio’s independent registered public accounting firm for the fiscal year ending December 31, 2026.
What if other matters are properly brought before the Annual Meeting?
As of the date of this proxy statement, we are not aware of any other matters that will be presented for consideration at the Annual Meeting. If any other matters are properly brought before the Annual Meeting, the persons named as proxies will be authorized to vote or otherwise act on those matters in accordance with their judgment. If, for any reason, a director nominee is not available as a candidate for director, the persons named as proxy holders will vote your proxy for such other candidate as may be nominated by the Board.
ImmunityBio, Inc. | 1 | 2026 Proxy Statement
How does the Board recommend that I vote?
The Board recommends that you vote your shares:
•“FOR” the election of each of the nine nominees for director named in this proxy statement; and
•“FOR” the ratification of the selection of Deloitte & Touche LLP as ImmunityBio’s independent registered public accounting firm for the fiscal year ending December 31, 2026.
Who may vote at the Annual Meeting?
Only stockholders of record as of the close of business as of the Record Date on April 13, 2026 are entitled to vote at the Annual Meeting. As of the Record Date, there were 1,047,345,861 shares of our common stock outstanding, held by 65 holders of record.
You are invited to attend the Annual Meeting and vote your shares via Internet Webcast at virtualshareholdermeeting.com/IBRX2026 by entering the 16-digit Control Number included in our Notice of Internet Availability of Proxy Materials, on your proxy card, or in the instructions that accompanied your proxy materials.
Stockholder of Record: Shares Registered in Your Name. If, at the close of business on the Record Date, your shares were registered directly in your name with our transfer agent, Equiniti Trust Company, LLC, then you are a stockholder of record. As a stockholder of record, you have the right to grant your proxy directly to the individuals listed on the proxy card or to vote via Internet Webcast.
Beneficial Owner: Shares Registered in the Name of a Broker, Bank or Other Nominee. If, at the close of business on the Record Date your shares were held, not in your name, but rather in an account at a brokerage firm, bank or other nominee, then you are the beneficial owner of shares held in “street name” and these proxy materials will be forwarded to you by that organization. The organization holding your account is considered the stockholder of record for purposes of voting at the Annual Meeting. As a beneficial owner, you have the right to direct your broker, bank or other nominee regarding how to vote the shares in your account by following the voting instructions your broker, bank or other nominee provides or vote via Internet Webcast.
How can I vote my shares?
Stockholder of Record: Shares Registered in Your Name. If you are a stockholder of record, you may vote in one of the following ways:
•You may vote during the Annual Meeting live via the Internet. If you plan to attend the Annual Meeting via Internet Webcast, you may vote your shares via Internet Webcast at virtualshareholdermeeting.com/IBRX2026 by entering the 16-digit Control Number found in your proxy materials.
•You may vote by mail. Complete, sign and date your proxy card and return it promptly in the postage-prepaid envelope provided (if you requested and received printed proxy materials). Your completed, signed and dated proxy card must be received prior to the Annual Meeting.
•You may vote by telephone. To vote over the telephone, dial toll-free 800-690-6903 and follow the recorded instructions. You will be asked to provide the 16-digit Control Number found in your proxy materials. Telephone voting is available 24 hours a day, 7 days a week. Please note that telephone voting closes at 11:59 p.m. Eastern Time on June 8, 2026.
•You may vote via the Internet. To vote via the Internet, go to proxyvote.com to complete an electronic proxy card. You will be asked to provide the 16-digit Control Number found in your proxy materials. Internet voting is available 24 hours a day, 7 days a week. Please note that Internet voting closes at 11:59 p.m. Eastern Time on June 8, 2026.
ImmunityBio, Inc. | 2 | 2026 Proxy Statement
Beneficial Owner: Shares Registered in the Name of a Broker, Bank or Other Nominee. If you are a beneficial owner of shares held of record by a broker, bank or other nominee, you will receive voting instructions from your broker, bank or other nominee. You must follow the voting instructions provided by your broker, bank or other nominee in order to instruct your broker, bank or other nominee on how to vote your shares. Beneficial owners of shares should generally be able to vote by returning the voting instruction form, or by telephone or via the Internet. However, the availability of telephone or Internet voting will depend on the voting process of your broker, bank or other nominee. You may also vote your shares via Internet Webcast at virtualshareholdermeeting.com/IBRX2026 by entering the 16-digit Control Number found in your proxy materials.
Can I change my vote or revoke my proxy?
Stockholder of Record: Shares Registered in Your Name. If you are a stockholder of record, you can change your vote or revoke your proxy at any time before the Annual Meeting by:
•Entering a new vote via the Internet or by telephone (until the applicable deadline for each method as set forth above);
•Returning a new proxy card bearing a later date (which automatically revokes the earlier proxy);
•Providing a written notice of revocation to ImmunityBio, Inc., Attention: Corporate Secretary, 3530 John Hopkins Court, San Diego, California 92121 that is received before the polls close for voting at the Annual Meeting or any adjournment or postponement thereof; or
•Attending the Annual Meeting and submitting your vote live via the Internet prior to the closing of the polls (which would automatically revoke the previously submitted proxy). If you are a stockholder of record, you cannot change your vote in advance of the Annual Meeting using the Internet or telephone voting methods after 11:59 p.m. Eastern Time on June 8, 2026.
Beneficial Owner: Shares Registered in the Name of a Broker, Bank or Other Nominee. If you are a beneficial owner of your shares, you may change your vote by timely submitting new voting instructions to your broker, bank or other nominee (which would revoke your earlier instructions), or by attending the Annual Meeting and submitting your vote live via the Internet prior to the closing of the polls (which would revoke the previously submitted instructions). Beneficial owners of shares should follow the voting instructions provided by their broker, bank or other nominee.
Attending the Annual Meeting will not, in itself, cause your previously granted proxy or previously submitted voting instructions to be revoked. You must submit a new vote during the Annual Meeting prior to the closing of the polls in order to do so.
What is the effect of giving a proxy?
Proxies are solicited by and on behalf of the Board. The persons named in the proxy, Richard Adcock, Philip LoScalzo, and David Sachs, have been designated as proxies for the Annual Meeting by the Board. When proxies are properly dated, executed and returned, the shares represented by such proxies will be voted at the Annual Meeting in accordance with the instruction of the stockholder. If no specific instructions are given, however, the shares will be voted in accordance with the recommendations of the Board as described above and, if any other matters are properly brought before the Annual Meeting, the shares will be voted in accordance with the proxies’ judgment.
How many votes do I have?
For the election of directors, each stockholder is entitled to cast one vote for each director nominee for each share of the company’s common stock held by them as of the Record Date. Cumulative voting is not permitted for the election of directors. On the other matters to be voted upon, each stockholder is entitled to one vote for each share of the company’s common stock held by them as of the Record Date.
ImmunityBio, Inc. | 3 | 2026 Proxy Statement
What is the quorum requirement for the Annual Meeting?
A quorum is the minimum number of shares required to be present or represented at the Annual Meeting for the meeting to be properly held under the company’s Amended and Restated Bylaws (the Bylaws) and Delaware law. Holders of a majority of the voting power of the company’s issued and outstanding common stock entitled to vote at the Annual Meeting must be present live via the Internet or represented by proxy to hold and transact business at the Annual Meeting. On the Record Date, there were 1,047,345,861 shares issued and outstanding and entitled to vote. Thus, the holders of at least 523,672,931 shares must be present in person or represented by proxy at the Annual Meeting to have a quorum.
Abstentions, “withhold” votes, and “broker non-votes” are counted as present and entitled to vote for purposes of determining a quorum. If there is no quorum, the chair of the meeting or the holders of a majority of the voting power present live via the Internet or represented by proxy at the Annual Meeting and entitled to vote at the Annual Meeting may adjourn the meeting to another date.
What are broker non-votes?
Broker non-votes occur when a beneficial owner of shares held in street name does not give instructions to the broker holding the shares as to how to vote on matters deemed “non-routine” and there is at least one “routine” matter to be voted upon at the meeting. Generally, if shares are held in street name, the beneficial owner of the shares is entitled to give voting instructions to the broker holding the shares. If the beneficial owner does not provide voting instructions, the broker can still vote the shares with respect to matters that are considered to be routine, but not with respect to non-routine matters. In the event that a broker votes shares on the routine matters but does not vote shares on the non-routine matters, those shares will be treated as broker non-votes with respect to the non-routine proposals. Accordingly, if you own shares through a nominee, such as a broker or bank, please be sure to instruct your nominee how to vote to ensure that your vote is counted on each of the proposals or vote your shares at the meeting.
What matters are considered “routine” and “non-routine”?
The ratification of the selection of Deloitte & Touche LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2026 (Proposal 2) is considered routine under applicable federal securities rules. The election of directors (Proposal 1) is considered non-routine under applicable rules.
What are the effects of abstentions and broker non-votes?
An abstention represents a stockholder’s affirmative choice to decline to vote on a proposal. If a stockholder indicates on its proxy card that it wishes to abstain from voting its shares, or if a broker, bank or other nominee holding its customers’ shares of record cause abstentions to be recorded for shares, these shares will be considered present and entitled to vote at the Annual Meeting. As a result, abstentions will be counted for purposes of determining the presence or absence of a quorum and will also count as votes against a proposal in cases where approval of the proposal requires the affirmative vote of a majority of the shares present live via the Internet or represented by proxy and entitled to vote at the Annual Meeting, including Proposal 2. However, because the outcome of Proposal 1 (election of directors) will be determined by a plurality vote, abstentions will have no impact on the outcome of such proposal as long as a quorum exists.
Broker non-votes will be counted for purposes of calculating whether a quorum is present at the Annual Meeting but will not be counted for purposes of determining the number of votes cast on a non-routine proposal. Therefore, a broker non-vote will make a quorum more readily attainable but will not otherwise affect the outcome of the vote on any non-routine proposal.
What is the voting requirement to approve each of the proposals?
Proposal 1: Election of Directors. The election of directors requires a plurality of the voting power of shares present live via the Internet or represented by proxy at the Annual Meeting and entitled to vote on the election of directors. This means that the nine nominees for director receiving the highest number of “FOR” votes will be elected as directors. You may vote (i) “FOR” each director or (ii) “WITHHOLD” for each director nominee. Because the outcome of this proposal will be determined by a plurality vote, shares voted “WITHHOLD” will have no impact on the outcome of this proposal but will count towards the quorum requirement for the Annual Meeting.
ImmunityBio, Inc. | 4 | 2026 Proxy Statement
Proposal 2: Ratification of Selection of Deloitte & Touche LLP. The ratification of the selection of Deloitte & Touche LLP requires the affirmative vote of a majority of the shares present live via the Internet or represented by proxy at the Annual Meeting and entitled to vote thereon to be approved. You may vote “FOR,” “AGAINST,” or “ABSTAIN” on this proposal. Abstentions will count towards the quorum requirement for the Annual Meeting and will have the same effect as a vote against the proposal.
Dr. Patrick Soon-Shiong, our Founder, Executive Chairman and Global Chief Scientific and Medical Officer, and his affiliates owned, in the aggregate, approximately 62.5% of the company’s outstanding common stock as of the Record Date and has advised us that he intends to vote (1) “FOR” the election of each of the nine nominees for director named in this proxy statement; and (2) “FOR” the ratification of the selection of Deloitte & Touche LLP as ImmunityBio’s independent registered public accounting firm for the fiscal year ending December 31, 2026.
What is the interest of the company’s executive officers and directors in the proposals being voted upon?
None of our executive officers or directors or their associates has any substantial interest in any matter to be acted upon, other than our directors, with respect to the election to office of the directors so nominated because their compensation could be impacted.
What if I do not specify how my shares are to be voted or fail to provide timely directions to my broker, bank or other nominee?
Stockholder of Record: Shares Registered in Your Name. If you are a stockholder of record and you submit a signed proxy but you do not provide voting instructions, your shares will be voted:
•“FOR” the election of each of the nine nominees for director named in this proxy statement; and
•“FOR” the ratification of the selection of Deloitte & Touche LLP as ImmunityBio’s independent registered public accounting firm for the fiscal year ending December 31, 2026.
In addition, if any other matters are properly brought before the Annual Meeting, the persons named as proxies will be authorized to vote or otherwise act on those matters in accordance with their judgment.
Beneficial Owner: Shares Registered in the Name of a Broker, Bank or Other Nominee. Brokers, banks and other nominees holding shares of common stock in street name for customers are generally required to vote such shares in the manner directed by their customers. In the absence of timely directions, your broker, bank or other nominee will have discretion to vote your shares on our sole routine matter – Proposal 2 to ratify the selection of Deloitte & Touche LLP. Absent direction from you, however, your broker, bank or other nominee will not have the discretion to vote on Proposal 1 relating to the election of directors.
How can I contact ImmunityBio’s transfer agent?
You may contact our transfer agent by writing to Equiniti Trust Company, LLC, Attn: EQ-Automated Scanning Team, 1110 Centre Point Curve, Suite 101, Mendota Heights, MN 55120-4100. You may also contact our transfer agent via email at HelpAST@equiniti.com or by telephone Monday through Friday from 8:00 a.m. to 8:00 p.m. Eastern Time at 800.937.5449 (Toll Free) or 718.921.8124 (Local & International).
How can I attend the virtual Annual Meeting?
We will host the Annual Meeting online. You will not be able to attend the Annual Meeting in person. A summary of the information you need in order to attend the Annual Meeting online is provided below:
•Any stockholder can attend the Annual Meeting by visiting virtualshareholdermeeting.com/IBRX2026.
•The Annual Meeting starts at 10:30 a.m. Pacific Time on Tuesday, June 9, 2026.
•We encourage you to access the Annual Meeting online prior to its start time.
•Stockholders may vote electronically online while attending the Annual Meeting.
ImmunityBio, Inc. | 5 | 2026 Proxy Statement
•Please have the Control Number we have provided you to join the Annual Meeting.
•Instructions on how to attend and participate at the Annual Meeting are available at virtualshareholdermeeting.com/IBRX2026.
•If a stockholder wishes to ask a question of directors and/or members of management in attendance at the Annual Meeting, please note that such questions must be submitted in advance of the Annual Meeting. You may submit your question at proxyvote.com on or before 5:00 p.m. Pacific Time, June 5, 2026 by clicking on the “Submit a Question to Management” button. You will need your 16-digit Control Number to access the website to submit your question.
•Stockholders may vote during the Annual Meeting Internet Webcast at virtualshareholdermeeting.com/IBRX2026.
Who will count the votes?
A representative of Broadridge Investor Communications, Inc. will tabulate the votes and act as inspector of election.
What happens if the Annual Meeting is adjourned or postponed?
Your proxy will still be effective and will be voted at the rescheduled or adjourned Annual Meeting. You will still be able to change or revoke your proxy until the rescheduled or adjourned Annual Meeting.
Who is paying for the costs of this proxy solicitation?
Your proxy is being solicited by and on behalf of the Board. The expense of preparing, printing and providing this proxy solicitation will be borne by the company. We do not plan to retain a proxy solicitor to assist in the solicitation of proxies. Certain directors, officers, representatives and employees of the company may solicit proxies by telephone and personal interview. Such individuals will not receive any additional compensation from the company for solicitation of proxies, but may be reimbursed by the company for reasonable out-of-pocket expenses in connection with such solicitation. In accordance with the regulations of the SEC, banks, brokers and other custodians, nominees and fiduciaries also will be reimbursed by the company, as necessary, for their reasonable expenses for sending proxy solicitation materials to the beneficial owners of common stock.
Copies of the proxy materials will be supplied to brokers and other nominees for the purpose of soliciting proxies from beneficial owners, and we will reimburse such brokers or other nominees for their reasonable expenses.
If you choose to access the proxy materials and/or vote via the Internet, you are responsible for Internet access charges you may incur. If you choose to vote by telephone, you are responsible for telephone charges you may incur.
How can I find the results of the Annual Meeting?
Preliminary results will be announced at the Annual Meeting. Final results will be published in a Current Report on Form 8-K to be filed with the SEC within four business days after the Annual Meeting. If the official results are not available at that time, we will provide preliminary voting results in the Form 8-K and will provide the final results in an amendment to the Form 8-K as soon as they become available.
When are stockholder proposals due for next year’s annual meeting?
ImmunityBio, Inc. | 6 | 2026 Proxy Statement
| | |
Proposal 1 – Election of Directors |
Pursuant to the company’s Restated Certificate of Incorporation (the Certificate of Incorporation) and the company’s Bylaws, the total number of directors constituting the Board shall be fixed exclusively by the Board. The Board is currently comprised of nine directors, as listed below. Based upon the recommendation of the Nominating and Corporate Governance Committee (the Governance Committee), the Board has resolved that the number of directors constituting the entire Board remain at nine, and has nominated each of the company’s current directors set forth below for election at the Annual Meeting. At the Annual Meeting, nine directors will be elected to the Board by holders of the company’s common stock to serve for a one year term expiring at the 2027 annual meeting of stockholders. Each director’s term continues until the election and qualification of his or her successor, or until his or her earlier retirement, resignation, disqualification, removal, or death.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | Committee Composition |
| Name | | Independent | | Age | | Board
Tenure
(in Years) | | Audit | | Compensation | | Nominating
and
Corporate
Governance | | Related
Party
Transaction | | Special |
| | | | | | | | | | | | | | | | |
Patrick Soon-Shiong, M.D.,
FRCS (C), FACS | | | | 73 | | 11 | | | | | | C | | | | |
| Cheryl L. Cohen | | ü | | 60 | | 6 | | M | | | | M | | | | M |
| Richard Adcock | | | | 57 | | 5 | | | | | | | | | | |
| Michael D. Blaszyk | | ü | | 73 | | 10 | | C | | M | | | | | | C |
| Wesley Clark | | ü | | 81 | | 5 | | | | | | M | | C | | |
Linda Maxwell, M.D.,
M.B.A., FRCSC | | ü | | 52 | | 5 | | M | | C | | | | M | | |
| Christobel Selecky | | ü | | 71 | | 5 | | M | | M | | | | M | | |
| Barry J. Simon, M.D. | | | | 61 | | 19 | | | | | | | | | | |
Bruce Wendel (1) | | ü | | 72 | | — | | | | | | | | M | | |
_______________
“C” Indicates Chair of the committee.
“M” Indicates member of the committee.
(1)Mr. Wendel was appointed to the Board on December 12, 2025 and appointed to the Related Party Transaction Committee on April 29, 2026.
The Board, upon the recommendation of the Governance Committee, has nominated Dr. Soon-Shiong, Ms. Cohen, Messrs. Adcock and Blaszyk, Gen. Clark, Dr. Maxwell, Ms. Selecky, Dr. Simon, and Mr. Wendel, each a current director, as nominees for re-election to the Board at the Annual Meeting. If elected, each of the aforementioned nominees will serve as a director until the 2027 annual meeting and until his or her respective successor is duly elected and qualified.
Each of our nominees for director has agreed to serve, if elected, and management has no reason to believe that they will be unavailable to serve. In the event a nominee is unable or declines to serve as a director at the time of the Annual Meeting, proxies will be voted for any nominee who may be designated by the present Board to fill the vacancy.
Required Vote
The directors elected to the Board will be elected by a plurality of the voting power of shares present live via the Internet or represented by proxy and entitled to vote on the election of directors. In other words, the nine nominees receiving the highest number of “FOR” votes will be elected as directors. Shares represented by executed proxies will be voted, if authority to do so is not expressly withheld, for the election of Dr. Patrick Soon-Shiong, Cheryl L. Cohen, Richard Adcock, Michael D. Blaszyk, Gen. Wesley Clark, Dr. Linda Maxwell, Christobel Selecky, Dr. Barry J. Simon, and Bruce Wendel. Broker non-votes have no effect on this proposal.
| | | | | | | | | | | | | | |
THE BOARD RECOMMENDS A VOTE “FOR” EACH OF THE NINE NOMINEES. PROXIES WILL BE VOTED “FOR” THE ELECTION OF THE NOMINEES UNLESS OTHERWISE SPECIFIED. |
ImmunityBio, Inc. | 7 | 2026 Proxy Statement
Set forth below is biographical information for each nominee and a summary of the specific qualifications, attributes, skills, and experiences which led the Board to conclude that each nominee should serve on the Board at this time. There are no family relationships among any of our directors or among any of our directors and our executive officers.
| | | | | |
| Patrick Soon-Shiong, M.D., FRCS (C), FACS | Executive Chairman of the Board |
| Dr. Soon-Shiong is the Founder of ImmunityBio, Inc. He was appointed Executive Chairman of the Board in October 2020 and Global Chief Scientific and Medical Officer of the company on August 11, 2021. Previously, he served as the Chairman of the Board and Chief Executive Officer from March 2015 to October 2020, as the Co-Chairman of the Board from December 2014 to March 2015, and as our Chief Medical Officer from January 2015 to March 2015. In 2011, he founded NantWorks, LLC (NantWorks), an ecosystem of companies to create a transformative global health information and next generation pharmaceutical development network, for the secure sharing of genetic and medical information. Dr. Soon-Shiong invented and developed Abraxane®, the nation’s first Food and Drug Administration (FDA)-approved protein nanoparticle albumin-bound delivery technology for the treatment of cancer. Abraxane was approved by the FDA for metastatic breast cancer in 2005, lung cancer in 2012, and pancreatic cancer in 2013. Abraxane is now approved in many countries across the globe. From 1997 to 2010, Dr. Soon-Shiong served as founder, chairman, and chief executive officer of two global pharmaceutical companies, American Pharmaceutical Partners (sold to Fresenius SE for $4.6 billion in 2008) and Abraxis BioScience (sold to Celgene Corporation for $3.8 billion in 2010). In 2018, he became the owner and executive chairman of the Los Angeles Times, Los Angeles Times en Español and other publications under the California Times. Dr. Soon-Shiong is chairman of the Chan Soon-Shiong Family Foundation and the Chan Soon-Shiong Institute of Molecular Medicine, a nonprofit medical research organization. He also serves as chair of the board of directors of the Access to Advanced Health Institute, a nonprofit biotech research institute located in Seattle focused on combating some of the world’s deadliest diseases, including COVID-19, cancer, fungal and parasitic infections, and other non-communicable diseases. He is a visiting Professor at the Imperial College of London. Dr. Soon-Shiong holds a degree in medicine from the University of the Witwatersrand and a M.Sc. in science from the University of British Columbia. Qualifications We believe Dr. Soon-Shiong is qualified to serve as a member of the Board based on his depth of expertise as chairman and chief executive officer of multiple multi-billion dollar companies in the life sciences industry, his broad experience in the research and development of pioneering technologies, and his educational background. Pursuant to the Subscription and Investment Agreement between the company and Cambridge Equities, LP (Cambridge), Cambridge has the right to designate one individual to be nominated and recommended for election by the Board for as long as Cambridge continues to hold at least 20% of the issued and outstanding shares of the company’s common stock. Dr. Soon-Shiong has been selected by Cambridge to hold this Board seat. |
Director since: 2014 Committee: •Nominating and Corporate Governance (Chair) |
ImmunityBio, Inc. | 8 | 2026 Proxy Statement
| | | | | |
| Cheryl L. Cohen | Lead Independent Director |
| Ms. Cohen was appointed Lead Independent Director in March 2023 and has served as a member of the Board since June 2019. Since 2014, she has served as principal owner and president of CLC Consulting, LLC (CLC), a pharmaceutical and biotechnology consulting firm specializing in new product start-up and commercialization. Prior to CLC, she served as chief commercial officer of Medivation, Inc., a publicly traded bio-pharmaceutical company, from 2011 until 2014. From 2007 to 2008, she served as Vice President, Strategic Commercial Group, of Health Care Systems, Inc., a Johnson & Johnson company, and from 1998 to 2007, she worked at Janssen Biotech, Inc. (formerly Centocor Biotech, Inc.), a Johnson & Johnson company, in a variety of senior sales roles including Vice President, Rheumatology Franchise. Ms. Cohen has served on the board of directors of Celldex Therapeutics, Inc. (Nasdaq:CLDX) since June 2022. She previously served on the board of directors of MEI Pharma, Inc. (Nasdaq:MEIP), Ignyte Acquisition, Corp. (Nasdaq:IGNY), Aerpio Pharmaceuticals, Inc., Eledon Pharmaceuticals, Inc. (formerly known as Novus Therapeutics, Inc.) (Nasdaq:ELDN), Vital Therapies, Inc., Protein Sciences Corporation, and LadRx Corporation (formerly CytRx Corporation (OTCQB:LADX)). She began her career at Solvay Pharmaceuticals in a variety of sales positions. Ms. Cohen received her B.A. from Saint Joseph College. Qualifications We believe Ms. Cohen is qualified to serve as a member of the Board based on her extensive experience with and knowledge of the healthcare industry, commercialization expertise, and experience serving on boards of directors of public companies. |
Director since: 2019 Committees: •Audit •Nominating and Corporate Governance •Special Other Public Company Board: •Celldex Therapeutics
|
| | | | | |
| Richard Adcock | Director |
| Mr. Adcock has served as our CEO since October 2020 and as President of the company and a member of the Board since March 2021. He has over 30 years of leadership experience in the healthcare industry. From January 2018 to September 2020, he served as chief executive officer of Verity Health System of California, Inc. (Verity Health), a nonprofit public benefit corporation healthcare provider, that he steered through a successful restructuring event. Prior to joining Verity Health, he served in various capacities at Sanford Health, a not-for-profit integrated healthcare delivery system, including as its chief innovation officer, president, executive vice president and director from 2004 to 2017. Prior to Sanford Health, he served as global engineering director at GE Healthcare. Mr. Adcock holds a B.S. in Business Administration from Northern State University and an M.B.A. in Healthcare Management from University of Phoenix. Qualifications We believe Mr. Adcock is qualified to serve as a member of the Board based on his extensive knowledge of the healthcare industry and his experience in senior management roles at leading healthcare companies. |
Director since: 2021
|
ImmunityBio, Inc. | 9 | 2026 Proxy Statement
| | | | | |
Michael D. Blaszyk | Independent Director |
| Mr. Blaszyk has served as a member of the Board since July 2015. From July 2017 through April 2025, he served as an operating partner of Beecken Petty O'Keefe & Company, LLC (BPOC), a capital markets company. Prior to BPOC, he served as the senior executive vice president, chief financial officer and chief corporate officer of Dignity Health (formerly known as Catholic Healthcare West), a not-for-profit public benefit corporation, from December 2000 until his retirement in December 2015. Prior to joining Dignity Health, he served as the senior vice president and chief financial officer of University Hospitals Health System in Cleveland, Ohio, a healthcare system affiliated with Case Western University, from 1997 to 2000. He also previously served as the managing partner of the Northeast Region Health Care Provider Consulting Practice for Mercer LLC (formerly known as William M. Mercer), a global consulting firm, and the executive vice president of Boston Medical Center, a not-for-profit academic medical center. Mr. Blaszyk is a director/manager for NantHealth (a company affiliated with Dr. Soon-Shiong). He has served on numerous public and private company boards and continues to serve on charitable purpose organization boards. He received his B.S. in Life Sciences from Wayne State University and his Masters in Health Administration from the Leeds School of Business, University of Colorado. Qualifications We believe Mr. Blaszyk is qualified to serve as a member of the Board based on his extensive experience with and knowledge of the healthcare industry and his significant financial and accounting background. |
Director since: 2015 Committees: •Audit (Chair) •Compensation •Special (Chair) Audit Committee financial expert |
| | | | | |
Wesley Clark | Independent Director |
| General Clark, USA, Retired, has served as a member of the Board since March 2021. Gen. Clark previously served on the board of directors of NantCell, Inc. (formerly known as ImmunityBio, Inc., a private company) from February 2021 until the Merger in March 2021. He served for 34 years in the U.S. Army, rising through the ranks to earn his fourth star as a full general in 1996. He served as the Supreme Allied Commander Europe of NATO from 1997 to 2000, where he commanded Operation Allied Force in the Kosovo War. Highly decorated throughout his career, Gen. Clark was awarded the U.S. Presidential Medal of Freedom by President William J. Clinton. Since March 2003, he has served as chairman and chief executive officer of Wesley K. Clark & Associates, LLC, a strategic consulting firm specializing in business development, crisis support and strategic communications. Since 2010, he has served as chairman and chief executive officer of Enverra, Inc., a boutique investment bank. Gen. Clark has served on the board of directors of Blue Acquisition Corp. (NASD:BACC) since June 2025 and joined the board of directors of Blockfusion Data Centers, Inc. in March 2026. He previously served on the boards of directors of Equinox Gold Corp. from 2020 to 2023 and Rentech, Inc. from 2010 to 2018. He is a graduate of the U.S. Military Academy at West Point, where he was class valedictorian. After graduating from West Point, he was awarded a Rhodes Scholarship to the University of Oxford where he earned degrees in philosophy, politics and economics. He earned a Master’s degree in military science from the Command and General Staff College. Qualifications We believe Gen. Clark is qualified to serve as a member of the Board based on his extensive leadership experience, success in both the public and private sectors, and experience serving on other public company boards of directors. |
Director since: 2021 Committees: •Nominating and Corporate Governance •Related Party Transaction (Chair) Other Public Company Boards: •Directa Plus S.p.A. (Italy) •MCF Energy Ltd. (Canada) |
ImmunityBio, Inc. | 10 | 2026 Proxy Statement
| | | | | |
Linda Maxwell, M.D., M.B.A., FRCSC | Independent Director |
| Dr. Maxwell has served as a member of the Board since March 2021. She is an experienced physician and surgeon, having managed her own head and neck surgical practice since 2006. She is a medical educator, a published scientific author, and a health technology entrepreneur and innovator. As of March 2022, she is an Operating Partner at DCVC Management Co, LLC, a deep tech venture capital investment fund. She was previously an Adjunct Professor of Surgery at University of Toronto, a Distinguished Visiting Professor at Toronto Metropolitan University, and an Associate Scientist at the Li Ka Shing Knowledge Institute in Toronto. She is the Founder and former Executive Director of the Biomedical Zone at Toronto Metropolitan University, Canada’s premier hospital-embedded medical technology incubator for early-stage digital health and medical technology companies, and has guided a wide variety of startup companies through clinical development, capitalization and commercialization. She has also managed a life sciences tech transfer portfolio at the University of Oxford and the UK National Health Service, executing patent strategy, spin-out company formation, and early-stage capital raising. She has also served as a healthcare innovation expert to various Canadian federal, provincial, and local government entities, as a member of the Department Audit Committee of the Public Health Agency of Canada, and as an advisor to the Canadian Medical Association and the Canadian Space Agency. She currently serves on the board of directors of United Therapeutics Corporation (Nasdaq:UTHR). Dr. Maxwell earned an A.B. with honors from Harvard University, an M.D. from Yale University School of Medicine, and an M.B.A. from the Saïd Business School at the University of Oxford. Dr. Maxwell is a certified Independent Corporate Director in both the U.S. (NACD.DC) and Canada (ICD.D). Qualifications We believe Dr. Maxwell is qualified to serve as a member of the Board based on her extensive medical and scientific knowledge and experience, public company governance experience, and her experience advising and cultivating companies in the health technology industry. |
Director since: 2021 Committees: •Audit •Compensation (Chair) •Related Party Transaction Other Public Company Board: •United Therapeutics Corporation |
ImmunityBio, Inc. | 11 | 2026 Proxy Statement
| | | | | |
Christobel Selecky | Independent Director |
| Ms. Selecky has served as a member of the Board since March 2021. She served on the board of directors of NantCell, Inc. (formerly known as ImmunityBio, Inc., a private company) from August 2020 until the Merger in March 2021. Ms. Selecky has more than 35 years of healthcare industry experience. She is the principal at Population Health Strategies, her healthcare consultancy, where since 2009 she has provided strategic consulting to management teams, companies and investors, helping improve patient engagement, population health outcomes, and healthcare cost management. Since 2014 she has served as a Strategic Advisor at Ceresti Health, a privately-held healthcare technology company, and has served as a lecturer in Healthcare Entrepreneurship for the M.B.A. program at the University of California, Irvine since 2017. She held several leadership positions over her 14-year career at FHP International Corporation, which ended in 1995 including as President of the FHP California Health Plan. She subsequently co-founded, and served as President, CEO, and Executive Chairman of LifeMasters Supported SelfCare, a national leader in the field of disease and population health management. She currently serves on the boards of directors of Teleperformance SE (TEP – ISIN: FR0000051807 – Reuters: TEPRF.PA – Bloomberg: TEP FP), a French public company providing business services, Satellite Healthcare, a leading not-for-profit provider of kidney dialysis and related services, and Griswold Home Care, a privately-held non-medical home care company. She is active in several board governance organizations such as NACD (where she is a board member of the Pacific Southwest Chapter), the Private Directors Association, and Women Corporate Directors and has been a member of audit, compensation, governance and other committees of boards of directors on which she has served. Ms. Selecky received her B.A. from the University of Delaware and her M.A. from Syracuse University. Qualifications We believe Ms. Selecky is qualified to serve as a member of the Board based on her extensive experience in and knowledge of the healthcare industry, experience in board governance, financial expertise, and experience in advising stakeholders of healthcare companies at various stages of growth. |
Director since: 2021 Committees: •Audit •Compensation •Related Party Transaction Other Public Company Board: •Teleperformance SE (France) Audit Committee financial expert
|
ImmunityBio, Inc. | 12 | 2026 Proxy Statement
| | | | | |
Barry J. Simon, M.D. | Director |
| Dr. Simon has served as a member of the Board since 2007 and as Chief Corporate Affairs Officer of the company since March 2021. He previously served as our President and Chief Administrative Officer from January 2017 to March 2021, and as President and Chief Operating Officer from 2015 to 2016. From 2007 to 2015, he was our President and Chief Executive Officer. He has served as president, chief executive officer and chairman of Brink Biologics Inc., a bioanalytics, reagents and testing services company (a company affiliated with Dr. Soon-Shiong) since March 2015. Previously, he held vice president, senior level, and advisory positions at F. Hoffmann-La Roche, a global healthcare company, Roche Labs, a pharmaceuticals company, Connetics Corporation, a specialty pharmaceutical company, Immunomedics, a biopharmaceutical company, Immusol, a biopharmaceutical company, HealthPro BioVentures, LLC, a healthcare and life sciences investment bank, and NorthSound Capital, LLC, a U.S.-based hedge fund. He previously served as a director of Viracta Therapeutics, Inc., a biopharmaceutical company, from July 2017 to November 2020 and March 2021 to December 2024, and as a director of Cue Biopharma, Inc. (Nasdaq:CUE), a biopharmaceutical company, from 2016 to June 2021. He has broad experience in public and private settings, having led private and public equity offerings, product and portfolio divestitures and acquisitions, strategic licensing and joint ventures, as well as commercial product launches, investigational new drug (IND) and biologics license application (BLA) regulatory filings, human-enabling programs, manufacturing, quality control and life cycle management. He has worked across several therapeutic areas including oncology, virology, ophthalmology and dermatology on products launches including Xeloda®, Pegasys®, Fortovase®, Tamiflu®, Camptobell®, Boniva®, Fuzeon®, Valcyt®, and Accutane®. He attended corporate training programs through the London School of Business and Amos Tuck School of Business at Dartmouth College. Dr. Simon trained clinically in Infectious Diseases at Albert Einstein College of Medicine, Anesthesiology at The Mount Sinai Medical Center, and Internal Medicine at New York University and received his M.D. from the SUNY Downstate Health Sciences Center in New York. Qualifications We believe Dr. Simon is qualified to serve as a member of the Board based on his extensive medical and scientific knowledge and experience, and senior management experience in the biopharmaceutical industry. |
Director since: 2007
|
ImmunityBio, Inc. | 13 | 2026 Proxy Statement
| | | | | |
Bruce Wendel | Independent Director |
| Mr. Wendel has served as a member of the Board since December 2025. He served as Chief Business Development Officer for ProMetic Biosciences Ltd., a pharmaceutical development company, from April 2018 to May 2019. He also served as Chief Strategic Officer of Hepalink USA, the U.S. subsidiary of Shenzhen Hepalink Pharmaceutical Company, from July 2012 to July 2018 and as Chief Executive Officer of Scientific Protein Laboratories, LLC from December 2014 to June 2015. Prior to this, having served in roles of increasing responsibility at Abraxis BioScience, LLC, he was appointed Vice Chairman and Chief Executive Officer, overseeing the commercialization of Abraxane® and leading the negotiations that culminated in the acquisition of the company by Celgene Corporation in 2010. He began his 14 years at Bristol-Myers Squibb Company (NYSE:BMY) in 1988 as in-house counsel before shifting to global business and corporate development where he served in roles of increasing responsibility. Subsequently, he was Vice President of Business Development at IVAX Corporation, and at American Pharmaceutical Partners, Inc. From November 2020 to May 2025, he served on the board of directors of GT BioPharma, Inc. (Nasdaq:GTBP), and from June 2016 through May 2021, he served on the board of directors of Verastem, Inc. (Nasdaq:VSTM). He also served as a director of ProMetic Biosciences Ltd. He earned a juris doctor degree from Georgetown University Law School, and a B.S. from Cornell University. Qualifications We believe Mr. Wendel is qualified to serve as a member of the Board based on his extensive leadership experience, experience with and knowledge of the healthcare industry, and experience serving on boards of directors of public companies. |
Director since: 2025 Committee: •Related Party Transaction |
ImmunityBio, Inc. | 14 | 2026 Proxy Statement
Our business affairs are managed under the direction of the Board, which is currently comprised of nine members. Six of the nine directors qualify as “independent” within the meaning of such term as set forth in the listing standards of the Nasdaq Stock Exchange (Nasdaq). Under Nasdaq rules, a director will only qualify as an “independent director” if, among other things, in the opinion of that company’s board of directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
On June 18, 2015, we entered into a letter agreement with Cambridge (Cambridge Nominating Agreement). Pursuant to the Cambridge Nominating Agreement, Cambridge has the right to designate one individual to be nominated and recommended for election by the Board or a duly authorized committee thereof, for as long as Cambridge or its affiliates continues to hold at least 20% of the issued and outstanding shares of the company’s common stock, subject to adjustment for stock splits, stock dividends, recapitalizations and similar transactions. Dr. Soon-Shiong, our Founder, Executive Chairman and Global Chief Scientific and Medical Officer, controls the entity that is the general partner of Cambridge and was selected by Cambridge to hold this board seat. The Cambridge director nominee shall be nominated and recommended for election to the Board, subject to any applicable limitations imposed by Delaware General Corporation Law (DGCL), the Board’s fiduciary duties to our stockholders, and any other applicable law. Cambridge’s right to have a designee nominated or appointed to serve on the Board shall automatically terminate whenever Cambridge owns less than 20% of the company’s issued and outstanding shares of common stock.
Under the terms of his employment agreement, Dr. Barry J. Simon, the company’s Chief Corporate Affairs Officer, for so long as he remains an employee of the company, shall be nominated and recommended for election to the Board at each annual meeting of stockholders by the Board or a duly authorized committee thereof. If Dr. Simon’s employment with us is terminated for any reason, his membership on the Board will also terminate, unless otherwise agreed in writing by the company and Dr. Simon.
Pursuant to the company’s Certificate of Incorporation and Bylaws, the total number of directors constituting the Board shall be fixed exclusively by the Board. Vacancies and newly created directorships resulting from any increase in the authorized number of directors may be filled only by a majority of the directors then in office, although less than a quorum, or by a sole remaining director, and not by stockholders, and the directors so chosen will hold office until the next annual or special meeting of stockholders called for that purpose and until their successors are duly elected and qualified, or until their earlier resignation or removal.
The company’s Corporate Governance Guidelines require that at least a majority of the Board shall consist of United States citizens and/or directors residing in the United States.
At each annual meeting of stockholders, the terms of each of our nine incumbent directors expire and all members of the Board will stand for election. The nine directors elected at the annual meeting will serve from the time of election and qualification until the earliest of the next annual meeting of stockholders following such election or their resignation or removal.
| | | | | | | | | | | | | | |
| Board Leadership Structure |
As described below, the Board is led by Dr. Soon-Shiong, our Founder and Executive Chairman since October 2020, and Ms. Cohen, our Lead Independent Director since March 2023.
Lead Independent Director
Our Corporate Governance Guidelines provide that, in the event that the Board’s chairperson is our CEO, or if the Governance Committee and Board determine that it is in the best interest of the company and its stockholders, the Board will annually elect a Lead Independent Director upon the recommendation of the Governance Committee. The Lead Independent Director will have the following responsibilities: (i) maintain honest and transparent board culture; (ii) serve as a liaison between the Chairperson of the Board and the independent directors; (iii) facilitate discussion among the independent directors on key issues and concerns outside of Board meetings; (iv) ensure Board discussions
ImmunityBio, Inc. | 15 | 2026 Proxy Statement
demonstrate effective oversight of management; (v) facilitate teamwork and communication among the independent directors; (vi) organize and preside over executive sessions; (vii) set the agendas for and lead executive sessions; (viii) solicit feedback from and engage the CEO on executive session topics; (ix) act as a key advisor to the CEO on company matters where appropriate; (x) facilitate the Board’s engagement with the CEO and the Board’s CEO succession planning; (xi) has authority to call meetings of the independent directors; (xii) with the Chairperson, approve meeting agendas for the Board; (xiii) with the Chairperson, approve information and materials sent to the Board in advance of meetings; (xiv) with the Chairperson, approve Board meeting schedules and work with the Chairperson of the Board and Committee Chairs to assure that there is sufficient time for discussion of all agenda items, and ensuring that the independent directors can perform their duties responsibly while not interfering with on-going company operations; (xv) preside at all meetings of the Board at which the Chairperson of the Board is not present; (xvi) advise the Chairman on the retention of advisors and consultants who report directly to the Board; and (xvii) have such other duties as the Board may delegate to assist in meeting its responsibilities. All members of the Board are encouraged to communicate with the CEO and Chairperson.
Roles of Chairperson, Lead Independent Director, and Chief Executive Officer
Our Corporate Governance Guidelines provide that the Board will fill the chairperson and CEO positions based upon its view of what is in the best interests of the company at any point in time. The Board believes that Dr. Soon-Shiong’s service as Executive Chairman and Mr. Adcock’s service as CEO is in the best interests of the company and its stockholders.
Given his long tenure with and status within ImmunityBio, Inc., the Board believes Dr. Soon-Shiong possesses detailed and in-depth knowledge of the issues, opportunities, and challenges facing the company, and management believes he is best positioned, in consultation with Ms. Cohen and Mr. Adcock, to develop agendas that ensure that the Board’s time and attention is focused on the most critical matters. Management also believes that Dr. Soon-Shiong’s extensive scientific and medical knowledge and background with the company enables him to work with Ms. Cohen and Mr. Adcock to ensure clear accountability and enhance the company’s ability to communicate its message and strategy clearly and consistently to our stockholders and employees.
| | | | | | | | | | | | | | |
| The Board’s Role in Risk Oversight |
One of the key functions of the Board is informed oversight of the company’s risk management process. The Board does not have a standing risk management committee, but rather administers this oversight function directly through the Board as a whole, as well as through its standing committees that address risks inherent in their respective areas of oversight. In particular, the Board is responsible for monitoring and assessing strategic risk exposure. The Audit Committee is responsible for reviewing and discussing the company’s major financial and cybersecurity risk exposures and the steps taken by management to monitor and control these exposures, including guidelines and policies with respect to risk assessment and risk management. The Audit Committee also monitors compliance with legal and regulatory requirements, in addition to oversight of the performance of the company’s internal and external audit functions. The Related Party Transaction Committee reviews related-party transactions and informs the Audit Committee of all related-party transactions approved by them. The Governance Committee assists the Board in fulfilling its oversight responsibilities with respect to the management of risk associated with board organization, membership and structure, and corporate governance. The Compensation Committee of the Board (Compensation Committee) assesses and monitors whether any of the company’s compensation policies and programs has the potential to encourage excessive risk-taking. The Board believes its leadership structure is consistent with and supports the administration of its risk oversight function.
| | |
| Code of Global Business Conduct and Ethics |
The Board has adopted a written Code of Global Business Conduct and Ethics that applies to our directors, officers and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions and agents and representatives, including consultants. A copy of the Code of Global Business Conduct and Ethics is available on our website, ir.immunitybio.com under “Corporate Governance.” If we make any substantive amendments to, or grant any waivers from, the Code of Global Business Conduct and Ethics for any officer or director, we will disclose the nature of such amendment or waiver and provide a copy of any amendments on our website.
ImmunityBio, Inc. | 16 | 2026 Proxy Statement
During the fiscal year ended December 31, 2025, the Board held nine meetings (including regularly scheduled and special meetings), and each director attended at least 75% of the aggregate of (i) the total number of Board meetings held during the period for which he or she served as a director and (ii) the total number of Board committee meetings held during the periods that he or she served on such committee(s). Independent directors shall meet in executive session at each regularly scheduled meeting no less than four times per year.
We make every effort to schedule our annual meeting of stockholders at a date and time to maximize attendance by directors taking into account the directors’ schedules. It is the company’s policy that all current directors attend our annual meetings of stockholders, subject to extenuating circumstances. Each of our directors serving at the time of our 2025 annual meeting of stockholders were present at such meeting.
| | |
| Communication with the Board |
In cases where stockholders or other interested parties wish to communicate directly with our directors, messages can be sent to ImmunityBio, Inc., Attention: Corporate Secretary, 3530 John Hopkins Court, San Diego, California 92121. Our Corporate Secretary monitors these communications and will forward them to our designated legal counsel to provide a summary of all received messages to the Board at each regularly scheduled meeting. The Board typically meets on a quarterly basis. Where the nature of a communication warrants, our designated legal counsel may determine, in his or her judgment, to obtain the more immediate attention of the appropriate committee of the Board or non-management director, of independent advisors or of our management, as our designated legal counsel considers appropriate.
Our designated legal counsel may decide in the exercise of his or her judgment whether a response to any stockholder or interested party communication is necessary.
This procedure for stockholder and other interested party communications with the non-management directors is administered by the Governance Committee. This procedure does not apply to (a) communications to non-management directors from our officers or directors who are stockholders, (b) stockholder proposals submitted pursuant to Rule 14a-8 under the Securities Exchange Act of 1934, as amended (the Exchange Act), or (c) communications to the Audit Committee pursuant to our procedures for complaints regarding accounting and auditing matters.
| | |
| Board Committees and Charters |
The Board currently has four standing committees: the Audit Committee; the Compensation Committee; the Governance Committee; and the Related Party Transaction Committee. Additionally, in October 2020 the Board established a Special Committee in connection with the merger between NantKwest, Inc. and NantCell, Inc. (formerly known as ImmunityBio, Inc., a private company) (the Merger), which merger closed on March 9, 2021.
The Board maintains charters for each of its standing committees and these charters are evaluated annually. In addition, the Board has adopted a written set of Corporate Governance Guidelines that generally formalize practices the Board has in place. The charters of our standing Board committees and our Corporate Governance Guidelines are available on our website, ir.immunitybio.com under “Corporate Governance.”
Controlled Company Exemption
Dr. Soon-Shiong, our Founder, Executive Chairman and Global Chief Scientific and Medical Officer and entities affiliated with him, control a significant majority of our common stock. As a result, we are a “controlled company” within the meaning of the Nasdaq corporate governance rules. Under these rules, a company of which more than 50% of the voting power is held by an individual, group or another company is a “controlled company” and may elect not to comply with certain corporate governance standards, including (1) the requirement that a majority of the Board consist of independent directors, and (2) the requirement that the company have a nominating and corporate governance
ImmunityBio, Inc. | 17 | 2026 Proxy Statement
committee. Although not required by Nasdaq corporate governance rules, the Board is currently comprised of a majority of independent directors. The Board has determined that each of Ms. Cohen, Mr. Blaszyk, Gen. Clark, Dr. Maxwell, Ms. Selecky, and Mr. Wendel, representing six of our nine directors, is independent as that term is defined under Nasdaq rules. In addition, although not required by Nasdaq rules, the Board established a Governance Committee.
The Audit Committee was established to oversee our accounting and financial reporting processes and assist the Board in monitoring our financial systems and our legal and regulatory compliance. The committee also:
•Approves the appointment, compensation, retention, and, where appropriate, the replacement of the independent registered public accounting firm;
•Oversees and evaluates, at least annually, the work of the independent registered public accounting firm;
•Approves engagement of the independent registered public accounting firm to render any audit or permissible non-audit services;
•Reviews the qualifications, independence and performance of the independent registered public accounting firm;
•Reviews our consolidated financial statements and our critical accounting policies and estimates;
•Reviews the adequacy and effectiveness of our internal controls;
•Oversees our compliance with applicable law (including U.S. federal securities laws and other legal and regulatory requirements);
•Reviews our policies with respect to risk assessment and risk management;
•Reviews and monitors our policies and procedures relating to related-party transactions in consultation with the Related Party Transaction Committee;
•Reviews and discusses the results of our annual audit and reviews our quarterly consolidated financial statements and publicly filed reports with management and the independent registered public accounting firm;
•Reviews and recommends to the Board for approval our proposed budget;
•Reviews the risks relating to significant cybersecurity matters and concerns, including information security, data privacy, backup of information systems, and related regulatory matters and compliance; and
•Reviews a quarterly report from the Disclosure Committee regarding the adequacy of our disclosure controls and the effectiveness, activities, and recommendations of the Disclosure Committee.
The current members of the committee are Mr. Blaszyk (Chair), Ms. Cohen, Dr. Maxwell, and Ms. Selecky. The Board has determined that each member of the committee is an independent director under the Nasdaq corporate governance rules, satisfies the additional independence criteria for audit committee members under Rule 10A-3 of the Exchange Act, and satisfies the requirements for financial literacy under the Nasdaq corporate governance rules, as applicable. The Board has also determined that each of Mr. Blaszyk and Ms. Selecky qualifies as an audit committee financial expert within the meaning of the applicable rules and regulations of the SEC and satisfies the financial sophistication requirements of the Nasdaq corporate governance rules. The committee held six meetings during the fiscal year ended December 31, 2025.
ImmunityBio, Inc. | 18 | 2026 Proxy Statement
The Compensation Committee was established to oversee the company’s corporate compensation programs. The committee also:
•Reviews and approves corporate goals and objectives relevant to the company’s CEO compensation, evaluates the CEO’s performance in light of those goals and objectives, and, either as a committee or together with the other independent directors (as directed by the Board), determines and approves the CEO’s compensation level based on this evaluation;
•Reviews and approves, or makes recommendations to the Board with respect to, non-CEO executive officer compensation, and incentive compensation and equity-based plans that are subject to Board approval;
•Provides oversight of the company’s compensation policies and plans and benefits programs, and overall compensation philosophy, as well as human capital management programs;
•Administers the company’s equity compensation plans for its executive officers and employees and the granting of equity awards pursuant to such plans or outside of such plans;
•Prepares the report of the Compensation Committee required by the rules and regulations of the SEC; and
•Engages a compensation consultant, legal counsel or other advisors to advise on executive compensation and assess the independence of each in accordance with Nasdaq corporate governance and SEC rules, among other duties and responsibilities.
The current members of the committee are Dr. Maxwell (Chair), Mr. Blaszyk, and Ms. Selecky. The Board has determined that each member of the committee is an independent director under Nasdaq corporate governance rules, satisfies the additional independence criteria for compensation committee members under Rule 10C-1 of the Exchange Act and Nasdaq corporate governance rules, and is a “non-employee director” within the meaning of Rule 16b-3 under the Exchange Act. The committee held five meetings during the fiscal year ended December 31, 2025.
| | | | | | | | | | | | | | |
| Nominating and Corporate Governance Committee |
Although not required by Nasdaq corporate governance rules because ImmunityBio, Inc. is a “controlled company”, the Governance Committee was established to:
•Assist the Board in identifying individuals who are qualified to become members of the Board in accordance with the criteria approved by the Board, and select, or recommend to the Board that the Board select, specific individuals as the director nominees for each meeting of stockholders at which directors are to be elected;
•Recommend members for each Board committee;
•Develop and maintain our corporate governance policies, including our Corporate Governance Guidelines; and
•Oversee the evaluation of the Board.
The Governance Committee also:
•Evaluates the current composition, organization and governance of the Board and its committees;
•Evaluates the “independence” of directors and director nominees;
•Reviews the structure and composition of each committee of the Board and make recommendations, if any, to the Board for changes to the committees;
•Oversees the company’s corporate governance practices, including reviewing and recommending to the Board for approval any changes to the company’s corporate governance framework, including its Certificate of Incorporation and Bylaws; and
ImmunityBio, Inc. | 19 | 2026 Proxy Statement
•Considers questions of possible conflicts of interest of Board members and of corporate officers.
The current members of the committee are Dr. Soon-Shiong (Chair), Gen. Clark and Ms. Cohen. Former director Mr. Brennan served as a member of the Governance Committee through February 7, 2025, the date he resigned from the Board and his committee positions. The Board has determined that each of Gen. Clark and Ms. Cohen, and Mr. Brennan during the time in which he served on the committee, is an independent director under Nasdaq corporate governance rules. The committee held three meetings during the fiscal year ended December 31, 2025.
| | | | | | | | | | | | | | |
| Related Party Transaction Committee |
The Related Party Transaction Committee was established to review and approve related-party transactions. The current members of the committee are Gen. Clark (Chair), Dr. Maxwell, Ms. Selecky, and Mr. Wendel. Former director Mr. Brennan served as Chair of the Related Party Transaction Committee through February 7, 2025, the date he resigned from the Board and his committee positions. Gen. Clark served as a member of the committee during the fiscal year ended December 31, 2025 and was appointed Chair of the committee in April 2025 following the resignation of Mr. Brennan. Ms. Selecky was appointed as a member of the committee in April 2025 following the resignation of Mr. Brennan. Mr. Wendel was appointed as a member of the committee in April 2026. The Board has determined that each member of the committee is an independent director under Nasdaq corporate governance rules. The committee held three meetings during the fiscal year ended December 31, 2025.
The Special Committee was established in October 2020 to, among other things, evaluate and negotiate the terms and conditions of the Merger and to make recommendations to the Board regarding the Merger. The current members of the committee are Mr. Blaszyk (Chair) and Ms. Cohen, each of whom is an independent director. The committee held no meetings during the fiscal year ended December 31, 2025. The Special Committee was dissolved in the first half of 2026.
| | | | | | | | | | | | | | |
| Process for Selecting Directors and Director Qualifications |
As described above, under the terms of the Cambridge Nominating Agreement, Cambridge has the right to designate one individual to be nominated and recommended for election by the Board. Additionally, under the terms of Dr. Simon’s employment agreement with the company, the Board is obligated to nominate and recommend Dr. Simon for election to the Board as long as he is employed by the company.
The Governance Committee uses the following procedures to identify and evaluate any individual recommended or offered for nomination to the Board:
•The Governance Committee will consider candidates recommended by stockholders in the same manner as candidates recommended to the Governance Committee from other sources.
•In its evaluation of director candidates, including the members of the Board eligible for re-election, the Governance Committee will consider the following:
•The current size and composition of the Board and the needs of the Board and its respective committees;
•Factors such as character, integrity, judgment, diversity, age and experience, independence, area of expertise, corporate experience, length of service, potential conflicts of interest, other commitments, and other similar factors, which are evaluated, among others, and not assigned any particular weighting or priority by the Governance Committee; and
•Other factors that the Governance Committee may consider appropriate.
ImmunityBio, Inc. | 20 | 2026 Proxy Statement
The Governance Committee requires that any nominee for a position on the Board must satisfy the following minimum qualifications:
•The highest personal and professional ethics and integrity;
•Proven achievement and competence in the nominee’s field and the ability to exercise sound business judgment;
•Skills that are complementary to those of the existing Board;
•The ability to assist and support management and make significant contributions to the company’s success; and
•An understanding of the fiduciary responsibilities required of a member of the Board and the commitment of time and energy necessary to diligently carry out those responsibilities.
If the Governance Committee determines that an additional or replacement director is required, the Governance Committee may take such measures as it considers appropriate in connection with its evaluation of a director candidate, including candidate interviews, inquiry of the person or persons making the recommendation or nomination, engagement of an outside search firm to gather additional information, or reliance on the knowledge of the members of the Governance Committee, the Board, or management.
The Governance Committee has discretion to decide which individuals to recommend for nomination as directors, and the Board has the final authority in determining the selection of director candidates for nomination to the Board. After completing its review and evaluation of director candidates, the Governance Committee and the Board unanimously recommend all nine of the director nominees for election named in this proxy statement.
To be considered independent for purposes of Rule 10A-3 and Nasdaq rules, a member of an audit committee of a listed company may not, other than in his or her capacity as a member of the audit committee, board of directors, or any other board committee: (1) accept, directly or indirectly, any consulting, advisory, or other compensatory fee from the listed company or any of its subsidiaries; or (2) be an affiliated person of the listed company or any of its subsidiaries.
The Board undertook a review of the independence of our directors and considered whether any director has a material relationship with the company that could compromise his or her ability to exercise independent judgment in carrying out his or her responsibilities. Based upon information requested from and provided by each director concerning his or her background, employment and affiliations, including family relationships, the Board has determined that each of Mr. Blaszyk, Gen. Clark, Mses. Cohen and Selecky, Dr. Maxwell, and Mr. Wendel, representing six of our nine directors, is independent as that term is defined by the SEC and Nasdaq rules. In addition, former director Mr. Brennan had served as a director until his resignation on February 7, 2025. During such time, the Board determined that Mr. Brennan was independent.
In making these determinations, the Board considered the relationships that each non-employee director has with us and all other facts and circumstances the Board deemed relevant in determining their independence, including consulting relationships, family relationships and the beneficial ownership of the company’s capital stock by each non-employee director.
In addition, our Corporate Governance Guidelines require that each independent member of the Board: (i) shall not have and have not been at any time during the past five years, an employee of the company; (ii) shall have received only de minimis remuneration from service as (or compensation paid to an entity the director is an officer of or controls) an advisor, consultant, or legal counsel to the company and/or its senior management for the preceding three years; (iii) shall have no personal service contract(s) with the company or a member of its senior management; and (iv) shall have no business relationships with the company during the preceding three years that required the company to make a disclosure under Regulation S-K of the Exchange Act (other than directorship/de minimis remuneration).
ImmunityBio, Inc. | 21 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
| Prohibited Trading Activities |
Under our Insider Trading Policy, all Board members, employees, consultants and certain other service providers are prohibited from participating directly or indirectly in transactions involving trading activities in the company’s securities that, by their nature, are aggressive or speculative, or may give rise to an appearance of impropriety. Such service providers may not:
•Engage in short sales (sales of stock that the seller does not own or a sale that is completed by delivery of borrowed stock) with respect to the company’s securities;
•Engage in transactions that hedge or offset, or are designed to hedge or offset, any decrease in the market value of the company’s stock;
•Purchase or pledge the company’s securities on margin or as collateral to secure a loan or other obligation, except with prior Board approval; or
•Enter into any derivative or similar transactions with respect to the company’s securities.
| | | | | | | | | | | | | | |
| Compensation Committee Interlocks and Insider Participation |
None of our executive officers currently serves, or in the past year has served, as a member of the Compensation Committee, or other Board committees performing equivalent functions (or in the absence of any such committee, the entire Board), or as a director of any entity that has one or more executive officers serving on the Compensation Committee or the Board. None of the members of the Compensation Committee during the fiscal year ended December 31, 2025, which included Dr. Maxwell, Mr. Blaszyk, and Ms. Selecky, has been an officer or employee of the company.
| | |
| Compensation Discussion and Analysis |
| | | | | | | | | | | | | | |
| Our Named Executive Officers |
The names of our executive officers, their ages, and their positions with the company as of April 29, 2026 are set forth below. There are no family relationships among any of our directors or executive officers.
| | | | | | | | | | | | | | |
| Name | | Age | | Position |
| | | | |
Richard Adcock | | 57 | | CEO and President |
| David Sachs | | 48 | | CFO |
| Patrick Soon-Shiong, M.D., FRCS (C), FACS | | 73 | | Founder, Global Chief Scientific and Medical Officer |
| Regan Lauer | | 56 | | Chief Accounting Officer |
David Sachs has served as our CFO since March 2021. He previously served as the chief financial officer of NantCell, Inc. (formerly known as ImmunityBio, Inc., a private company) from July 2019 to March 2021. He also served as chief financial officer of Integrity Healthcare, LLC (a NantWorks subsidiary) from February 2018 to August 2020. From April 2011 to June 2019, he held various executive and board positions at NantWorks and its subsidiaries and acted as a member of the investment committee. He also served as chief financial officer of NantHealth, Inc. from 2013 to 2015. Prior to NantWorks, he served in business development roles at Celgene Corporation and Abraxis BioScience and as an investment banker with Bank of America Merrill Lynch. Mr. Sachs received his B.A. in Economics from the University of California at Los Angeles and his M.B.A. in Finance and Strategy from the UCLA Anderson School of Management.
ImmunityBio, Inc. | 22 | 2026 Proxy Statement
Regan Lauer has served as the company’s Chief Accounting Officer since November 2021. Prior to ImmunityBio, she was Corporate Vice President of Accounting at Progenity, Inc., a biotechnology company, from October 2019 to November 2021. Previously, Ms. Lauer worked for Silvergate Capital Corporation where she was Chief Financial Officer from 2016 to 2019 and Senior Vice President and Corporate Controller from 2013 to 2016. Prior to that, Ms. Lauer worked for First PacTrust Bancorp, Inc. from 2000 to 2012 holding many executive positions, including Principal Accounting Officer. Ms. Lauer began her career with Deloitte & Touche LLP and holds a Bachelor’s degree in Accounting from the Saint Louis University.
This Compensation Discussion and Analysis (CD&A) is intended to help our stockholders understand our executive compensation program by providing an overview of our executive compensation-related policies, practices, and decisions for 2025. It also explains how we determined the material elements of compensation for our principal executive officer (PEO), our principal financial officer (PFO), and our two other named executive officers as of December 31, 2025 (referred to as our NEOs). We had a total of four NEOs in 2025, all of whom served for the full year. For fiscal year 2025, our NEOs were:
•Richard Adcock — President and CEO
•David Sachs — CFO
•Patrick Soon-Shiong, M.D., FRCS (C), FACS — Global Chief Scientific and Medical Officer
•Regan Lauer — Chief Accounting Officer
Specifically, this CD&A provides an overview of our executive compensation philosophy, the overall objectives of our executive compensation program, and each compensation element that we provide to our executive officers. In addition, it explains how and why the Compensation Committee arrived at the specific compensation decisions for our NEOs in 2025.
Overview
ImmunityBio, Inc. is a biotechnology company focused on innovating, developing, and commercializing next-generation immunotherapies designed to activate the patient’s immune system and deliver durable protection against cancer and infectious diseases. Our approach harnesses both the adaptive and innate immune systems with the goal of restoring immune function and generating lasting immunological memory in patients. At the core of our strategy is the Cancer BioShield™ platform, which is designed to stimulate critical lymphocytes, including natural killer (NK) cells, cytotoxic T cells, and memory T cells via our proprietary IL-15 superagonist, ANKTIVA® (nogapendekin alfa inbakicept). Our Cancer BioShield platform is anchored by this antibody-cytokine fusion protein and is complemented by a portfolio that includes adenovirus-vectored vaccines, allogeneic (off-the-shelf) and autologous NK-cell therapies, and additional immunomodulators intended to promote immunogenic cell death and support durable immune responses while potentially reducing reliance on high-dose chemo-radiation therapy.
2025 Business Highlights
We believe that 2025 was a pivotal year in the evolution of the company, laying the foundation for continued growth and success. Highlights include:
•Receiving approval from the UK Medicines and Healthcare products Regulatory Agency for the United Kingdom and a recommendation for approval from the European Medicines Agency for the European Union for ANKTIVA with Bacillus Calmette-Guérin (BCG) for the treatment of adult patients with BCG-unresponsive non-muscle invasive bladder cancer (NMIBC) carcinoma in situ (CIS)
•Raising over $400 million of capital through multiple registered direct offerings and “at-the-market” offerings
•Accelerated enrollment in BCG-Naïve NMIBC with CIS trial (QUILT-2.005)
ImmunityBio, Inc. | 23 | 2026 Proxy Statement
•Obtaining U.S. Food and Drug Administration (FDA) Expanded Access Program (EAP) authorization for use of ANKTIVA to treat lymphopenia in adult patients with refractory or relapsed solid tumors, regardless of tumor type, who have progressed following first-line standard-of-care treatment, including chemotherapy, radiation, or immunotherapy
•Obtaining FDA Regenerative Medicine Advanced Therapy (RMAT) designation for use of ANKTIVA and PD-L1 CAR-NK in combination with standard-of-care chemotherapy/radiotherapy for the reversal of lymphopenia and treatment of multiply relapsed locally advanced or metastatic pancreatic cancer
2025 Executive Compensation Summary
Consistent with our performance and compensation philosophy as further detailed below, the Compensation Committee took the following key actions with respect to the NEOs during fiscal 2025:
•Annual Base Salary. Consistent with our approach to provide compensation competitive with our peer companies approximating the market median, in February 2025, the Compensation Committee approved an annual target base salary increase of 5% for Mr. Adcock and Dr. Soon-Shiong, and 3% for Mr. Sachs and Ms. Lauer, in each case effective as of March 10, 2025.
•Short-Term Incentive Compensation. No changes in target bonus opportunities were made for our NEOs in 2025, which remained at 75% of annual target base salary for Mr. Adcock and Dr. Soon-Shiong and 50% for Mr. Sachs and Ms. Lauer, as the existing target bonus opportunities were considered market competitive.
In March 2026, the Compensation Committee determined that the company achieved approximately 77% of its corporate goals, which were comprised of specific financial and operating objectives, which had been established by the Compensation Committee in March 2025 (the 2025 STIP). See the section below entitled “—Corporate Performance in Fiscal 2025 and Payouts” for information regarding our 2025 performance goals, weightings, and payout methodology. In addition, in order to reward our NEOs for their achievements for 2025 not otherwise reflected in corporate performance measures of the 2025 STIP, the Compensation Committee determined to award additional discretionary bonuses to our NEOs such that the aggregate value of the annual bonus payout each NEO would receive was equal to 100% of each NEO’s target annual bonus opportunity. •Long-Term Incentive Compensation. In order to strike a balance between company performance, individual performance, and retention while fostering an ownership mindset and creating wealth creation opportunities for NEOs, in February 2025, the Compensation Committee approved grants under our long-term incentive plan (2025 LTIP) to our NEOs in the form of stock options and restricted stock units (RSUs). The awards vest annually in approximately equal increments over a period of three years, provided the executive continues to provide services to the company through each vesting date. These awards are intended to provide long-term incentive compensation that is comparable with similar roles in our peer companies.
Our executive compensation program is designed to be reasonable and competitive, and balance our goal of attracting, motivating, rewarding and retaining top-performing senior executives in a highly competitive biotechnology industry with our goal of aligning their interests with those of our stockholders, and emphasizing pay-for-performance.
Executive Compensation Policies and Practices
We strive to maintain sound executive compensation policies and practices, including compensation-related corporate governance standards, consistent with our executive compensation philosophy. During fiscal 2025, the following executive compensation policies and practices were in place. We believe these policies and practices drive performance and either prohibit or minimize behaviors that we do not believe serve our stockholders’ long-term interests.
ImmunityBio, Inc. | 24 | 2026 Proxy Statement
| | | | | |
| Our Compensation Best Practices |
| What We Do | What We Do Not Do |
Maintain an Independent Compensation Committee. The Compensation Committee consists solely of independent directors who establish our compensation policies and practices. | No “Single Trigger” Change in Control Severance Payments or Benefits. We do not provide “single trigger” change in control severance payments or benefits to our NEOs. |
Retain an Independent Compensation Advisor. The Compensation Committee engages and retains its own advisors. During 2025, the Compensation Committee engaged Mercer to provide information, analysis, and other advice to assist with its responsibilities. | No Excise Tax Payments on Post-Employment Compensation Arrangements. We do not provide any excise tax reimbursement payments (including “gross-ups”) on payments or benefits contingent upon a change in control of the company. |
Annual Executive Compensation Review. The Compensation Committee conducts an annual review and approval of our compensation strategy, including a review and determination of our compensation peer group used for comparative purposes and a review of our compensation-related risk profile to ensure that our compensation programs do not encourage excessive or inappropriate risk-taking and that the level of risk that they do encourage is not reasonably likely to have a material adverse effect on us. | Limited Perquisites. We provide minimal perquisites and other personal benefits to our NEOs. |
Emphasize Long-Term Equity Compensation. The Compensation Committee uses equity awards to deliver long-term incentive compensation opportunities to our NEOs. These equity awards vest or may be earned over multi-year periods, which better serves our long-term value creation goals and retention objectives. | No Tax Reimbursement on Perquisites. We do not provide any tax reimbursement payments (including “gross-ups”) on any perquisites or other personal benefits, except in limited circumstances to achieve specific business objectives. |
Reasonable Severance Arrangements. The post-employment compensation arrangements for our NEOs provide for amounts and multiples that are within reasonable market norms. | No Special Retirement, Health or Welfare Benefits. We do not provide our NEOs with any retirement, health or welfare benefits programs, other than participation in our broad-based employee plans and programs on the same basis as our other full-time, salaried employees, except in limited circumstances to achieve specific business objectives. |
Compensation Philosophy
In order to create value for our stockholders, it is critical to attract, motivate and retain key executive officer talent by providing competitive compensation packages. The market for talented individuals in the life sciences industry is highly competitive and becoming more challenging for employers. The Compensation Committee believes our compensation program should align executive interests with the drivers of growth and stockholder returns, and support achievement of the company’s key business mission, goals, and objectives. Consequently, the Compensation Committee believes the substantial majority of NEO compensation should be at-risk and performance-based to facilitate the successful execution of our business strategy. In evaluating the company’s executive compensation level and programs for NEOs in comparison to the company’s peer group, the Compensation Committee generally targets compensation levels consistent with the market median, but considers total direct compensation above or below the median depending on level of experience and criticality of executive talent. Although the Compensation Committee considers a multitude of factors in its deliberations, including competitive positioning relative to its peer group, it places no formal weighting on any one factor.
ImmunityBio, Inc. | 25 | 2026 Proxy Statement
Decision-Making Process
Role of the Compensation Committee
The Compensation Committee, which is comprised entirely of independent directors, is responsible for discharging the Board’s responsibilities relating to compensation of our CEO and other executive officers, overseeing our overall compensation structure, policies and programs, and reviewing our processes and procedures for the consideration and determination of director and executive compensation. The primary objective of the Compensation Committee is to develop and implement compensation policies and plans to attract and retain key management personnel, motivate management to achieve our corporate goals and strategies, and align the interests of management with the long-term interests of our stockholders. We have not adopted formal guidelines for allocating total compensation between long-term and short-term compensation, cash compensation and non-cash compensation, or among different forms of non-cash compensation. As we continue to grow, the Compensation Committee will evaluate our compensation philosophy and program objectives as circumstances require.
The Compensation Committee annually reviews the compensation, plans, policies and programs for our CEO and our other executive officers. In connection therewith, the Compensation Committee considers, among other things, each executive officer’s performance in light of established individual and corporate goals and objectives and the recommendations of our CEO, in consultation with management as well as an independent compensation consultant, as described below.
Role of Management
In discharging its responsibilities, the Compensation Committee works with management, including our CEO. Our management assists the Compensation Committee by providing information on corporate and individual performance, market compensation data and management’s perspective on compensation matters. In particular, the Compensation Committee considers the recommendations of our CEO when reviewing base salary and incentive performance compensation levels of our executive officers and when setting specific individual and corporate performance targets under our annual incentive bonus plan for our executive officers. Our CEO and Global Chief Scientific and Medical Officer have no input and are not present during voting or deliberations about their respective compensation.
Independent Compensation Consultant
Pursuant to its charter, the Compensation Committee has the authority to retain compensation consultants to assist in its evaluation of executive and director compensation. Consistent with the prior year, the Compensation Committee engaged Mercer as an independent outside compensation consultant to perform compensation advisory services during 2025 and to provide information, recommendations and other advice relating to director and executive compensation. The Compensation Committee believes working with an independent compensation consultant furthers the company’s objectives to recruit and retain qualified executives, align executive interests with those of stockholders and ensure that executive compensation packages will appropriately motivate and reward ongoing achievement of business goals. The Compensation Committee intends to continue to retain a consultant to provide similar information and analyses in future years for consideration in determining annual cash, equity and other compensation for our employees, executive officers and directors.
The Compensation Committee has reviewed the independence of Mercer’s advisory role relative to the consultant independence factors adopted by the SEC to guide listed companies in determining the independence of their compensation consultants, legal counsel and other advisors. Following its review, the Compensation Committee determined that no conflict of interest arose from the work performed by Mercer during the fiscal year ended December 31, 2025.
Peer Group
For purposes of comparing our executive compensation against the competitive market, the Compensation Committee reviews and considers the compensation levels and practices of a group of peer companies. The Compensation Committee reviews our compensation peer group at least annually and makes adjustments to our peer group if necessary, taking into account changes in both our business and our peer companies’ businesses. The Compensation Committee also uses market data from our compensation peer group and from additional Mercer databases as one factor in evaluating whether the compensation for our executive officers is competitive in the market. The Compensation Committee and the Board, as applicable, also rely on their own knowledge and judgment in evaluating market data and making compensation decisions.
ImmunityBio, Inc. | 26 | 2026 Proxy Statement
In July 2024, the Compensation Committee, in consultation with Mercer, reassessed and updated its peer group of companies to assist with its 2025 compensation assessment. Natera, Inc., Exact Sciences Corporation, and Mirati Therapeutics, Inc. were removed from the peer group, while Blueprint Medicines Corporation, TG Therapeutics, Inc., and Immunocore Holdings plc were added to better align with ImmunityBio’s industry, financial size, and business model. The peer group for 2025 consists of public biotechnology companies that are relatively similar to us in terms of revenue, market capitalization and industry focus.
The peer group of companies for 2025 for executive and director compensation decisions included the following companies:
| | | | | |
| Neurocrine Biosciences, Inc. | Novavax, Inc. |
| BridgeBio Pharma, Inc. | Arcus Biosciences, Inc. |
| Blueprint Medicines Corporation | Syndax Pharmaceuticals, Inc. |
| TG Therapeutics, Inc. | Kura Oncology, Inc. |
| Ultragenyx Pharmaceutical Inc. | Allogene Therapeutics, Inc. |
| Immunocore Holdings plc | Fate Therapeutics, Inc. |
This compensation peer group was used by the Compensation Committee in connection with its annual review of our executive compensation program in 2025. Specifically, the Compensation Committee reviewed the compensation data drawn from the compensation peer group, in combination with industry-specific compensation survey data, to develop a subjective representation of the “competitive market” with respect to current executive compensation levels and related policies and practices. The Compensation Committee then evaluated how our pay practices and the compensation levels of our executive officers compared to the competitive market. As part of this evaluation, the Compensation Committee also reviewed the performance measures and performance goals generally used within the competitive market to reward performance.
We do not believe that it is appropriate to make compensation decisions, whether regarding base salaries or short-term or long-term incentive compensation, solely based upon competitive market data with respect to a peer or other representative group of companies. However, the Compensation Committee believes that information regarding the compensation practices of other companies is useful in at least two respects: (1) the Compensation Committee recognizes that our compensation policies and practices must be competitive in the marketplace and (2) this information is useful in assessing the reasonableness and appropriateness of individual executive compensation elements and of our overall executive compensation packages. In evaluating the company’s executive compensation level and programs for NEOs in comparison to the company’s peer group, the Compensation Committee generally targets compensation levels consistent with the market median, but considers total direct compensation above or below the median depending on level of experience and criticality of executive talent. This information is only one of several factors that the Compensation Committee considers, however, in making its decisions with respect to the compensation of our executive officers, including our NEOs.
2024 Say-on-Pay Results
At our 2024 annual meeting of stockholders, we held a non-binding stockholder advisory vote on executive compensation (commonly referred to as a “say-on-pay” vote). The say-on-pay vote received support from approximately 99% of the votes cast by our stockholders on the matter (excluding broker non-votes). Although a majority of stockholders expressed support for the compensation of our NEOs, the Compensation Committee values any additional stockholder feedback and endeavors to respond to stockholders’ concerns. Additionally, the Compensation Committee obtains feedback, advice, and recommendations on compensation best practices from its independent external compensation consultant, Mercer, and assesses the reports and publications of proxy advisory firms. The Compensation Committee also reviews the company’s performance, the compensation practices of our peers, and compensation surveys and other materials regarding general and executive compensation.
We hold a say-on-pay vote every three years, the next of which will be in connection with our 2027 annual meeting of stockholders. We will also hold a vote on the frequency of say-on-pay at our 2027 annual meeting.
We value the opinions of our stockholders. The Compensation Committee will consider the outcome of the say-on-pay vote, as well as feedback received throughout the year, when making compensation decisions for our NEOs.
ImmunityBio, Inc. | 27 | 2026 Proxy Statement
Elements of Compensation for 2025
The company’s executive compensation program consists of three primary elements: base salaries, annual cash incentive opportunities, and long-term incentive compensation in the form of stock options and RSU awards. Our executives, including our NEOs, also participate in the standard employee benefit plans available to most of the company’s employees. In addition, as described below, our executives, including our NEOs, are entitled to certain severance payments pursuant to their employment agreements or offer letters. Each of these compensation elements is discussed in detail below, including a description of the particular element and how it fits into our overall executive compensation and a discussion of the amounts of compensation earned by our NEOs in 2025 under each of these elements.
Variable compensation represents approximately 93% of our CEO’s target total direct compensation and, on average, approximately 78% of the target total direct compensation for our other NEOs, as shown in the following charts:
Base Salary
We pay base salaries to our executive officers, including our NEOs, as the fixed portion of their compensation to provide them with a reasonable degree of personal income, and to attract and retain our executives. Annual salary decisions are typically made in recognition of competitive data as well as the skills and experience that each individual brings to the company. During 2025, there were several key dates at the company and individual level that served as a catalyst for Compensation Committee determinations of base salary.
The Compensation Committee, in consultation with Mercer, engaged in further review of compensation for NEOs and other executives. In March 2025 as a result of its review of peer group data, the Compensation Committee determined to increase the base salary of Mr. Adcock and Dr. Soon-Shiong by 5% and the base salary of Mr. Sachs and Ms. Lauer by 3%. The adjustment levels for Mr. Adcock and Dr. Soon-Shiong considered, among other factors, that their salaries had not been adjusted since 2022. In reaching its determination, the Compensation Committee also considered the specialized skill, knowledge and expertise that Dr. Soon-Shiong provided to the company in his operational role as Global Chief Scientific and Medical Officer of the company.
ImmunityBio, Inc. | 28 | 2026 Proxy Statement
The table below illustrates the changes in base salary amounts for the NEOs over the course of 2025 as described above:
| | | | | | | | | | | | | | | | | | | | |
| Name | | 2024 Annual
Base Salary
($) | | 2025 Annual
Base Salary (1)
($) | | Year-Over-Year
Increase in Base
Salary
(2025 vs. 2024) |
| | | | | | |
| Richard Adcock | | 796,000 | | | 835,800 | | | 5 | % |
| David Sachs | | 575,358 | | | 592,619 | | | 3 | % |
| Patrick Soon-Shiong | | 621,000 | | | 652,050 | | | 5 | % |
| Regan Lauer | | 329,410 | | | 339,292 | | | 3 | % |
_______________
(1) Effective March 10, 2025.
Short-Term Incentive Compensation Program
We provide short-term cash incentive compensation opportunities under our short-term incentive compensation plan (STIP) to our executive officers, including our NEOs, in the form of annual cash bonuses to drive our short-term success and reward NEOs for their individual performance and the performance of the company over the fiscal year. Bonuses are tied to achievement of annual corporate performance goals, as determined by the Compensation Committee in its discretion. Notwithstanding the foregoing, the Compensation Committee may adjust bonuses payable under the STIP based on achievement of individual performance goals or pay bonuses (including, without limitation, discretionary bonuses) to executives, including our NEOs, based upon such other terms and conditions as the Compensation Committee may in its discretion determine. Each executive participating in the STIP, including our NEOs, has a target bonus opportunity set for each performance period.
In March 2025, the Compensation Committee established a target cash bonus amount for each NEO expressed as a percentage of annual base salary, and a set of pre-established objective company performance criteria with various relative weighting under the STIP. For fiscal year 2025, the target amounts were 75% for Mr. Adcock and Dr. Soon-Shiong and 50% for Mr. Sachs and Ms. Lauer, which were the same as for fiscal year 2024.
| | | | | | | | | | | | | | | | | | | | |
| Name | | 2025 Annual
Base Salary
($) | | 2025 Target
Bonus Opportunity
(as a % of Annual
Base Salary) | | 2025 Target
Bonus Opportunity
($) |
| | | | | | |
| Richard Adcock | | 835,800 | | | 75 | % | | 626,850 | |
| David Sachs | | 592,619 | | | 50 | % | | 296,310 | |
| Patrick Soon-Shiong | | 652,050 | | | 75 | % | | 489,038 | |
| Regan Lauer | | 339,292 | | | 50 | % | | 169,646 | |
In aggregate, the target bonus opportunities for the NEOs were positioned within a range that the Compensation Committee viewed as competitive.
Corporate Performance in Fiscal 2025 and Payouts
The Compensation Committee worked with senior management, the Board and Mercer to develop the performance goals for 2025 based on the company’s strategic and operational goals and objectives and a review of peer group practices for short-term incentive programs at companies at a similar stage as the company. Following these discussions, the Compensation Committee determined that 50% of the 2025 STIP metrics would be linked to Commercial and Financial results and 50% to Clinical results. The 50% Commercial and Financial component includes Revenue, weighted at 40% of the total target bonus opportunity, and Adjusted EBITDA, weighted at 10% of the total target bonus opportunity. The Clinical component was broken down further into four separate regulatory and submission objectives, with two weighted at 15% and two weighted at 10%. The annual fiscal 2025 goals for each metric were established in March 2025. The Compensation Committee believes that these metrics are highly linked to creating both short- and long-term value for our stockholders. The Compensation Committee applied business judgment in setting these objectives, related weighting and goals.
ImmunityBio, Inc. | 29 | 2026 Proxy Statement
A summary of key elements and outcomes of the 2025 STIP are as follows:
•Payouts under the 2025 STIP could range from 0% to 140% of each NEO’s target bonus opportunity, based on achievement against the established performance goals.
◦Commercial results were weighted 40% and consisted of Revenue objectives (total revenue as presented in our Annual Report on Form 10-K for the year ended December 31, 2025). Payout under the Commercial component could range from 0% to 150%. Revenue achievement at 25% below target performance or at maximum performance would have resulted in payouts of 75% and 150%, respectively. Final achievement for the Revenue objective was 66%.
◦Financial results were weighted 10% and consisted of Adjusted Earnings Before Interest, Taxes, Depreciation, and Amortization objectives (Adjusted EBITDA – as defined below). Payout under the Financial component could range from 0% to 150%. Adjusted EBITDA achievement at 25% below target performance or at maximum performance would have resulted in payouts of approximately 50% and 150%, respectively. Final achievement for the Adjusted EBITDA objective was 86%.
◦Clinical development results were weighted 50% and included four objectives related to domestic and international regulatory approvals and submissions. Payout under the Clinical component could range from 0% to 130%. Final achievement for the clinical development objectives was 85%. The clinical development operational performance goals for the 2025 STIP are not disclosed because we believe it would be competitively harmful to do so, as it would provide competitors insight into our strategic and financing planning processes.
| | | | | | | | | | | | | | | | | | | | |
| Corporate Performance Category | | Objectives | | Weighting
(%) | | Actual
Achievement
(%) |
| | | | | | |
| Commercial and Financial Results | | Revenue | | 40 | % | | 66 | % |
| | Adjusted EBITDA | | 10 | % | | 86 | % |
| | | | | | |
| Clinical Development | | Objectives related to domestic and
international regulatory approvals and
submissions | | 50 | % | | 85 | % |
| | | | | | |
| | Total Bonus Achievement (prior to
Compensation Committee discretion) | | | | 77 | % |
In February 2026, based on the assessment of our performance against these objectives, the Compensation Committee determined to award bonuses to our NEOs pursuant to the 2025 STIP and approved bonus payouts of approximately 77% of target for our NEOs. Awards were determined based on the objectives, related targets, and weightings set in March 2025. In addition, in order to reward our NEOs for their achievements for 2025 not otherwise reflected in corporate performance measures of the 2025 STIP set early in the year, the Compensation Committee determined to award additional discretionary bonuses to our NEOs such that the aggregate value of the annual bonus payout each NEO would receive was equal to 100% of each NEO’s target annual bonus opportunity. These achievements included, among others:
•Successful commercial launch of our first FDA-approved drug, ANKTIVA. 2025 was our first full year of commercial sales after implementation of our permanent J-code (J9028).
•Significant capital raised over the year by management to accelerate key programs
•Accelerated enrollment in critical trials
•Extensive regulatory successes globally and FDA Expanded Access authorizations and RMAT designation for core platforms in extremely large potential future indications
ImmunityBio, Inc. | 30 | 2026 Proxy Statement
The annual bonuses paid to our NEOs under the 2025 STIP and discretionary bonuses are set forth in the Summary Compensation Table below:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| Name | | 2025 Target
Bonus Opportunity
($) | | Annual Payout
Under 2025
STIP
($) | | Discretionary
Bonus
($) | | Total Bonus
for 2025
($) |
| | | | | | | | |
| Richard Adcock | | 626,850 | | | 485,166 | | | 141,684 | | | 626,850 | |
| David Sachs | | 296,310 | | | 229,336 | | | 66,974 | | | 296,310 | |
| Patrick Soon-Shiong | | 489,038 | | | 378,503 | | | 110,535 | | | 489,038 | |
| Regan Lauer | | 169,646 | | | 131,302 | | | 38,344 | | | 169,646 | |
Long-Term Incentive Compensation Program
We believe that long-term incentive compensation in the form of equity awards is an important component of executive compensation to ensure that a significant portion of a NEO’s compensation opportunity is related to factors that directly or indirectly influence stockholder value. Further, equity participation establishes a sense of ownership and aligns executive interests with those of other stockholders. In 2015, the company adopted the ImmunityBio, Inc. 2015 Equity Incentive Plan (the 2015 Plan), and in 2025 the company adopted the ImmunityBio, Inc. 2025 Equity Incentive Plan (the 2025 Plan). Historically, the company has granted RSU awards and option awards to employees and executives at various times. Option awards, when granted with exercise prices equal to the fair market value of our common stock on the date of grant, are inherently performance oriented and provide an appropriate long-term incentive for our executive officers, since the option awards reward them only to the extent that our stock price increases and stockholders realize value following their grant date. RSU awards provide tangible value to executive officers and serve as an incentive and retention tool during a difficult operating or volatile business environment, while still being tied to stockholder value. Additionally, RSU awards enable our executive officers to accumulate stock ownership in the company to further increase their alignment with stockholders.
The company proposes awards for participants in the LTIP, including executive management, based on peer data and analysis in consultation with Mercer. Proposed grant values for participants are discussed and modified or approved by the Compensation Committee.
The approved target grant value is divided into a dollar value for RSU awards and a dollar value for option awards based on the vehicle mix as approved by the Compensation Committee (which was allocated 25% to RSU awards and 75% to option awards, respectively, for NEOs in 2025). The aggregate number of RSU awards is determined by dividing (i) the value of the RSU award by (ii) the average closing sales price (the closing price) of the company’s common stock in trading on the Nasdaq on each of the twenty (20) trading days encompassing from the date the Compensation Committee approves the award. The aggregate number of shares of the company’s common stock subject to option awards is determined by dividing (i) the value of the option award in U.S. dollars by (ii) the grant date fair value, as determined under accounting principles generally accepted in the U.S. (U.S. GAAP), of an option award covering one share of the company’s common stock, with such determination utilizing the Black-Scholes option pricing model, provided that such model will incorporate the average closing price of the company’s common stock in trading on the Nasdaq on each of the twenty (20) trading days encompassing the date the Compensation Committee approves the award.
In February 2025, in recognition of our financial and operational results and each NEO’s individual performance, the Compensation Committee approved the grant of RSU awards and option awards to our NEOs under the 2025 LTIP, effective February 10, 2025. In determining the size of each NEO’s equity awards, the Compensation Committee took into consideration the recommendations of our CEO, except with respect to his own equity awards, as well as the factors described above. The Compensation Committee also considered the existing equity holdings of each NEO, including the current economic value of their unvested equity awards and the ability of these unvested holdings to satisfy our retention objectives. The Compensation Committee also considered the dilutive effect of our long-term incentive compensation practices, and the overall impact that these equity awards, as well as awards to other employees, will have on stockholder value.
ImmunityBio, Inc. | 31 | 2026 Proxy Statement
The 2025 LTIP awards granted to our NEOs on February 10, 2025 are shown in the table below:
| | | | | | | | | | | | | | | | | | | | |
| | Stock Awards | | Option Awards |
| Name | | Number
of Shares | | Number
of Shares | | Exercise
Price (1)
($) |
| | | | | | |
| Richard Adcock | | 822,368 | | | 2,812,500 | | | 3.39 | |
| David Sachs | | 246,710 | | | 843,750 | | | 3.39 | |
| Patrick Soon-Shiong | | 822,368 | | | 2,812,500 | | | 3.39 | |
| Regan Lauer | | 16,447 | | | 56,250 | | | 3.39 | |
_______________
(1) Equal to the closing price of the company’s common stock as reported on the Nasdaq on the date of grant.
Each award granted to our NEOs in connection with the 2025 LTIP vests ratably over three years, subject in each instance to the award recipient remaining a “service provider” (as that term is defined in the 2015 Plan) through each applicable vesting date.
Perquisites and Other Benefits
The company provides certain of our management team and more senior executives, including our NEOs, with certain perquisites and other personal benefits that the company believes are reasonable and consistent with the overall compensation program to better enable the company to attract and retain employees for key positions.
The company has established a 401(k) retirement savings plan for its employees, including the NEOs, who satisfy certain eligibility requirements. Under the 401(k) plan, eligible employees may elect to contribute pre-tax amounts, up to a statutorily prescribed limit, to the 401(k) plan. The company believes that providing a vehicle for tax-preferred retirement savings through the 401(k) plan adds to the overall desirability of its executive compensation package and further incentivizes the company’s employees, including the NEOs, in accordance with the company’s compensation policies. For 2025, the company approved payment of employer matching contributions of 4% to each eligible participant’s plan account in an amount equal to 100% of each participant’s contributions, with eligible compensation limited to $350,000.
The company provides Mr. Adcock with the reimbursement of premiums associated with his personal life insurance policies pursuant to the terms of his Offer of Employment letter dated October 20, 2020, as well as related tax neutrality payments to make such reimbursements tax neutral to Mr. Adcock.
In addition, the company provides other benefits to the NEOs on the same basis as all of our full-time employees. These benefits include health, dental and vision insurance, short- and long-term disability insurance, accidental death and dismemberment insurance, and basic life insurance coverage. We also provide paid time off and other paid holidays to all employees, including our NEOs. We do not offer our employees a non-qualified deferred compensation plan or pension plan. We design our employee benefits programs to be affordable and competitive in relation to the market, as well as compliant with applicable laws and practices. We adjust our employee benefits programs as needed based upon regular monitoring of applicable laws and practices, the competitive market, and our employees’ needs.
ImmunityBio, Inc. | 32 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
| Employment and Change in Control Agreements |
Overview of Employment Agreements
Richard Adcock
On October 20, 2020, we entered into an Offer of Employment letter with Mr. Adcock pursuant to which he agreed to serve as the company’s CEO. Mr. Adcock is eligible to participate in the company’s annual bonus plan and had an initial target bonus of 50% of his then-current annual base salary. Bonuses awarded under the company’s bonus plan are based on established performance criteria; however, the Compensation Committee ultimately exercises discretion over awards made under the plan. In order to receive any bonus payment, Mr. Adcock must remain continuously employed through, and still be employed by the company on, the date any such bonus is paid. As of April 29, 2026, Mr. Adcock’s annual base salary is $860,874 and his target bonus is 75%.
Mr. Adcock is eligible to participate in any regular health insurance, retirement, and other benefit plans established by the company for its employees from time to time. The company will also reimburse Mr. Adcock for costs associated with his two current life insurance policies, inclusive of tax neutrality payments to make such reimbursements tax neutral to Mr. Adcock.
If Mr. Adcock’s employment is terminated without “cause” (as such term is defined in his Offer of Employment letter), he will receive a single cash payment equal to 12 months of his then-current annual base salary, less all applicable federal, state, and local withholding and deductions. Such payment will be made within 2.5 months following the date on which termination occurs, subject to his timely execution and non-revocation of a release of claims in a form reasonably satisfactory to the company.
David Sachs
On August 3, 2020, NantCell, Inc. (formerly known as ImmunityBio, Inc., a private company) entered into an Offer of Employment letter with Mr. Sachs (who previously provided services through a shared services agreement with NantWorks) as its chief financial officer. In connection with the Merger, Mr. Sachs became the company’s CFO effective March 9, 2021. Mr. Sachs is eligible to participate in the company’s annual bonus plan with a target bonus of 50% of his then-current annual base salary. Bonuses awarded under the company’s bonus plan are based on established performance criteria; however, the Compensation Committee ultimately exercises discretion over awards made under the plan. In order to receive any bonus payment, Mr. Sachs must remain continuously employed through, and still be employed by the company on, the date any such bonus is paid. As of April 29, 2026, Mr. Sachs’ annual base salary is $610,397 and his target bonus is 50%.
Mr. Sachs is eligible to participate in any regular health insurance, retirement, and other benefit plans established by the company for its employees from time to time.
Mr. Sachs is also eligible for a severance payment if the company terminates his employment without “cause” or if he resigns for “good reason” (each as defined in his Offer of Employment Letter) (the Severance Payment). The Severance Payment will be equal to: (i) 83.33% of his then-current base salary (i.e., 10 months of severance) plus (ii) a prorated bonus paid out at 100% of his target annual bonus (i.e., the target 50% bonus) for the calendar year in which the termination occurs, less all applicable federal, state, and local withholding and deductions. Such payment will be made within 60 days following the date on which termination occurs, subject to his timely execution and non-revocation of a release of claims in a form reasonably satisfactory to the company.
ImmunityBio, Inc. | 33 | 2026 Proxy Statement
Patrick Soon-Shiong
On August 11, 2021, the Board appointed Dr. Soon-Shiong as Global Chief Scientific and Medical Officer of the company. Dr. Soon-Shiong is eligible to participate in the company’s annual bonus plan and had an initial target bonus of 50% of his then-current annual base salary. Bonuses awarded under the company’s bonus plan are based on established performance criteria; however, the Compensation Committee ultimately exercises discretion over awards made under the plan. In order to receive any bonus payment, Dr. Soon-Shiong must remain continuously employed through, and still be employed by the company on, the date any such bonus is paid. As an employee of the company, he does not receive compensation for his service as a director under the company’s Director Compensation Program. As of April 29, 2026, Dr. Soon-Shiong’s annual base salary is $671,612 and his target bonus is 75%.
Dr. Soon-Shiong is eligible to participate in any regular health insurance, retirement, and other benefit plans established by the company for its employees from time to time.
Regan Lauer
On October 12, 2021 (effective November 1, 2021), ImmunityBio, Inc. entered into an Offer of Employment with Ms. Lauer pursuant to which she agreed to serve as the company’s Senior Vice President, Accounting, with the expectation that the role would transition to Chief Accounting Officer upon approval by the Board. Ms. Lauer is eligible to participate in the company’s annual discretionary bonus plan and had an initial target bonus of 40% of her then-current base salary. Bonuses awarded under the company’s bonus plan are based on established performance criteria; however, the Compensation Committee ultimately exercises discretion over awards made under the plan. In order to receive any bonus payment, Ms. Lauer must remain continuously employed through, and still be employed by the company on, the date any such bonus is paid. As of April 29, 2026, Ms. Lauer’s annual base salary is $349,471, and her target bonus is 50%.
Ms. Lauer is eligible to participate in any regular health insurance, retirement, and other benefit plans established by the company for its employees from time to time.
If the company terminates her employment without “cause” (as such term is defined in her Offer of Employment letter), Ms. Lauer will receive a severance cash payment equal to 18 weeks of her then‑current annual base salary, less required withholdings, provided she executes a satisfactory release within 60 days of termination; the payment will be made on the 60th day following termination, subject to any applicable Section 409A timing requirements of the U.S. Internal Revenue Code of 1986, as amended.
Merger and Change in Control
ImmunityBio, Inc. 2025 Equity Incentive Plan
The 2025 Plan provides that in the event of a merger or change in control, as defined under the 2025 Plan, each outstanding award will be treated as the Administrator determines, which may include continuation by the successor corporation, acceleration of vesting, or termination in exchange for cash or property. If the successor corporation does not continue an award, the participant will fully vest in 100% of the then-unvested shares subject to outstanding options and stock appreciation rights, all restrictions on restricted stock and RSUs will lapse, and all performance goals or other vesting criteria for performance-based awards will be treated as achieved at 100% of target levels.
ImmunityBio, Inc. | 34 | 2026 Proxy Statement
ImmunityBio, Inc. 2015 Equity Incentive Plan
The 2015 Plan provides that in the event of a merger or change in control, as defined under the 2015 Plan, each then outstanding award will be treated as the administrator determines, except that if a successor corporation or its parent or subsidiary does not assume or substitute an equivalent award for any outstanding award, then such award will fully vest, all restrictions on the shares subject to such award will lapse, all performance goals or other vesting criteria applicable to the shares subject to such award will be deemed achieved at 100% of target levels and all of the shares subject to such award will become fully exercisable, if applicable, for a specified period prior to the transaction. The award will then terminate upon the expiration of the specified period of time.
2015 NantCell Stock Incentive Plan (NC 2015 Plan)
The 2015 NC Plan provides that in the event of a corporate transaction, as defined in the 2015 NC Plan, each outstanding award will terminate effective upon the consummation of such corporate transaction, except to the extent assumed or replaced in connection with such corporate transaction. The administrator may provide that awards granted under the 2015 NC Plan become vested and exercisable in connection with such a corporate transaction and release such awards from restrictions on transfer and repurchase or forfeiture rights.
| | | | | | | | | | | | | | |
| Potential Payments Upon Termination or Change in Control |
The employment agreements with Messrs. Adcock and Sachs and Ms. Lauer provide for payment of severance as follows:
| | | | | | | | | | | | | | |
| | Triggering Event |
| Name | | Termination
Without
Cause
($) | | Termination
Without
Cause or for
Good Reason
($) |
| | | | |
| Richard Adcock | | 835,800 | | | — | |
| David Sachs | | — | | | 790,159 | |
Patrick Soon-Shiong (1) | | — | | | — | |
| Regan Lauer | | 117,447 | | | — | |
_______________
(1)Dr. Soon-Shiong does not receive any form of severance in connection with his employment terms.
Our NEOs’ employment agreements do not provide for any other form of termination benefits, other than severance.
| | |
Policies and Practices Related to the Grant of Certain Equity Awards |
The Board and Compensation Committee do not take material non-public information into account when determining the timing and terms of any grants of equity awards, including option grants. The timing of any equity awards to recipients in connection with new hires, promotions, or other non-routine grants is tied to the event giving rise to the award (such as an employee’s commencement of employment or promotion effective date). We have not timed the disclosure of material non-public information for the purpose of affecting the value of executive compensation.
No option awards were issued to executive officers in the fiscal year ended December 31, 2025 during any period beginning four business days before the filing of a periodic report or Current Report on Form 8-K disclosing material non-public information and ending one business day after the filing or furnishing of such report with the SEC.
ImmunityBio, Inc. | 35 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
| Executive Compensation Recovery Policy |
Effective as of November 29, 2023, the Board adopted a compensation recovery policy, a copy of which was filed as Exhibit 97.1 to our Annual Report for the fiscal year ended December 31, 2023. The compensation recovery policy is intended to further the company’s pay-for-performance philosophy and to comply with applicable SEC and Nasdaq requirements by providing for recovery (i.e., a clawback) of certain executive compensation in the event of an accounting restatement. The application of the compensation recovery policy to executive officers is not discretionary, with limited exceptions, and applies without regard to whether an executive officer was at fault.
| | |
| Insider Trading Policies and Procedures |
ImmunityBio, Inc. and its subsidiaries have adopted a written Insider Trading Policy that applies to our employees, including our executive officers, and the non-employee members of our Board. Under this policy, these persons are prohibited from, directly or indirectly, among other things: (1) engaging in short sales, (2) trading in publicly-traded options, such as puts and calls, and other derivative securities with respect to our securities (other than RSUs, options, and other compensatory awards issued to such individuals by us), (3) purchasing financial instruments (including prepaid variable forward contracts, equity swaps, collars and exchange funds), or otherwise engaging in transactions that hedge or offset, any decrease in the market value of equity securities granted to them by us as part of their compensation or held, directly or indirectly, by them, (4) pledging any of our securities as collateral for any loans, and (5) holding our securities in a margin account.
We believe our Insider Trading Policy is reasonably designed to promote compliance with insider trading laws, rules and regulations, and applicable listing standards. A copy of our Insider Trading Policy was filed as Exhibit 19.1 to our Annual Report on Form 10-K for the year ended December 31, 2024.
| | | | | | | | | | | | | | |
| Tax and Accounting Considerations |
Deductibility of Executive Compensation
Section 162(m) of the Code, generally limits the amount a public company may deduct for federal income tax purposes for compensation paid to its chief executive officer, chief financial officer, and certain other executive officers up to $1 million per executive office per year. As a publicly-traded company, the Compensation Committee is mindful of the benefits of full deductibility of compensation, and intends to operate our executive compensation program to be most efficient and effective for our stockholders.
The Compensation Committee believes it is important to maintain cash and equity incentive compensation at the requisite level to attract and retain the individuals essential to our financial success, even if all or part of that compensation may not be deductible by reason of the Section 162(m) limitation.
Taxation of “Parachute” Payments
Sections 280G and 4999 of the Code provide that executive officers and directors who hold significant equity interests and certain other service providers may be subject to additional excise taxes if they receive payments or benefits in connection with a change in control of the company that exceeds certain prescribed limits, and that we (or a successor) may forfeit a deduction on the amounts subject to this additional tax.
Accounting for Stock-Based Compensation
The Compensation Committee takes accounting considerations into account when designing compensation plans and arrangements for our executive officers and other employees. Chief among these is Financial Accounting Standards Board (FASB) Accounting Standards Codification (ASC) Topic 718 (ASC 718), Stock Compensation (or any of its successors), the standard which governs the accounting treatment of stock-based compensation awards.
The grant date fair value of our share-based payment awards is reported in the Summary Compensation Table below (or the year-end fair value or vesting date fair value in the case of the Pay Versus Performance Tables), even though recipients may never realize any value from their equity awards.
ImmunityBio, Inc. | 36 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
| Compensation Committee Report |
The Compensation Committee has reviewed and discussed with management the Compensation Discussion and Analysis required by Item 402(b) of Regulation S-K. Based on this review and discussion, the Compensation Committee recommended to the Board that the section entitled “Compensation Discussion and Analysis” be included in this proxy statement and incorporated by reference in the company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2025.
| | |
| Compensation Committee of the Board of Directors |
|
| Linda Maxwell, M.D. (Chair) |
| Michael D. Blaszyk |
| Christobel Selecky |
ImmunityBio, Inc. | 37 | 2026 Proxy Statement
| | |
| Executive Compensation Tables |
| | | | | | | | | | | | | | |
| Summary Compensation Table |
The following table provides information regarding the compensation of our PEO, PFO, and our two other named executive officers during the fiscal year ended December 31, 2025, together referred to as our NEOs, for the fiscal years ended December 31, 2025, 2024 and 2023. Our NEOs are our only executive officers.
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
Name and
Principal Position
(a) | | Year
(b) | | Salary
($) (1)
(c) | | Bonus
($) (2)
(d) | | Stock
Awards
($) (3)
(e) | | Option
Awards
($) (4)
(f) | | Non-Equity
Incentive Plan
Compensation
($) (5)
(g) | | All Other
Compensation
($) (6)
(i) | | Total ($)
(j) |
| | | | | | | | | | | | | | | | |
| Richard Adcock | | 2025 | | 831,667 | | | 141,684 | | | 2,787,828 | | | 8,353,406 | | | 485,166 | | | 117,202 | | | 12,716,953 | |
President and
CEO | | 2024 | | 802,123 | | | — | | | 2,396,341 | | | 7,214,767 | | | 477,600 | | | 64,044 | | | 10,954,875 | |
| | 2023 | | 796,000 | | | 298,500 | | | — | | | — | | | — | | | 62,403 | | | 1,156,903 | |
| David Sachs | | 2025 | | 591,711 | | | 66,974 | | | 836,347 | | | 2,506,022 | | | 229,336 | | | 14,153 | | | 4,244,543 | |
| CFO | | 2024 | | 574,515 | | | — | | | 639,023 | | | 1,923,937 | | | 230,143 | | | 13,989 | | | 3,381,607 | |
| | 2023 | | 537,525 | | | 273,980 | | | 233,457 | | | — | | | — | | | 13,120 | | | 1,058,082 | |
| Patrick Soon-Shiong, M.D. | | 2025 | | 648,826 | | | 110,535 | | | 2,787,828 | | | 8,353,406 | | | 378,503 | | | 14,157 | | | 12,293,255 | |
Global Chief
Scientific and
Medical Officer | | 2024 | | 625,777 | | | — | | | 1,797,252 | | | 5,411,074 | | | 372,600 | | | 13,991 | | | 8,220,694 | |
| | 2023 | | 621,000 | | | 232,875 | | | — | | | — | | | — | | | 11,465 | | | 865,340 | |
Regan Lauer (7) | | 2025 | | 338,773 | | | 38,344 | | | 55,755 | | | 167,068 | | | 131,302 | | | 14,071 | | | 745,313 | |
Chief Accounting
Officer | | | | | | | | | | | | | | | | |
_______________
(1)Amounts shown in Column (c) for the fiscal year ended December 31, 2025 reflect the base salary earned during the year, including salary increases effective during the year, if any.
(2)Amounts shown in Column (d) for the fiscal year ended December 31, 2025 reflect discretionary cash bonuses awarded by the Compensation Committee (approximately 23% of target for Messrs. Adcock and Sachs, Dr. Soon-Shiong, and Ms. Lauer) earned in 2025 that were paid in 2026 based on the performance of each NEO in 2025 and after taking any relevant factors into consideration. Amounts shown for the fiscal year ended December 31, 2023 reflect discretionary cash bonuses awarded by the Compensation Committee (75.0% of target for Mr. Adcock and Dr. Soon-Shiong, respectively, and 100.0% of target for Mr. Sachs) earned in 2023 that were paid in 2024 based on the performance of each NEO in 2023 and after taking any relevant factors into consideration. No discretionary cash bonuses were earned by our NEOs during the fiscal year ended December 31, 2024.
(3)Amounts shown in Column (e) reflect the grant date fair value of RSU awards calculated on the basis of the fair market value of the underlying shares of common stock on the grant date in accordance with ASC 718. The company did not grant RSU awards to Mr. Adcock and Dr. Soon-Shiong during the fiscal year ended December 31, 2023.
(4)Amounts shown in Column (f) reflect the grant date fair value of option awards as determined in accordance with ASC 718 using the Black-Scholes option pricing model with the following key assumptions: risk-free interest rate of 4.3%; expected term of 6.0 years; expected volatility of the price of our common stock of 120.2%; dividend yield of 0.0%; and an exercise price of $3.39 per share, the closing price of the company’s common stock as reported on the Nasdaq on February 10, 2025, the date of grant. The company did not grant option awards to Messrs. Adcock and Sachs and Dr. Soon-Shiong during the fiscal year ended December 31, 2023.
(5)Amounts shown in Column (g) for the fiscal year ended December 31, 2025 reflect performance-based cash awards awarded by the Compensation Committee (approximately 77% of target for Messrs. Adcock and Sachs, Dr. Soon-Shiong, and Ms. Lauer) earned in 2025 that were paid in 2026 based on metrics in the 2025 STIP. Amounts shown for the fiscal year ended December 31, 2024 reflect performance-based cash awards awarded by the Compensation Committee (80% of target for Messrs. Adcock and Sachs, and Dr. Soon-Shiong) earned in 2024 that were paid in 2025 based on metrics in the 2024 STIP. No performance-based cash awards were earned by our NEOs during the fiscal year ended December 31, 2023. See “Compensation Discussion and Analysis—Short-Term Incentive Compensation Program” above, for additional information regarding our 2025 performance-based cash awards. (7)Ms. Lauer was appointed an NEO in the fiscal year ended December 31, 2025.
ImmunityBio, Inc. | 38 | 2026 Proxy Statement
| | |
All Other Compensation–Other Compensation and Perquisites |
The following table provides the detail of all other compensation provided to our NEOs during the fiscal year ended December 31, 2025:
| | | | | | | | | | | | | | | | | | | | |
| Name | | Company
Contributions to
Retirement Plans (1)
($) | | Reimbursement of
Personal Life
Insurance
Premiums (2)
($) | | Total
($) |
| | | | | | |
| Richard Adcock | | 14,202 | | | 103,000 | | | 117,202 | |
| David Sachs | | 14,153 | | | — | | | 14,153 | |
| Patrick Soon-Shiong | | 14,157 | | | — | | | 14,157 | |
| Regan Lauer | | 14,071 | | | — | | | 14,071 | |
_______________
(1)Amounts shown reflect employer contributions made to the company’s 401(k) plan for our NEOs.
(2)Amount shown reflects reimbursement of premiums associated with Mr. Adcock’s personal life insurance policies pursuant to the terms of his Offer of Employment letter dated October 20, 2020 and related tax neutrality payments to make such reimbursements tax neutral to Mr. Adcock.
| | |
| Grants of Plan-Based Awards |
The following table sets forth summary information regarding grants of plan-based awards made to our NEOs during the fiscal year ended December 31, 2025:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | Estimated Future
Payouts Under
Non-Equity
Incentive
Plan Awards | | | | | | | | | | |
Name
(a) | | Grant Date
(b) | | Compensation
Committee
Approval
Date | | Target
($) (1)
(d) | | Maximum
($) (2)
(e) | | All Other
Stock
Awards:
Number of
Shares
of Stock
or Units
(#) (3)
(i) | | | | All Other
Option
Awards:
Number of
Securities
Underlying
Options
(#) (4)
(j) | | Exercise or
Base Price of
Option
Awards
($/Share)
(k) | | Grant Date
Fair Value of
Stock and
Option
Awards
($)
(l) |
| | | | | | | | | | | | | | | | | | |
| Richard Adcock | | 2/10/2025 | | 2/7/2025 | | 626,850 | | | 877,590 | | | | | | | | | | | |
| | 2/10/2025 | | 2/7/2025 | | | | | | 822,368 | | | | | | | | | 2,787,828 | |
| | 2/10/2025 | | 2/7/2025 | | | | | | | | | | 2,812,500 | | | 3.39 | | | 8,353,406 | |
| David Sachs | | 2/10/2025 | | 2/7/2025 | | 296,310 | | | 414,833 | | | | | | | | | | | |
| | 2/10/2025 | | 2/7/2025 | | | | | | 246,710 | | | | | | | | | 836,347 | |
| | 2/10/2025 | | 2/7/2025 | | | | | | | | | | 843,750 | | | 3.39 | | | 2,506,022 | |
| Patrick Soon-Shiong | | 2/10/2025 | | 2/7/2025 | | 489,038 | | | 684,653 | | | | | | | | | | | |
| | 2/10/2025 | | 2/7/2025 | | | | | | 822,368 | | | | | | | | | 2,787,828 | |
| | 2/10/2025 | | 2/7/2025 | | | | | | | | | | 2,812,500 | | | 3.39 | | | 8,353,406 | |
| Regan Lauer | | 2/10/2025 | | 2/7/2025 | | 169,646 | | | 237,504 | | | | | | | | | | | |
| | 2/10/2025 | | 2/7/2025 | | | | | | 16,447 | | | | | | | | | 55,755 | |
| | 2/10/2025 | | 2/7/2025 | | | | | | | | | | 56,250 | | | 3.39 | | | 167,068 | |
ImmunityBio, Inc. | 39 | 2026 Proxy Statement
_______________
(1)Amounts shown in Column (d) represent performance-based cash awards granted under the company’s 2025 STIP shown at 100% of target calculated as follows: for Mr. Adcock, 75% of his annual base salary ($835,800); for Mr. Sachs, 50% of his annual base salary ($592,619); for Dr. Soon-Shiong, 75% of his annual base salary ($652,050); and for Ms. Lauer, 50% of her annual base salary ($339,292). The company’s non-equity incentive plan for 2025 does not have a minimum payout. Performance-based cash awards earned by Messrs. Adcock and Sachs, Dr. Soon-Shiong, and Ms. Lauer during the fiscal year ended December 31, 2025 that were paid in 2026 are described in Note 5 of the Summary Compensation Table. See “Compensation Discussion and Analysis—Short-Term Incentive Compensation Program” above for additional information regarding our 2025 performance-based cash awards. (2)Amounts shown in Column (e) represent performance-based cash awards granted under the company’s 2025 STIP shown at 140% of target calculated as follows: for Mr. Adcock, 75% of his annual base salary ($835,800); for Mr. Sachs, 50% of his annual base salary ($592,619); for Dr. Soon-Shiong, 75% of his annual base salary ($652,050); and for Ms. Lauer, 50% of her annual base salary ($339,292). See “Compensation Discussion and Analysis—Short-Term Incentive Compensation Program” above for additional information regarding our 2025 performance-based cash awards. (3)Amounts shown in Column (i) represent RSU awards granted by the Compensation Committee pursuant to the 2025 LTIP. Subject to the recipient continuing to be a “service provider” (as defined in the 2015 Plan) through each applicable vesting date, 33.33% of the shares subject to the RSU award shall vest in equal annual installments on each of the first and second anniversaries of the vesting commencement date and 33.34% of the shares subject to the RSU award shall vest on the third anniversary of the vesting commencement date, such that all shares shall be fully vested on the third anniversary of the vesting commencement date. The vesting commencement date for the RSU awards is February 10, 2025.
Amounts shown in Column (l) represent the grant date fair value of RSU awards determined in accordance with ASC 718 based on the number of RSU awards granted multiplied by $3.39 per share, the closing price of the company’s common stock as reported on the Nasdaq on February 10, 2025, the date of grant.
(4)Amounts shown in Column (j) represent option awards to purchase shares of the company’s common stock granted by the Compensation Committee pursuant to the 2025 LTIP. Subject to the recipient continuing to be a “service provider” (as defined in the 2015 Plan) through each applicable vesting date, 33.33% of the shares subject to the option awards shall vest in equal annual installments on each of the first, second, and third anniversaries of the vesting commencement date, such that all shares shall be fully vested on the third anniversary of the vesting commencement date. The vesting commencement date for the option awards is February 10, 2025.
Amounts shown in Column (k) represent the exercise price of the option awards.
Amounts shown in Column (l) represent the grant date fair value of option awards determined in accordance with ASC 718 using the Black-Scholes option pricing model with the following key assumptions: risk-free interest rate of 4.3%; expected term of 6.0 years; expected volatility of the price of our common stock of 120.2%; dividend yield of 0.0%; and an exercise price of $3.39 per share, the closing price of the company’s common stock as reported on the Nasdaq on February 10, 2025, the date of grant.
ImmunityBio, Inc. | 40 | 2026 Proxy Statement
| | |
| Outstanding Equity Awards at Fiscal Year-End |
The following table sets forth summary information regarding the outstanding equity awards held by our NEOs as of December 31, 2025:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Option Awards | | Stock Awards |
Name
(a) | | Number of
Securities
Underlying
Unexercised
Options
Exercisable (#)
(b) | | Number of
Securities
Underlying
Unexercised
Options
Unexercisable (#)
(c) | | | | Option
Exercise
Price ($)
(e) | | Option
Expiration
Date
(f) | | Number of
Shares or Units
of Stock That
Have Not
Vested (#)
(g) | | Market Value
of Shares
or Units
of Stock
That Have
Not Vested
($) (1)
(h) | | | | |
| | | | | | | | | | | | | | | | | | | | | | |
| Richard Adcock | | 750,000 | | | — | | | | | | | 23.72 | | | 2/5/2031 | | | | | | | | | | |
| | | | | | | | | | | | | | 51,731 | | (2) | | 102,427 | | | | | | |
| | 700,000 | | | — | | | | | | | 5.83 | | | 3/23/2032 | | | | | | | | | | |
| | 530,487 | | | 1,060,976 | | (3) | | | | | 5.24 | | | 2/22/2034 | | | | | | | | | | |
| | | | | | | | | | | | | | 304,878 | | (4) | | 603,658 | | | | | | |
| | — | | | 2,812,500 | | (5) | | | | | 3.39 | | | 2/10/2035 | | | | | | | | | | |
| | | | | | | | | | | | | | 822,368 | | (6) | | 1,628,289 | | | | | | |
| David Sachs | | | | | | | | | | | | | | 45,602 | | (2) | | 90,292 | | | | | |
| | 250,000 | | | — | | | | | | | 5.83 | | | 3/23/2032 | | | | | | | | | | |
| | 141,463 | | | 282,927 | | (3) | | | | | 5.24 | | | 2/22/2034 | | | | | | | | | | |
| | | | | | | | | | | | | | 81,301 | | (4) | | 160,976 | | | | | |
| | — | | | 843,750 | | (5) | | | | | 3.39 | | | 2/10/2035 | | | | | | | | | | |
| | | | | | | | | | | | | | 246,710 | | (6) | | 488,486 | | | | | |
| Patrick Soon-Shiong | | 26,064 | | | — | | | | | | | 14.91 | | | 6/10/2031 | | | | | | | | | | |
| | 700,000 | | | — | | | | | | | 5.83 | | | 3/23/2032 | | | | | | | | | | |
| | 397,865 | | 795,732 | | (3) | | | | | 5.24 | | | 2/22/2034 | | | | | | | | | | |
| | | | | | | | | | | | | | 228,658 | | (4) | | 452,743 | | | | | | |
| | — | | | 2,812,500 | | (5) | | | | | 3.39 | | | 2/10/2035 | | | | | | | | | | |
| | | | | | | | | | | | | | 822,368 | | (6) | | 1,628,289 | | | | | | |
| Regan Lauer | | 14,286 | | | — | | | | | | | 5.83 | | | 3/23/2032 | | | | | | | | | | |
| | 14,146 | | | 28,293 | | (3) | | | | | 5.24 | | | 2/22/2034 | | | | | | | | | | |
| | | | | | | | | | | | | | 8,130 | | (4) | | 16,097 | | | | | | |
| | — | | | 56,250 | | (5) | | | | | 3.39 | | | 2/10/2035 | | | | | | | | | | |
| | | | | | | | | | | | | | 16,447 | | (6) | | 32,565 | | | | | | |
_______________
(1)Amounts shown in Column (h) represent the market value of the unvested RSUs shown in Column (g) based on the closing price of $1.98 per share of the company’s common stock as reported on the Nasdaq on December 31, 2025.
(2)Amount shown in Column (g) represents RSU awards granted on March 4, 2021. Subject to the recipient remaining in “continuous service” (as defined in the NC 2015 Plan) through the vesting date, the RSU awards shall vest in full on December 31, 2026.
(3)Amounts shown in Column (c) represent option awards granted on February 22, 2024. Subject to the recipient continuing to be a “service provider” (as defined in the 2015 Plan) through each applicable vesting date, 33.3% of the shares subject to the option awards shall vest on the second (February 22, 2026) anniversary of the vesting commencement date and 33.4% of the shares subject to the option awards shall vest on the third (February 22, 2027) anniversary of the vesting commencement date, such that all shares shall be fully vested on the third anniversary of the vesting commencement date.
ImmunityBio, Inc. | 41 | 2026 Proxy Statement
(4)Amounts shown in Column (g) represent RSU awards granted on February 22, 2024. Subject to the recipient continuing to be a “service provider” (as defined in the 2015 Plan) through each applicable vesting date, 33.3% of the shares subject to the RSU awards shall vest on the second (February 22, 2026) anniversary of the vesting commencement date and 33.4% of the shares subject to the RSU awards shall vest on the third (February 22, 2027) anniversary of the vesting commencement date, such that all shares shall be fully vested on the third anniversary of the vesting commencement date.
(5)Amounts shown in Column (c) represent option awards granted on February 10, 2025. Subject to the recipient continuing to be a “service provider” (as defined in the 2015 Plan) through each applicable vesting date, 33.33% of the shares subject to option awards shall vest in equal annual installments on each of the first, second, and third anniversaries of the vesting commencement date, such that all shares shall be fully vested on the third anniversary of the vesting commencement date. The vesting commencement date for the option awards is February 10, 2025.
(6)Amounts shown in Column (g) represent RSU awards granted on February 10, 2025. Subject to the recipient continuing to be a “service provider” (as defined in the 2015 Plan) through each applicable vesting date, 33.33% of the shares subject to the RSU awards shall vest in equal annual installments on each of the first and second anniversaries of the vesting commencement date and 33.34% of the shares subject to the RSU awards shall vest on the third anniversary of the vesting commencement date, such that all shares shall be fully vested on the third anniversary of the vesting commencement date. The vesting commencement date for the RSU awards is February 10, 2025.
| | | | | | | | | | | | | | |
| Option Exercises and Stock Vested |
The following table sets forth certain information concerning option exercises and stock vested for our NEOs during the fiscal year ended December 31, 2025:
| | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Option Awards | | Stock Awards |
Name
(a) | | Number of
Shares
Acquired on
Exercise (#)
(b) | | Value
Realized on
Exercise ($)
(c) | | Number of
Shares
Acquired on
Vesting (#) (1)
(d) | | Value
Realized on
Vesting ($) (2)
(e) |
| | | | | | | | |
| Richard Adcock | | — | | | — | | | 186,922 | | | 589,618 | |
| David Sachs | | — | | | — | | | 71,047 | | | 199,209 | |
| Patrick Soon-Shiong | | — | | | — | | | 114,329 | | | 391,005 | |
| Regan Lauer | | — | | | — | | | 4,065 | | | 13,902 | |
_______________
(1)For Mr. Adcock, the company withheld 95,677 shares to cover his tax withholding obligations. For Mr. Sachs, the company withheld 31,295 shares to cover his tax withholding obligations. For Dr. Soon-Shiong, the company withheld 42,071 shares to cover his tax withholding obligations. For Ms. Lauer, the company withheld 1,669 shares to cover her tax withholding obligations.
(2)Amounts shown in Column (e) represent the closing price of a share of our common stock on the date of vesting multiplied by the number of RSUs vested.
| | |
| Other Compensation Matters |
| | | | | | | | | | | | | | |
| 2025 Pay Ratio of CEO to Median Employee |
As required by Section 953(b) of the Dodd-Frank Wall Street Reform Act, and Item 402(u) of Regulation S-K, the following information describes the relationship of the annual total compensation of our employees and the annual total compensation of Mr. Adcock, our CEO and President, as of December 31, 2025:
| | | | | | | | | | | | | | |
| | 2025 Annual
Total Compensation
($) | | Pay Ratio
Estimate |
| | | | |
| Richard Adcock (President and CEO) | | 12,716,953 | | | |
| The median of all employees, other than Mr. Adcock | | 183,848 | | | |
| | | | 69 to 1 |
ImmunityBio, Inc. | 42 | 2026 Proxy Statement
To identify the median employee and determine the annual total compensation of the median employee and our CEO:
•We identified the median employee using our total employee population of 665 persons as of December 31, 2025 (including all employees, whether employed on a full-time, part-time, seasonal or temporary basis, but excluding employees based outside of the U.S). The company employs a de minimis number of employees located in Italy that constitute less than 4% of our total employee population.
•Under the relevant rules, we are required to identify the median employee by use of a “consistently applied compensation measure” (CACM). We chose a CACM that closely approximates the annual total direct compensation of our employees. Specifically, we identified the median employee by aggregating, for each employee as of December 31, 2025: (1) annual base pay, (2) annual target cash incentive opportunity, (3) the grant date fair value for equity awards granted during the fiscal year ended December 31, 2025, and (4) employer contributions made to the company’s 401(k) plan during the fiscal year ended December 31, 2025. In identifying the median employee, we annualized the compensation values of individuals who joined the company during the fiscal year ended December 31, 2025.
•All of the elements of such employee’s compensation for the fiscal year ended December 31, 2025 in accordance with the requirements of Item 402(c)(2)(x) of Regulation S-K were totaled, resulting in annual total compensation of $183,848.
This information is being provided for compliance purposes and is a reasonable estimate calculated in a manner consistent with the SEC rules, based on our internal records and the methodology described above. The SEC rules for identifying the median compensated employee allow companies to adopt a variety of methodologies, to apply certain exclusions and to make reasonable estimates and assumptions that reflect their employee populations and compensation practices. Accordingly, the pay ratio reported by other companies may not be comparable to the pay ratio reported above, as other companies have different employee populations and compensation practices and may use different methodologies, exclusions, estimates and assumptions in calculating their own pay ratios. Neither the Compensation Committee nor management of the company used the CEO Pay Ratio measure in making compensation decisions.
| | | | | | | | | | | | | | |
| 2025 Performance Measures |
Pursuant to Item 402(v) of Regulation S-K, the Compensation Committee identified the following Financial and Commercial performance measures used in our STIP and LTIP programs as representing the performance measures used to link “compensation actually paid” in 2025 to the company’s performance:
•Revenue
•Adjusted EBITDA
•Clinical Development Goals (1)
_______________
(1)The Clinical Development goals are an operational metric, not a financial performance measure.
For a description of these measures and their application to our compensation programs for 2025, please see our “Compensation Discussion and Analysis.”
ImmunityBio, Inc. | 43 | 2026 Proxy Statement
The following table sets forth information concerning: (1) the compensation of our PEO and the average compensation for our Non-PEO NEOs, both as reported in the Summary Compensation Table and with certain adjustments to reflect the “compensation actually paid” to such individuals, as defined under SEC rules, for each of the fiscal years ended December 31, 2025, 2024, 2023, 2022, and 2021, (2) our cumulative total shareholder return (TSR), (3) the cumulative TSR for the Nasdaq Biotechnology Index, (4) our net loss, and (5) our Revenue over such years in accordance with SEC rules for each such fiscal year.
For information about our compensation philosophy and how we align executive compensation with our performance, please see our “Compensation Discussion and Analysis.”
| | |
| Pay Versus Performance Table |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | Value of Initial Fixed
$100 Investment Based on: | | | | |
Year
(a) | | Summary
Compensation
Table Total for
PEO ($)
(b) | | Compensation
Actually Paid
to PEO ($) (1)(2)
(c) | | Average
Summary
Compensation
Table Total
for Non-PEO
NEOs ($)
(d) | | Average
Compensation Actually Paid
to Non-PEO
NEOs ($) (1)(2)
(e) | | ImmunityBio
Total
Shareholder
Return ($) (3)
(f) | | Peer Group
Total
Shareholder
Return ($) (4)
(g) | | Net Loss
($M)
(h) | | Revenue
($M)
(i) |
| | | | | | | | | | | | | | | | |
2025 | | 12,716,953 | | | 6,222,909 | | | 5,761,037 | | | 2,920,536 | | | 14.85 | | | 119.92 | | | (351.5) | | | 113.3 | |
2024 | | 10,954,875 | | | 4,065,929 | | | 5,801,150 | | | 2,319,758 | | | 19.20 | | | 90.58 | | | (413.6) | | | 14.7 | |
2023 | | 1,156,903 | | | 262,372 | | | 961,711 | | | 622,399 | | | 37.66 | | | 91.84 | | | (583.9) | | | 0.6 | |
2022 | | 4,008,507 | | | 2,751,454 | | | 2,724,907 | | | 2,481,143 | | | 38.03 | | | 88.53 | | | (417.3) | | | 0.2 | |
2021 | | 29,016,727 | | | 10,356,288 | | | 2,888,272 | | | 1,170,403 | | | 45.61 | | | 99.37 | | | (349.8) | | | 0.9 | |
_______________
(1)As the valuation methods for “compensation actually paid” required by the SEC differ from those required in the Summary Compensation Table (SCT), the table below provides a reconciliation of the SCT amounts to the Compensation Actually Paid (CAP) amounts in the Pay Versus Performance table for Mr. Adcock and the average SCT and CAP amounts for our other NEOs for each of the last five years. The other Non-PEO NEOs for 2025 include Mr. Sachs, Dr. Soon-Shiong, and Ms. Lauer. The other Non-PEO NEOs for each of 2024, 2023, 2022, and 2021 include Mr. Sachs and Dr. Soon-Shiong.
Summary Compensation Table to Compensation Actually Paid Reconciliation Table
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | Required Adjustments to Summary Compensation Table Totals to Determine “Compensation Actually Paid” | | |
| | | | Summary
Compensation
Table Total ($) | | Summary
Compensation
Table Value
of Stock
Awards ($) | | Summary
Compensation
Table Value
of Option
Awards ($) | | Fair Value
as of Year
End of
Equity
Awards
Granted
During the
Year ($) | | Year-Over-Year
Increase
(Decrease) in
Fair Value as
of Year End
of Unvested
Awards
Granted in
Prior Years ($) | | Increase
(Decrease)
From Prior
Year End
in Fair
Value of
Awards That
Vested
During the
Year ($) | | Compensation
Actually
Paid ($) |
| | Year | | (a) | | (b) | | (c) | | (d) | | (e) | | (f) | | (g)=(a)-(b)-(c)
+(d)+(e)+(f) |
| | | | | | | | | | | | | | | | |
Richard
Adcock | | 2025 | | 12,716,953 | | | 2,787,828 | | | 8,353,406 | | | 4,780,201 | | | (626,555) | | | 493,544 | | | 6,222,909 | |
Average
of Other
NEOs | | 2025 | | 5,761,037 | | | 1,226,643 | | | 3,675,499 | | | 2,103,288 | | | (216,284) | | | 174,637 | | | 2,920,536 | |
(2)The fair value or change in fair value, as applicable, of equity awards was determined in accordance with ASC 718 by reference to (1) for RSU awards, the closing price of our common stock on the applicable measurement date and (2) for option awards, a Black-Scholes valuation model with updated assumptions for time to expiration, closing price of our common stock, volatility, and risk-free rate as of the applicable measurement date.
ImmunityBio, Inc. | 44 | 2026 Proxy Statement
(3)Pursuant to SEC rules, TSR reflects an initial investment of $100 in the company’s common stock on December 31, 2021. Cumulative TSR is calculated by dividing the sum of the cumulative amount of dividends for the measurement period (December 31, 2021 – December 31, 2025), assuming dividend reinvestment, and the difference between the share price at the end and the beginning of the measurement period by the share price at the beginning of the measurement period. The company has never paid cash dividends on its common stock.
(4)Peer Group TSR reflects an initial investment of $100 in the Nasdaq Biotechnology Index on December 31, 2021, which is the same index we used in the Stock Performance Graph in our Annual Report. Cumulative TSR is calculated by dividing the sum of the cumulative amount of dividends for the measurement period (December 31, 2021 – December 31, 2025), assuming dividend reinvestment, and the difference between the share price at the end and the beginning of the measurement period by the share price at the beginning of the measurement period.
| | |
| Compensation Actually Paid and Performance Measures |
Relationship between Financial Performance Measures
The graphs below compare the compensation actually paid to our PEO and the average of the compensation actually paid to our Non-PEO NEOs versus (i) our cumulative TSR, (ii) cumulative TSR for the Nasdaq Biotechnology Index, (iii) our net loss, and (iv) our Revenue, in each case, for the fiscal years ended December 31, 2025, 2024, 2023, 2022, and 2021, respectively.
TSR amounts reported in the graph assume an initial fixed investment of $100 on December 31, 2021, and that all dividends, if any, were reinvested.
(1) Compensation Actually Paid Compared to Total Shareholder Return
ImmunityBio, Inc. | 45 | 2026 Proxy Statement
(2) Compensation Actually Paid Compared to Net Loss
(3) Compensation Actually Paid Compared to Revenue
ImmunityBio, Inc. | 46 | 2026 Proxy Statement
The compensation policy for the company’s non-employee directors is intended to be competitive and fair so that we can attract the best talent to the Board. In addition to cash compensation, we provide equity awards and have stock ownership guidelines to align the directors’ interests with those of our stockholders and to focus on the long-term growth of the company. We reimburse our directors for expenses associated with attending Board and committee meetings. Directors who are employees of the company receive no compensation for serving on the Board, attending Board meetings or, in the case of Dr. Soon-Shiong, serving as the Executive Chairman of the Board and participating on Board committees. All cash payments to non-employee directors are paid quarterly in arrears on a prorated basis.
| | | | | | | | | | | | | | |
2025 Director Compensation Policy |
| | | | | | | | | | | | | | |
| Position | | Annual
Cash
Retainer
($) | | Equity
Awards
(Grant Date
Fair Value)
($) |
| | | | |
Executive Chairman of the Board (1) | | 35,000 | | | |
| | | | |
| Non-Employee Director Annual Base Retainer: | | 50,000 | | | |
| Lead Independent Director (Additional Retainer) | | 30,000 | | | |
| Initial option grant upon appointment to the Board | | | | 300,000 | |
| Annual option grant subject to continuing as a non-employee director | | | | 400,000 | |
| | | | |
| Committee Member Retainers: | | | | |
| Audit Committee | | 12,500 | | | |
| Compensation Committee | | 10,000 | | | |
| Nominating and Corporate Governance Committee | | 7,500 | | | |
| Related Party Transaction Committee | | 7,500 | | | |
| Special Committee | | 15,000 | | | |
| | | | |
| Committee Chair Additional Retainers: | | | | |
| Audit Committee | | 12,500 | | | |
| Compensation Committee | | 10,000 | | | |
| Nominating and Corporate Governance Committee | | 7,500 | | | |
| Related Party Transaction Committee | | 7,500 | | | |
| Special Committee | | 15,000 | | | |
_______________
(1)Dr. Soon-Shiong does not receive compensation for his services as Executive Chairman of the Board as he is an employee of the company.
There have been no changes to the Director Compensation Policy for 2026 as compared to the 2025 policy. The Special Committee was dissolved in the first half of 2026.
| | | | | | | | | | | | | | |
| Other Compensation Details |
Our Director Compensation Policy provides that in any given fiscal year, a non-employee director may not be paid, issued or granted cash retainer fees and equity awards with an aggregate value greater than $750,000 (increased to $1,000,000 in connection with the non-employee director’s initial year of service), with the value of each equity award based on its grant date fair value (as determined according to U.S. GAAP) for purposes of this limit. Any cash compensation paid, or awards granted to an individual for his or her services as an employee or consultant (other than as a non-employee director) will not count toward this limit.
ImmunityBio, Inc. | 47 | 2026 Proxy Statement
With respect to the automatic initial grant to new non-employee directors, each grantee shall receive an initial option award to purchase shares of our common stock at a per-share exercise price equal to the fair market value of a share of our common stock on the first trading date on which the trading window under our Insider Trading Policy opens after becoming a non-employee director, and such option shall have a Black-Scholes value of $300,000 as computed in accordance with ASC 718 on the grant date. The initial award shall vest in three equal installments on each anniversary of the date the applicable non-employee director’s service commenced, in each case subject to the non-employee director continuing to be a service provider through the applicable vesting date.
With respect to the automatic annual grants to all continuing non-employee directors, such grant will be made on the date of each annual meeting of stockholders, and such option shall have a Black-Scholes value of $400,000 as computed in accordance with ASC 718 on the grant date. The annual award will vest on the earlier of the one-year anniversary of the date the annual award is granted, or the day prior to the date of next the annual meeting following the date the annual award is granted, in each case, subject to the non-employee director continuing to be a service provider through the applicable vesting date.
The 2015 Plan and 2025 Plan each provide that in the event of a merger or change in control, as defined in the applicable plan, each outstanding equity award granted under the applicable plan that is held by a non-employee director will fully vest, all restrictions on the shares subject to such award will lapse, and all of the shares subject to such award will become fully exercisable, if applicable.
| | | | | | | | | | | | | | |
| Non-Employee Director Stock Ownership Policy |
Each non-employee director is expected to have equity holdings of the company with a value equivalent of at least three times his or her annual base retainer (not including retainers for serving as Chairperson of the Board, Lead Independent Director or as a member or Chair of any Board committee) and to maintain this minimum amount of stock ownership throughout his or her tenure on the Board. Non-employee Board members who were members of the Board on or before December 1, 2020 are expected to achieve the applicable level of stock ownership on or before the fifth anniversary date of December 1, 2020. Non-employee directors who joined the Board after December 1, 2020 are expected to achieve the applicable level of stock ownership on or before five-year anniversary of joining the Board. Minimum equity holdings can be satisfied by the following: shares directly-owned by the non-employee director; shares indirectly-held by the non-employee director or his or her immediate family members; shares subject to RSUs or other awards that have vested for which the non-employee has elected to defer the settlement to a date beyond the vesting date; and shares underlying RSUs and other awards that are vested or unvested for which the only requirement to earn the award is continued service to the company or its subsidiaries.
As of December 31, 2025, due in part to volatility in our stock price, three of our non-employee directors, consisting of Gen. Clark, Dr. Maxwell, and Ms. Selecky, were not in compliance with our Non-Employee Director Stock Ownership Policy. The Board is assessing the appropriate procedure to ensure that each of our non-employee directors will achieve compliance with our Non-Employee Director Stock Ownership Policy going forward.
ImmunityBio, Inc. | 48 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
| Director Compensation Table |
The following table sets forth information regarding compensation earned by our non-employee directors during the fiscal year ended December 31, 2025. Dr. Soon-Shiong, Mr. Adcock and Dr. Simon are not included in the table below as they are employees of the company and thus receive no compensation for their services as directors.
| | | | | | | | | | | | | | | | | | | | | | | | | | |
Name
(a) | | Fees Earned or
Paid in Cash ($)
(b) | | Option Awards
($) (1)
(d) | | All Other
Compensation ($)
(g) | | Total ($)
(h) |
| | | | | | | | |
| Michael D. Blaszyk | | 113,384 | | | 400,000 | | | — | | | 513,384 | |
John Owen Brennan (2) | | — | | | — | | | 272,500 | | | 272,500 | |
| Wesley Clark | | 70,589 | | | 400,000 | | | — | | | 470,589 | |
| Cheryl L. Cohen | | 114,192 | | | 400,000 | | | — | | | 514,192 | |
| Linda Maxwell | | 89,192 | | | 400,000 | | | — | | | 489,192 | |
| Christobel Selecky | | 77,281 | | | 400,000 | | | — | | | 477,281 | |
Bruce Wendel (3) | | 2,740 | | | 300,000 | | | — | | | 302,740 | |
_______________
(1)Amounts shown in Column (d) represent the aggregate grant date fair value of 162,786 options granted on June 18, 2025 to non-employee directors upon their re-election to the Board calculated in accordance with ASC 718 without regard to estimated forfeitures. See Note 18, Stock-Based Compensation, of the “Notes to Consolidated Financial Statements” that appear in Part II, Item 8. “Financial Statements and Supplementary Data” of the company’s Annual Report for the fiscal year ended December 31, 2025 filed with the SEC on February 23, 2026 for a discussion of valuation assumptions made in determining the grant date fair value of option awards.
(2)Mr. Brennan resigned as a director of the company as of February 7, 2025. Amount shown in Column (g) represents consulting fees paid to Mr. Brennan subsequent to his resignation from the Board.
(3)Mr. Wendel was appointed to the Board on December 12, 2025.
As of December 31, 2025, our non-employee directors held the following outstanding option awards:
•Each of Mr. Blaszyk and Ms. Cohen held 100,000 vested options exercisable at $6.21 per share, 26,064 vested options exercisable at $14.91 per share, 167,937 vested options exercisable at $2.98 per share, 158,263 vested options exercisable at $2.99 per share, 77,633 vested options exercisable at $5.96 per share, and 162,786 unvested options at an exercise price of $2.84 per share;
•Each of Gen. Clark, Dr. Maxwell and Ms. Selecky held 21,873 vested options exercisable at $17.24 per share, 26,064 vested options exercisable at $14.91 per share, 167,937 vested options exercisable at $2.98 per share, 158,263 vested options exercisable at $2.99 per share, 77,633 vested options exercisable at $5.96 per share, and 162,786 unvested options at an exercise price of $2.84 per share; and
•Mr. Wendel held 146,020 unvested options at an exercise price of $2.36 per share.
ImmunityBio, Inc. | 49 | 2026 Proxy Statement
| | |
| Security Ownership of Certain Beneficial Owners, Directors, and Named Executive Officers |
The following table sets forth certain information, as of April 13, 2026, with respect to the holdings of (i) each person who is the beneficial owner of more than 5% of our common stock, (ii) each of our directors, (iii) each executive officer, and (iv) all of our current directors and executive officers as a group. Beneficial ownership representing less than 1% is denoted with an asterisk (*) in the table below.
Beneficial ownership of our common stock is determined in accordance with the rules of the SEC and includes any shares of common stock over which a person exercises sole or shared voting or investment power, or of which a person has a right to acquire ownership at any time within 60 days of April 13, 2026.
Except as otherwise indicated, we believe that the persons named in this table have sole voting and investment power with respect to all shares of common stock held by them. Applicable percentage ownership in the following table is based on 1,047,345,861 shares of common stock outstanding as of April 13, 2026 plus, for each individual, any securities that individual has the right to acquire within 60 days of that date.
| | | | | | | | | | | | | | |
| Name and Address of Beneficial Owner (1) | | Number of
Shares of
Common
Stock
Beneficially
Owned | | Percentage
of
Common
Stock
Beneficially
Owned |
| | | | |
| 5% Stockholders: | | | | |
Patrick Soon-Shiong, M.D., FRCS (C), FACS (2) | | 745,518,285 | | 65.5 | % |
Nant Capital, LLC (3) | | 339,465,528 | | 29.9 | % |
Cambridge Equities, LP (4) | | 261,705,814 | | 25.0 | % |
California Capital Equity, LLC (5) | | 106,511,412 | | 10.2 | % |
NantWorks, LLC (6) | | 98,535,253 | | 9.4 | % |
| Other Directors and Named Executive Officers: | | | | |
Richard Adcock (7) | | 4,008,818 | | * |
David Sachs (8) | | 1,114,319 | | * |
Michael D. Blaszyk (9) | | 795,479 | | * |
Wesley Clark (10) | | 459,770 | | * |
Cheryl L. Cohen (11) | | 730,514 | | * |
Regan Lauer (12) | | 177,203 | | * |
Linda Maxwell, M.D., M.B.A., FRCSC (13) | | 451,770 | | * |
Christobel Selecky (14) | | 351,770 | | * |
Barry J. Simon, M.D. (15) | | 3,167,631 | | * |
Bruce Wendel (16) | | — | | * |
All directors and executive officers as a group (11 persons) (17) | | 756,775,559 | | 66.1 | % |
_______________
(1)The address of each of the individuals named in the table above is c/o ImmunityBio, Inc., 3530 John Hopkins Court, San Diego, California 92121. The address of each of the entities named in the table above is 450 Duley Road, El Segundo, California 90245.
(2)Consists of (i) 261,705,814 shares held by Cambridge; (ii) 29,757,911 shares directly held by Dr. Soon-Shiong; (iii) 251,018,873 shares held by Nant Capital, LLC (Nant Capital); (iv) 47,557,934 shares held by NantMobile, LLC (NantMobile); (v) 32,606,985 shares held by NantCancerStemCell, LLC (NCSC); (vi) 9,986,920 shares held by NantWorks; (vii) 8,383,414 shares held by NantBio, Inc. (NantBio); (viii) 7,976,159 shares held by California Capital Equity, LLC (California Capital); (ix) 5,618,326 shares held by the Chan Soon-Shiong Family Foundation; (x) 88,446,655 shares that Nant Capital has the right to acquire within 60 days of April 13, 2026 pursuant to the conversion of a promissory note; and (xi) 2,459,294 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
ImmunityBio, Inc. | 50 | 2026 Proxy Statement
(3)Consists of (i) 251,018,873 shares held by Nant Capital and (ii) 88,446,655 shares that Nant Capital has the right to acquire within 60 days of April 13, 2026 pursuant to the conversion of a promissory note. Dr. Soon-Shiong is the sole member of Nant Capital and thus may be deemed to have beneficial ownership of and voting and dispositive power over the shares held by Nant Capital.
(4)Consists of 261,705,814 shares held by Cambridge. MP 13 Ventures, LLC (MP 13 Ventures) is the general partner of Cambridge and may be deemed to have beneficial ownership of the shares held by Cambridge. Dr. Soon-Shiong is the sole member of MP 13 Ventures and has voting and dispositive power over the shares held by Cambridge.
(5)Consists of (i) 7,976,159 shares held by California Capital; (ii) 47,557,934 shares held by NantMobile; (iii) 32,606,985 shares held by NCSC; (iv) 9,986,920 shares held by NantWorks; and (v) 8,383,414 shares held by NantBio. Dr. Soon-Shiong owns all of the equity interests in California Capital and has voting and dispositive power over the shares held by California Capital.
(6)Consists of (i) 9,986,920 shares held by NantWorks; (ii) 8,383,414 shares held by NantBio; (iii) 47,557,934 shares held by NantMobile; and (iv) 32,606,985 shares held by NCSC. NantWorks is the majority stockholder of NantBio, NantMobile, and NCSC and thus may be deemed to control NantBio, NantMobile, and NCSC. NantBio is the sole member of NCSC. Dr. Soon-Shiong indirectly beneficially owns equity interests in NantWorks and thus may be deemed to control NantWorks and each entity directly or indirectly controlled by NantWorks (including NantBio, NantMobile, and NCSC) and has voting and dispositive power over the shares held by NantWorks.
(7)Consists of (i) 560,344 shares directly held by Mr. Adcock and (ii) 3,448,474 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
(8)Consists of (i) 300,143 shares directly held by Mr. Sachs and (ii) 814,176 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
(9)Consists of (i) 193,667 shares directly held by Mr. Blaszyk; (ii) 71,915 shares indirectly held; and (iii) 529,897 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
(10)Consists of (i) 8,000 shares directly held by Gen. Clark and (ii) 451,770 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
(11)Consists of (i) 200,617 shares directly held by Ms. Cohen and (ii) 529,897 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
(12)Consists of (i) 115,875 shares directly held by Ms. Lauer and (ii) 61,328 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
(13)Consists of 451,770 shares issuable to Dr. Maxwell upon the exercise of options that were exercisable as of April 13, 2026.
(14)Consists of 351,770 shares issuable to Ms. Selecky upon the exercise of options that were exercisable as of April 13, 2026.
(15)Consists of (i) 2,850,821 shares directly held by Dr. Simon and (ii) 316,810 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
(16)Mr. Wendel was appointed to the Board on December 12, 2025. He does not hold any shares of our common stock nor does he have any options that were exercisable as of April 13, 2026.
(17)Consists of (i) 33,987,378 shares directly held; (ii) 624,926,340 shares indirectly held; (iii) 88,446,655 shares that Nant Capital has the right to acquire within 60 days of April 13, 2026 pursuant to the conversion of a promissory note; and (iv) 9,415,186 shares issuable upon the exercise of options that were exercisable as of April 13, 2026.
ImmunityBio, Inc. | 51 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
Proposal 2 – Ratification of Selection of Independent Registered Public Accountants |
At the Annual Meeting, stockholders are being asked to ratify the selection of Deloitte & Touche LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2026. Stockholder ratification of the selection of Deloitte & Touche LLP is not required by our Bylaws or other applicable legal requirements. However, the Audit Committee is submitting the selection of Deloitte & Touche LLP to our stockholders because we value our stockholders’ views on our independent registered public accounting firm and as a matter of good corporate governance. If the selection is not ratified by our stockholders, the Audit Committee may reconsider whether it should appoint another independent registered public accounting firm. A representative of Deloitte & Touche LLP is expected to attend the Annual Meeting, where he or she will be available to respond to appropriate questions and, if he or she desires, to make a statement.
Required Vote
Ratification of the selection of Deloitte & Touche LLP as our independent registered public accounting firm for the fiscal year ending December 31, 2026 requires the affirmative “FOR” vote of a majority of the shares present live via the Internet or represented by proxy at the Annual Meeting and entitled to vote on the proposal. You may vote “FOR,” “AGAINST,” or “ABSTAIN” on this proposal. Abstentions have the same effect as a vote against the proposal.
| | |
THE BOARD RECOMMENDS A VOTE “FOR” THE RATIFICATION OF
THE SELECTION OF DELOITTE & TOUCHE AS IMMUNITYBIO’S
INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM. |
ImmunityBio, Inc. | 52 | 2026 Proxy Statement
The Audit Committee is a committee of the Board comprised solely of independent directors as required by Nasdaq corporate governance rules and the rules and regulations of the SEC. The Audit Committee operates under a written charter approved by the Board, which is available on our corporate website at ir.immunitybio.com. The composition of the Audit Committee, the attributes of its members and the responsibilities of the Audit Committee, as reflected in its charter, are intended to be in accordance with applicable requirements for corporate audit committees. The Audit Committee reviews and assesses the adequacy of its charter and the Audit Committee’s performance on an annual basis.
On March 13, 2025, the Audit Committee approved the dismissal of Ernst & Young LLP (EY) as the company’s independent registered public accounting firm. The dismissal was not related to any disagreements with EY on any matter of accounting principles or practices, financial statement disclosure, or auditing scope or procedure.
On March 13, 2025, the Audit Committee approved the engagement of Deloitte & Touche LLP (the Auditor) as the company’s independent registered public accounting firm for the fiscal year ending December 31, 2025.
With respect to its financial reporting process, the company’s management is responsible for (1) establishing and maintaining internal controls and (2) preparing its consolidated financial statements. The company’s independent registered public accounting firm is responsible for performing an independent audit of its consolidated financial statements in accordance with the auditing standards of the Public Company Accounting Oversight Board (United States) (PCAOB) and for issuing a report thereon. It is the responsibility of the Audit Committee to oversee these activities. It is not the responsibility of the Audit Committee to prepare the company’s consolidated financial statements. These are the fundamental responsibilities of management. In the performance of its oversight function, the Audit Committee has:
•Reviewed and discussed the audited financial statements for the fiscal year ended December 31, 2025 with the company’s management;
•Discussed with the Auditor the matters required to be discussed by the applicable requirements of Auditing Standard No. 1301, Communications with Audit Committees, as adopted by the PCAOB; and
•Received written disclosures and letters from the Auditor, as required by applicable requirements of the PCAOB regarding the independent registered public accounting firm’s communications with the Audit Committee concerning independence, and has discussed with the Auditor that firm’s independence.
Based on the Audit Committee’s review of the consolidated financial statements and various discussions with management and the Auditor, the Audit Committee recommended to the Board that the audited financial statements be included in the company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2025 for filing with the SEC.
| | |
| Audit Committee of the Board of Directors |
|
| Michael D. Blaszyk (Chair) |
| Cheryl L. Cohen |
| Linda Maxwell |
| Christobel Selecky |
ImmunityBio, Inc. | 53 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
| Fees Paid to Independent Registered Public Accounting Firm |
The following table presents fees for services provided by or to be provided by the Auditor for the audit of the fiscal year ended December 31, 2025, and fees for other services rendered by the Auditor during this period:
| | | | | | | | | | |
| | Year Ended
December 31,
2025 | | |
| | | |
| | | | |
Audit fees (1) | | $ | 2,775,615 | | | |
| Audit-related fees | | — | | | |
Tax fees (2) | | 43,668 | | | |
All other fees (3) | | 3,769 | | | |
| Total | | $ | 2,823,052 | | | |
_______________
(1)Audit fees consist of fees incurred for professional services provided by our independent registered public accounting firm for audit and quarterly reviews of our consolidated financial statements, including the audit of internal control over financial reporting, and related services that are normally provided in connection with statutory and regulatory filings or engagements including procedures relating to offering memorandums.
(2)Tax fees consist of various permissible tax compliance and tax advisory services fees provided by our independent registered public accounting firm.
(3)Amount relates to accounting research database subscription services.
Pre-Approval Policies and Procedures
Our Audit Committee’s policy is to pre-approve all audit and permissible non-audit services provided by the independent registered accounting firm. These services may include audit services, audit-related services, tax services, and other services. The Audit Committee pre-approves services or categories of services. The independent registered public accounting firm and the company’s management are required to periodically report to the Audit Committee regarding the extent of services provided by the independent registered public accounting firm in accordance with these pre-approvals, and the fees for the services performed to date. All services provided to us by and audit fees paid to Deloitte & Touche LLP during the fiscal year ended December 31, 2025 described above were pre-approved by the Audit Committee.
Auditor Independence
During the fiscal year ended December 31, 2025, all professional services provided were considered by the Audit Committee for compatibility with maintaining the independence of our independent registered public accounting firm.
ImmunityBio, Inc. | 54 | 2026 Proxy Statement
| | |
| Annual Report on Form 10-K |
Our Annual Report on Form 10-K, which contains the consolidated financial statements of the company for the fiscal year ended December 31, 2025, accompanies this proxy statement, but is not part of the company’s soliciting materials.
Stockholders may obtain, without charge, a copy of the company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2025 filed with the SEC on February 23, 2026, including the consolidated financial statements and schedules thereto, without the accompanying exhibits, by writing to: Investor Relations at our principal executive offices, contacting Broadridge by telephone at 800-579-1639, or sending an email to sendmaterial@proxyvote.com (including your control number in the subject line). The company’s Annual Report on Form 10-K is also available online on our website at ir.immunitybio.com (1) under “SEC Filings” and the SEC website at sec.gov.
_______________
(1)Reference to our website is not intended to function as a hyperlink and the information contained on our website is not intended to be part of this proxy statement.
ImmunityBio, Inc. | 55 | 2026 Proxy Statement
| | |
| Certain Relationships and Related-Party Transactions |
The following is a summary of transactions since January 1, 2023 to which we have been, or plan to be, a party in which the annual amount involved exceeded, or is expected to exceed, $120,000 and in which any of our executive officers, directors, promoters or beneficial holders of more than 5% of our capital stock had or will have a direct or indirect material interest, other than compensation arrangements which are described under the section of this proxy statement titled “Executive Compensation.” For information regarding Cambridge, including the basis of control and the percentage of voting securities owned, see “Security Ownership of Certain Beneficial Owners, Directors, and Named Executive Officers.” | | | | | | | | | | | | | | |
| Policies for Approval of Related-Party Transactions |
We have adopted a written Related Party Transactions Policy that sets forth our policies and procedures regarding the identification, review, consideration, approval and oversight of related-party transactions. For purposes of our policy only, a “related-party transaction” is a past, present, or future transaction, arrangement or relationship (or any series of similar transactions, arrangements or relationships) in which we and any “related person” are participants, the amount involved exceeds $120,000, and in which a related person had, has, or will have a direct or indirect material interest. Various transactions are not covered by this policy, including transactions involving compensation for services provided to us as an employee or director. A “related party” includes any executive officer, director or nominee to become director, and any holder of more than 5% of our common stock, including any immediate family members of such persons. Any related-party transaction may only be consummated if approved by the Related Party Transaction Committee in accordance with the policy guidelines set forth below.
Under the policy, where a transaction has been identified as a related-party transaction, management must present information regarding the proposed related-party transaction to the Related Party Transaction Committee for review and approval during a committee meeting. In considering related-party transactions, the Related Party Transaction Committee takes into account the relevant available facts and circumstances including, but not limited to whether the terms of such transaction are no less favorable than terms generally available to an unaffiliated third-party under the same or similar circumstances and the extent of the related person’s interest in the transaction.
On December 23, 2014, we entered into a subscription and investment agreement (Cambridge Subscription Agreement), a registration rights agreement (Cambridge Registration Rights Agreement), and a reclassification agreement (Reclassification Agreement) with Cambridge relating to the private placement of our Class A common stock. In the private placement, we issued to Cambridge an aggregate of 25,191,473 shares of Class A common stock at a price of $1.89 per share. We received aggregate gross proceeds of $47.5 million from Cambridge’s investment. Dr. Soon-Shiong, our Founder, Executive Chairman and Global Chief Scientific and Medical Officer, controls the entity that is the general partner of Cambridge, which beneficially owned approximately 25.0% of our common stock as of April 13, 2026.
Cambridge Subscription Agreement. Cambridge agreed in the Cambridge Subscription Agreement that, until the earlier of the consummation of our initial public offering (IPO) and December 23, 2015, neither it nor any of its affiliates shall acquire, including by way of the acquisition of control of another entity, beneficial ownership of any shares of our common stock which, when aggregated with all of the other shares of our common stock beneficially owned by Cambridge and its affiliates, would cause the total number of shares of our common stock beneficially owned by Cambridge and its affiliates to exceed 49.9% of our outstanding shares of common stock. The Cambridge Subscription Agreement was amended pursuant to a letter agreement dated January 20, 2015, to remove the limitation on Class A common stock beneficially owned by Cambridge in exchange for Cambridge agreeing to vote its shares in favor of certain matters approved by a majority of the Board.
ImmunityBio, Inc. | 56 | 2026 Proxy Statement
Cambridge Registration Rights Agreement. Under the terms of the Cambridge Registration Rights Agreement, we provided Cambridge with a right to demand registration of the shares of common stock issued under the Cambridge Subscription Agreement. We also granted to Cambridge “piggyback” registration rights exercisable at any time that allow them to include the shares of our common stock that they own in any public offering of equity securities initiated by us for our own account or the account of others (other than those public offerings pursuant to registration statements on forms that do not permit registration for resale by them). These “piggyback” registration rights are not available with respect to any shares of our common stock held by Cambridge which are eligible for resale pursuant to certain exemptions from registration under the Securities Act of 1933, as amended (the Securities Act) or that are the subject of a then-effective registration statement.
Cambridge Reclassification Agreement. Pursuant to the Reclassification Agreement, we agreed together with Cambridge, Bio IP Ventures, LLC, and Bonderman Family Limited Partnership subject to the effectiveness of certain transactions, to take all necessary actions and to vote such shares necessary to convert all of our issued and outstanding shares of Series B preferred stock into Class A common stock, all of our issued and outstanding Series C preferred stock into Class B common stock, and to reclassify all of our Series B preferred stock, Series C preferred stock and Class B common stock into our Class A common stock by filing an amendment to our Certificate of Incorporation.
Cambridge Nominating Agreement. On June 18, 2015, we entered into the Cambridge Nominating Agreement, pursuant to which Cambridge has the right to designate one individual to be nominated and recommended for election by the Board for as long as Cambridge and/or its affiliates directly own more than 20% of the issued and outstanding shares of our common stock, subject to adjustment for stock splits, stock dividends, recapitalizations and similar transactions. Dr. Soon-Shiong, who controls the entity that is the general partner of Cambridge and has the sole power to vote or direct to vote and the sole power to dispose or direct the disposition, was selected by Cambridge to hold this Board seat. The Cambridge director nominee shall be nominated and recommended for election by the Board or other duly authorized committee, subject to any applicable limitations imposed by the DGCL, the Board’s fiduciary duties to our stockholders and any other applicable law. Cambridge’s right to have a designee nominated or appointed to serve on the Board shall automatically terminate whenever Cambridge owns less than 20% of our issued and outstanding shares of common stock.
All references to consolidated financial statements in this section relate to the consolidated financial statements of ImmunityBio, Inc. and its subsidiaries that appear in Part II, Item 8. “Financial Statements and Supplementary Data” of our Annual Report for the fiscal year ended December 31, 2025 filed with the SEC on February 23, 2026.
Our related-party debt is summarized below (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Balance as of December 31, 2025 |
| | | | | | | Fair Value |
| Maturity
Year | | Interest
Rate | | Principal
Amount | | Amount | | Leveling |
| | | | | | | | | |
| Related-Party Convertible Note at Fair Value: | | | | | | | | | |
| $505 million December 2024 Promissory Note | 2027 | | Term
SOFR
+8.0% | | $ | 505,000 | | | $ | 477,093 | | | Level 3 |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Balance as of December 31, 2024 |
| | | | | | | Fair Value |
| Maturity
Year | | Interest
Rate | | Principal
Amount | | Amount | | Leveling |
| | | | | | | | | |
| Related-Party Convertible Note at Fair Value: | | | | | | | | | |
| $505 million December 2024 Promissory Note | 2027 | | Term
SOFR
+8.0% | | $ | 505,000 | | | $ | 461,877 | | | Level 3 |
ImmunityBio, Inc. | 57 | 2026 Proxy Statement
$505 million December 2024 Promissory Note (4)
On December 10, 2024 in connection with an equity offering, the company and Nant Capital entered into a second amended and restated promissory note. Pursuant to the terms of the second amended and restated promissory note, Tranche 1 of the December 2023 Promissory Note with a principal amount of $125.0 million and Tranche 2 of the December 2023 Promissory Note with a principal amount of $380.0 million were combined into one convertible promissory note with a principal amount of $505.0 million with a maturity date of December 31, 2027. The $505 million December 2024 Promissory Note bears an interest rate of Term SOFR plus 8.0% per annum, payable on a quarterly basis. Pursuant to the terms of the second amended and restated promissory note, the noteholder, in its sole discretion, may convert all of the outstanding principal amount into shares of common stock at a conversion price of $5.4270 per share. The company may prepay the outstanding promissory note, at any time, in whole or in part, without penalty. Upon receipt of a written notice of prepayment from the company, the noteholder has the option to convert the outstanding principal amount to be prepaid and the accrued and unpaid interest thereon into shares of the company’s common stock at the same conversion price of $5.4270 per share. In addition, the noteholder can request up to $50.0 million of the outstanding principal amount and accrued interest to be repaid upon consummation of a specified transaction.
On January 23, 2026, the company entered into an amendment to the $505 million December 2024 Promissory Note, to grant the holder the right to convert any portion of the outstanding principal amount into shares of the company’s common stock at any time prior to the maturity date.
In connection with the RIPA transaction, the outstanding related party convertible note was subordinated to the RIPA payment obligations.
Mandatory Prepayment Feature
The embedded derivative related to a contingently exercisable prepayment feature, which allows the noteholder to request up to a $50.0 million prepayment and accrued interest upon occurrence of a specified transaction (defined in the promissory note) was recorded as a derivative liability on the consolidated balance sheet and measured at fair value prior to the second amended and restated promissory note. The fair value of the derivative liability was determined at each period end using the with-and-without method, which assessed the likelihood and timing of a specified transaction that if triggered could have resulted in a repayment. Changes in the fair value of the derivative liability were reported in change in fair value of derivative liabilities, on the consolidated statement of operations.
Since the company elected the fair value option to account for the $505 million December 2024 Promissory Note, the embedded derivative is no longer required to be recorded separately but is considered when estimating the fair value of the $505 million December 2024 Promissory Note. The change in fair value of the contingently exercisable prepayment feature embedded derivative through December 10, 2024, the date of the $505 million December 2024 Promissory Note amendment, was $0.8 million.
$300 million December 2021 Promissory Note (1), (2), (3)
On December 17, 2021, the company executed a $300.0 million promissory note with Nant Capital, an affiliated entity under common control of our Founder, Executive Chairman and Global Chief Scientific and Medical Officer. The note bore an interest rate of Term SOFR plus 5.4% per annum, payable on a quarterly basis. The outstanding principal amount and any accrued and unpaid interest on advances were due on December 17, 2022. In the event of a default on the loan (as defined in both the original and amended and restated promissory notes), including if the company does not repay the loan at maturity, the company had the right, at its sole option, to convert the outstanding principal amount and accrued and unpaid interest due under this note into shares of the company’s common stock at a price of $5.67 per share.
On August 31, 2022, the company and Nant Capital entered into an amended and restated promissory note, pursuant to which the maturity date of the promissory note was extended to December 31, 2023 and the interest rate was amended to Term SOFR plus 8.0% per annum.
On September 11, 2023, the company and Nant Capital entered into a letter amendment pursuant to which the maturity date of the promissory note was further extended to December 31, 2024.
ImmunityBio, Inc. | 58 | 2026 Proxy Statement
On December 29, 2023 in connection with the RIPA, the company and Nant Capital entered into an amended and restated promissory note pursuant to which this existing promissory note was amended to be included in the Tranche 2 principal amount of the amended $505 million December 2023 promissory note, with a maturity date of December 31, 2025, and an interest rate of Term SOFR plus 7.5% per annum. Based on the terms of the amended and restated promissory note, the noteholder, at its sole discretion, could convert all of the Tranche 2 $380.0 million principal amount into shares of common stock at a conversion price of $8.2690 per share.
$125 million August 2022 Promissory Note (1), (2), (3)
On August 31, 2022, the company executed a $125.0 million promissory note with Nant Capital. The note bears an interest rate of Term SOFR plus 8.0% per annum, payable on a quarterly basis. The company may prepay the outstanding promissory note, at any time, in whole or in part, without penalty.
On September 11, 2023, the company and Nant Capital entered into a letter amendment pursuant to which the maturity date of the promissory note was extended to December 31, 2024.
On December 29, 2023 in connection with the RIPA, the company and Nant Capital entered into an amended and restated promissory note letter agreement pursuant to which the existing promissory note was amended to be included in the Tranche 1 principal amount of the amended $505 million December 2023 promissory note, with a maturity date of December 31, 2025 and an interest rate of Term SOFR plus 8.0% per annum.
$50 million December 2022 Promissory Note (2), (3)
On December 12, 2022, the company executed a $50.0 million promissory note with Nant Capital. The note bore an interest rate of Term SOFR plus 8.0% per annum, payable on a quarterly basis. The company could prepay the outstanding promissory note, at any time, in whole or in part, without penalty. In the event of a specified transaction (as defined in the note), the noteholder could request the outstanding principal and interest due on the loan to be repaid in full upon consummation of such specified transaction.
On September 11, 2023, the company and Nant Capital entered into a letter amendment pursuant to which the maturity date of the promissory note was further extended to December 31, 2024.
On December 29, 2023 in connection with the RIPA, the company and Nant Capital entered into an amended and restated promissory note pursuant to which the existing promissory note was amended to be included in the Tranche 2 principal amount of the amended $505 million December 2023 promissory note, with a maturity date of December 31, 2025, and an interest rate of Term SOFR plus 7.5% per annum. Pursuant to the terms of the amended and restated promissory note, the investor, in its sole discretion, could convert all of the outstanding Tranche 2 principal amount into shares of common stock at a conversion price of $8.2690 per share. The noteholder could request up to $50.0 million of the Tranche 2 principal amount and accrued interest to be repaid upon consummation of such specified transaction.
$30 million March 2023 Promissory Note (2), (4)
On March 31, 2023, the company executed a $30.0 million promissory note with Nant Capital. This note bore interest at Term SOFR plus 8.0% per annum, payable on a quarterly basis. The outstanding principal amount and any accrued and unpaid interest was originally due on December 31, 2023. The company may prepay the outstanding promissory note, at any time, in whole or in part, without penalty. Upon receipt of a written notice of prepayment from the company, the noteholder could choose to convert the outstanding principal amount to be prepaid and the accrued and unpaid interest thereon into shares of the company’s common stock at a price of $2.28 per share. Additionally, the noteholder could at its option convert the entire outstanding principal amount of the promissory note and accrued interest into shares of the company’s common stock at a conversion price of $2.28 per share, at the option of the noteholder.
The company received net proceeds of approximately $29.9 million from this financing, net of a $0.1 million origination fee paid to the noteholder.
On September 11, 2023, the company and Nant Capital entered into a letter agreement pursuant to which the maturity date of the $30.0 million promissory note described above was extended from December 31, 2023 to December 31, 2024.
ImmunityBio, Inc. | 59 | 2026 Proxy Statement
On December 29, 2023 in connection with the RIPA, the company and Nant Capital entered into a letter agreement pursuant to which the maturity date of this promissory note was extended to December 31, 2025.
On December 10, 2024 in connection with an equity offering, the company received a written notice from Nant Capital, the holder of the $30.0 million promissory note due December 31, 2025, of its election to convert the entire outstanding principal and accrued interest under the existing note into shares of the company’s common stock. As of such date, the total outstanding principal amount and accrued and unpaid interest due under the existing note were converted into shares of the company’s common stock.
$30 million June 2023 Promissory Note (2), (3)
On June 13, 2023, the company executed a $30.0 million promissory note with Nant Capital, pursuant to which, the company could request up to three (3) advances of $10.0 million each. The principal amount of each advance bore interest rate at Term SOFR plus 8.0% per annum, payable on a quarterly basis. The outstanding principal amount and any accrued and unpaid interest on advances was originally due on December 31, 2023. We could prepay the outstanding principal amount, together with any accrued interest, on any then-outstanding advances, at any time, in whole or in part, without premium or penalty, and without the prior consent of the noteholder upon five (5) days written notice to the noteholder.
We received net proceeds of approximately $29.9 million from this promissory note, net of a $0.1 million origination fee paid to Nant Capital.
On September 11, 2023, the company and Nant Capital entered into a letter amendment pursuant to which the maturity date of the promissory note was further extended to December 31, 2024.
On December 29, 2023 in connection with the RIPA, the company and Nant Capital entered into an amended and restated promissory note pursuant to which the existing promissory note was amended to be included in the Tranche 2 principal amount of the amended $505 million December 2023 promissory note, with a maturity date of December 31, 2025, and an interest rate of Term SOFR plus 7.5% per annum. Pursuant to the terms of the amended and restated promissory note the investor, in its sole discretion, could convert all of the Tranche 2 $380.0 million principal amount into shares of common stock at a conversion price of $8.2690 per share.
$200 million September 2023 Promissory Note (2), (4)
On September 11, 2023, the company executed a $200.0 million convertible promissory note with Nant Capital. The note bore interest at Term SOFR plus 8.0% per annum, payable on a monthly basis. The outstanding principal amount and any accrued and unpaid interest were due on September 11, 2026. We could prepay the outstanding principal amount, together with any accrued interest, at any time, in whole or in part, without premium or penalty upon five (5) days’ written notice to the noteholder. The noteholder had the sole option to convert all (but not less than all) of the outstanding principal amount and accrued but unpaid interest into shares of the company’s common stock at a conversion price of $1.9350 per share. The company received net proceeds of approximately $199.0 million from this financing, net of a $1.0 million origination fee paid to the noteholder.
On December 10, 2024 in connection with an equity offering, the company received a written notice from Nant Capital, the holder of the $200.0 million promissory note due September 11, 2026 of its election to convert the entire outstanding principal and accrued interest due under the existing note into shares of the company’s common stock. As of such date, the total outstanding principal amount and accrued and unpaid interest due under the note were converted into shares of the company’s common stock.
Footnotes to Related-Party Debt
Debt Modification and Debt Extinguishments
(1)August 2022 Debt Extinguishment
On August 31, 2022, the company amended and restated the above fixed-rate notes payables held by Nant Capital, NantWorks, NantMobile and NCSC, which are entities affiliated with Dr. Soon-Shiong. Prior to the amendments and restatements, these notes bore and thereafter continued to bear interest at a per annum rate ranging from 3.0% to
ImmunityBio, Inc. | 60 | 2026 Proxy Statement
6.0%, provided that the outstanding principal was and thereafter continued to be due and payable on September 30, 2025, and accrued and unpaid interest was or continued to be payable either upon maturity or, with respect to one of the notes, on a quarterly basis. Prior to the amendments and restatements, the company could and thereafter continued to be able to prepay the outstanding principal (together with accrued and unpaid interest), either in whole or in part, at any time without premium or penalty and without the prior consent of the noteholder, subject to an advance notice period of at least five business days during which the noteholder could convert the amount requested to be prepaid by the company into shares of the company’s common stock, as part of the amendment and restatement.
The terms of these fixed-rate promissory notes were amended and restated to include a conversion feature that gave each noteholder, at its sole option, at any time (other than when the noteholder is in receipt of a written notice of prepayment from the borrower), the right to convert the entire outstanding principal amount and accrued and unpaid interest due under each note at the time of conversion into shares of the company’s common stock at a price of $5.67 per share.
Since all of the above promissory notes were entered into or amended at the same time and with entities under common control, the company determined that the promissory notes were required to be evaluated collectively to accurately capture the economics of the transactions entered in contemplation of each other and contemporaneously. FASB ASC Topic 470-50 (ASC 470-50), Debt—Modifications and Extinguishments, provides that a modification or an exchange that adds or eliminates a substantive conversion option as of the conversion date would always be considered substantial and require extinguishment accounting. Accordingly, as a result of the addition of the conversion feature to the fixed-rate promissory notes, the fixed-rate promissory notes and the variable-rate promissory notes were determined to be extinguished given the contemporaneous nature of the amendments. The company performed a valuation of the fixed-rate promissory notes and variable-rate promissory notes before and after amendments. Under this model, the company calculated a gain on extinguishment of $82.9 million, representing the difference between the fair value of the new and amended promissory notes and the carrying value of the extinguished debt, net of any unamortized related-party notes discounts plus the cash proceeds from the new promissory note. Since the debt was obtained from entities under common control, such gain was recorded in additional paid-in capital, on the consolidated statement of stockholders’ deficit during the year ended December 31, 2022. Also, the difference between the face values of the new and amended promissory notes (and accrued interest on the date of the amendment) and the fair values of the new and restated promissory notes was recorded as a debt discount to be amortized to interest expense over the remaining term (or until conversion in the case of fixed-rate promissory notes) of the respective promissory notes. During the years ended December 31, 2023 and 2022, we recorded amortization of related-party notes discounts totaling $42.4 million and $16.3 million, respectively, in interest expense, on the consolidated statement of operations.
The fair values of the promissory notes without a holder conversion option were estimated using discounted cash flow analyses, based on market rates available to the company for similar debt at issuance after consideration of default and credit risk and the level of subordination. The fair values of the fixed-rate promissory notes, which were each modified to include a holder conversion option, were determined based on a binomial lattice convertible note model. The analysis involved the construction of various intermediate lattices: stock price tree, conversion value tree, conversion probability tree, and discount rate tree. Since certain of the factors analyzed are considered to be unobservable inputs, both the discounted cash flow model and the lattice model are considered to be Level 3 valuations. Significant unobservable inputs used for the discounted cash flow analysis included market yields from 18.0% to 24.8%, and the significant unobservable inputs used for the binomial lattice model included a volatility of 84.9%, and a market yield of 17.4%.
On December 12, 2022, the company received written notice from NantWorks, the holder of an existing convertible promissory note of NantCell, a wholly-owned subsidiary of the company, of its election to convert the NantCell promissory note into shares of the company’s common stock. As of such date, the holder of the NantCell note converted the entire $56.6 million of outstanding principal and accrued and unpaid interest due under the note into 9,986,920 shares of the company’s common stock at a price of $5.67 per share in accordance with the terms of the promissory note.
(2)September 11, 2023 Debt Modification
On September 11, 2023, the company entered into a stock purchase agreement with Nant Capital, NantMobile and NCSC pursuant to which the holders exchanged promissory notes totaling approximately $270.0 million in aggregate principal amount and accrued unpaid interest for an aggregate of 209,291,936 shares of common stock at an exchange price of $1.29 per share. As a result of the exchange, the company was forever released and discharged from all its obligations and liabilities under the notes.
ImmunityBio, Inc. | 61 | 2026 Proxy Statement
On September 11, 2023, the company and Nant Capital entered into a series of letter agreements pursuant to which the maturity date of the related-party nonconvertible notes described above totaling $505.0 million in principal and the $30.0 million March 2023 Promissory Note was extended from December 31, 2023 to December 31, 2024. In addition, the company entered into the $200.0 million September 2023 promissory note with Nant Capital. No other material terms or conditions of these promissory notes were modified.
Since all of the above promissory notes were entered into or amended at the same time on September 11, 2023 and with entities under common control, the company determined that the promissory notes were required to be evaluated collectively to accurately capture the economics of the transactions entered in contemplation of each other and contemporaneously. Pursuant to ASC 470-50, as the terms of the amendment were not substantially different than the terms of the promissory notes prior to the amendment, the amendment was accounted for as a debt modification. The unamortized debt discounts from the promissory notes are being amortized as an adjustment to interest expense over the remaining term of modified promissory notes that are not recorded at fair value using the effective interest rate method. Also, a $29.6 million increase in fair value of the embedded conversion feature from the debt modification was accounted for as a debt discount to the $200.0 million convertible note that is not recorded at fair value, and a $1.6 million increase in fair value of the embedded conversion feature related to the promissory note recorded at fair value was accounted for as interest expense during the year ended December 31, 2023. Such increase in fair values of the embedded conversion features totaling $31.2 million has been recorded with a corresponding increase in additional paid-in capital, on the consolidated statement of stockholders’ deficit.
(3)December 29, 2023 Debt Extinguishment
On December 29, 2023 in connection with the RIPA, the company and Nant Capital entered into an amended and restated promissory note and a letter amendment for the following outstanding promissory notes. Pursuant to the terms of the amended and restated promissory note, the amended $505 million December 2023 promissory note became comprised of a Tranche 1 with principal amount of $125.0 million which was previously the $125 million August 2022 promissory note before amendment, and a Tranche 2 with principal amount of $380.0 million, which was made up of the previous $300 million December 2021 promissory note, $50 million December 2022 promissory note and $30 million June 2023 promissory note. In addition, the amendment allowed Nant Capital, in its sole discretion, to convert all the Tranche 2 principal amount of $380.0 million of the amended promissory note into shares of common stock. The conversion price was subsequently determined at $8.2690 per share based on the agreement. The maturity date of the amended promissory note was December 31, 2025. Pursuant to the terms of the letter amendment, the maturity date of the $30 million March 2023 promissory note was extended from December 31, 2024 to December 31, 2025. Also, in connection with the RIPA transaction, all outstanding related-party promissory notes became subordinated to the RIPA payment obligations.
ImmunityBio, Inc. | 62 | 2026 Proxy Statement
The following table summarizes the Nant Capital promissory notes before the amendments on December 29, 2023 (principal amount in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Principal
Amount | | Maturity
Year | | Conversion
Price | | Interest
Rate |
| | | | | | | |
| Related-Party Nonconvertible Note: | | | | | | | |
| $125 million August 2022 Promissory Note | $ | 125,000 | | | 2024 | | | | Term
SOFR
+8.0% |
| | | | | | | |
| Related-Party Convertible Notes: | | | | | | | |
| $300 million December 2021 Promissory Note | $ | 300,000 | | | 2024 | | | | Term
SOFR
+8.0% |
| $50 million December 2022 Promissory Note | 50,000 | | | 2024 | | | | Term
SOFR
+8.0% |
| $30 million June 2023 Promissory Note | 30,000 | | | 2024 | | | | Term
SOFR
+8.0% |
| $30 million March 2023 Promissory Note | 30,000 | | | 2024 | | $2.2800 | | Term
SOFR
+8.0% |
| $200 million September 2023 Promissory Note | 200,000 | | | 2026 | | $1.9350 | | |
Total related-party promissory notes
before amendments | $ | 735,000 | | | | | | | |
The following table summarizes the Nant Capital promissory notes after the amendments on December 29, 2023 (principal amount in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Principal
Amount | | Maturity
Year | | Conversion
Price | | Interest
Rate |
| | | | | | | |
| Related-Party Nonconvertible Note: | | | | | | | |
$505 million December 2023 Promissory Note –
Tranche 1 | $ | 125,000 | | | 2025 | | | | Term
SOFR
+8.0% |
| | | | | | | |
| Related-Party Convertible Notes: | | | | | | | |
| $300 million December 2021 Promissory Note | $ | 300,000 | | | | | | | |
$50 million December 2022
Promissory Note | 50,000 | | | | | | | |
| $30 million June 2023 Promissory Note | 30,000 | | | | | | | |
$505 million December 2023 Promissory Note –
Tranche 2 | 380,000 | | | 2025 | | $8.2690 | | Term
SOFR
+7.5% |
| Total $505 million December 2023 Promissory Note | 505,000 | | | | | | | |
| $30 million March 2023 Promissory Note | 30,000 | | | 2025 | | $2.2800 | | Term
SOFR
+8.0% |
| $200 million September 2023 Promissory Note | 200,000 | | | 2026 | | $1.9350 | | Term
SOFR
+8.0% |
Total related-party promissory notes
after amendments | $ | 735,000 | | | | | | | |
ImmunityBio, Inc. | 63 | 2026 Proxy Statement
Since all of the above outstanding promissory notes were amended at the same time, with entities under common control, the company determined that the promissory notes were required to be evaluated collectively to accurately capture the economics of the transactions entered in contemplation of each other and contemporaneously. Also, in accordance with ASC 470-50 the company used the debt terms that existed before the September 11, 2023 modification to determine whether the modification was substantially different, as the September 11, 2023 modification was within a year of the transaction, and the promissory notes, at that time, had been modified without being deemed to be substantially different. As the modifications (September 11, 2023 and December 29, 2023 on a cumulative basis) added a substantive conversion feature to the promissory notes, these promissory notes were determined to be extinguished given the contemporaneous nature of the amendments. The company performed a valuation of all the promissory notes before and after amendments. Under this model, the company calculated a loss on extinguishment of $318.8 million, representing the difference between the fair value of the amended promissory notes and the carrying value of the extinguished debt, net of any unamortized related-party notes discounts. Since the debt was obtained from entities under common control, such loss was recorded in additional paid-in capital. In addition, a debt premium totaling $354.9 million, calculated as the difference between the fair values of certain promissory notes after modifications and their respective face values, was also recorded in additional paid-in capital. Collectively, a net gain on debt extinguishment of $36.1 million was recorded in additional paid-in capital, on the consolidated statement of stockholders’ deficit for the year ended December 31, 2023. Also, the difference between face values of certain new and amended promissory notes and their respective fair values of $53.1 million was recorded as a debt discount to be amortized as interest expense over the remaining term. During the year ended December 31, 2023, we recorded amortization of related-party notes discounts totaling $0.5 million in interest expense, on the consolidated statement of operations related to the new and amended promissory notes.
In regard to the Tranche 2 principal amount of the $505 million December 2023 promissory note, the company identified an embedded derivative related to a contingent exercisable prepayment feature of the promissory note, which allowed the noteholder to request up to a $50.0 million prepayment of the promissory note and accrued interest upon the occurrence of a specified transaction. After the debt extinguishment, the company concluded that this promissory note was issued at a substantial discount, so the embedded derivative that was contingently exercisable was required to be bifurcated and accounted separately from the debt host instrument. The fair value of the embedded derivative was estimated at $0.8 million as of December 31, 2023, using a “with-and-without” method, which assesses the likelihood and timing of the specified transaction to be triggered and result in a repayment. The estimated fair value was computed with the following unobservable inputs:
| | | | | |
| As of
Amendment Date
December 29, 2023 |
| |
| Expected market yield | 23.2 | % |
| Expected volatility | 118.0 | % |
The fair value of Tranche 1 principal amount of the amended $505 million December 2023 promissory note, which had no noteholder conversion option, was estimated using discounted cash flow analyses, based on market rates available to the company for similar debt at issuance after consideration of default and credit risk and the level of subordination. The fair value of Tranche 2 of the amended promissory note, which was modified to include a noteholder conversion option, was determined based on a binomial lattice convertible note model. The analysis involved the construction of various intermediate lattices: stock price tree, conversion value tree, conversion probability tree, and discount rate tree. Since certain of the factors analyzed were considered to be unobservable inputs, both the discounted cash flow model and the lattice model are considered to be Level 3 valuations. The effective unamortized debt discount rate of the amended Tranche 1 and Tranche 2 principal amount of the $505 million December 2023 promissory note was 23.65% and 18.04%, respectively. The estimated fair value was computed with the following unobservable inputs:
| | | | | |
| As of
Amendment Date
December 29, 2023 |
| |
| Expected market yield | 23.2 | % |
| Expected volatility | 118.0 | % |
ImmunityBio, Inc. | 64 | 2026 Proxy Statement
The fair value of the $200 million September 2023 promissory note was determined using the binomial lattice model with the following unobservable inputs:
| | | | | |
| As of
Amendment Date
December 29, 2023 |
| |
| Expected market yield | 23.3 | % |
| Expected volatility | 119.3 | % |
Prior to December 29, 2023, the $30 million March 2023 promissory note was accounted for under the FASB ASC Topic 825 (ASC 825), Financial Instruments, fair value election (FVO). Under the FVO election, the note was initially measured at its issue-date estimated fair value and subsequently remeasured at estimated fair value on a recurring basis at each reporting period date. The estimated fair value of the convertible note was computed using the binomial lattice model with the following unobservable inputs before it was modified on December 29, 2023. After the debt extinguishment, the note was accounted for under the amortized cost basis.
| | | | | |
| As of
Amendment Date
December 29, 2023 |
| |
| Expected market yield | 23.5 | % |
| Expected volatility | 138.0 | % |
The change in the carrying value of this note was as follows (in thousands):
| | | | | |
| Fair value at issuance date, March 31, 2023 | $ | 29,850 | |
| Change in fair value | 36,203 | |
| Gain on debt extinguishment with entities under common control | (36,053) | |
| Carrying value, December 29, 2023 | $ | 30,000 | |
(4)December 10, 2024 Debt Extinguishment
On December 10, 2024 in connection with an equity offering, the company and Nant Capital entered into a second amended and restated promissory note. Pursuant to the terms of the second amended and restated promissory note, Tranche 1 of the December 2023 Promissory Note with a principal amount of $125.0 million and Tranche 2 of the December 2023 Promissory Note with a principal amount of $380.0 million were combined into one convertible promissory note with a principal amount of $505.0 million with a maturity date of December 31, 2027. The $505 million December 2024 Promissory Note bears an interest rate of Term SOFR plus 8.0% per annum, payable on a quarterly basis. Pursuant to the terms of the second amended and restated promissory note, the noteholder, in its sole discretion, may convert all of the outstanding principal amount into shares of common stock at a conversion price of $5.4270 per share. In addition, the noteholder can request up to $50.0 million of the outstanding principal amount and accrued interest to be repaid upon consummation of a specified transaction. Also, on the same date, the company received a written notice from Nant Capital, the holder of the $30 million March 2023 Promissory Note due on December 31, 2025 and the $200 million September 2023 Promissory Note due on September 11, 2026 of its election to convert the entire outstanding principal and accrued interests of the such promissory notes into shares of the company’s common stock (the Converted Promissory Notes). As of such date, the total outstanding principal amount and accrued and unpaid interests due under the Converted Promissory Notes were converted into shares of the company’s common stock.
Since the $505 million December 2024 Promissory Note, together with the Converted Promissory Notes, were executed contemporaneously and in contemplation with one another with entities under common control, the company evaluated these transactions collectively as a single unit of account to accurately capture the economics of the transactions. The company determined the fair value of the conversion feature before and after the above transactions to assess whether there is a substantial change in fair value. The company used binomial lattice models for the “with” scenario and discounted cash flow model for the “without” scenario. The company determined that the fair value of the conversion
ImmunityBio, Inc. | 65 | 2026 Proxy Statement
feature before modification is substantially different than the fair value after modification, and as such, these transactions were accounted for as debt extinguishment in accordance with ASC 470-50. Under this model, the company calculated a gain on debt extinguishment of $10.4 million, which represents the difference between the: (a) fair value of the December 2024 Promissory Note and the reacquisition price of the Converted Promissory Notes on the date of conversion; and (b) carrying value of the extinguished debt, net of any unamortized related party notes discounts plus, the debt premium previously recognized in additional paid-in capital, on the consolidated statement of stockholders’ deficit as a result of the December 29, 2023 Debt Extinguishment. Since the debt was obtained from entities under common control, such gain on extinguishment was recorded in additional paid-in capital, on the consolidated statement of stockholders’ deficit.
Due to the conversion of the Converted Promissory Notes, the company also recorded $188.5 million in additional paid-in capital, on the consolidated statement of stockholders’ deficit as of December 31, 2024. This represents the difference between the reacquisition price of the Converted Promissory Notes on the date of conversion and the debt premium previously recognized in additional paid-in capital, on the consolidated statement of stockholders’ deficit as a result of the December 29, 2023 Debt Extinguishment.
The estimated fair value of the Converted Promissory Notes were computed with the following unobservable inputs at the amendment date on December 10, 2024:
| | | | | | | | | | | | | | |
| | $30 million March 2023 Promissory Note | | $200 million
September 2023
Promissory Note |
| | | | |
| Binomial lattice model: | | | | |
| Expected market yield | | 18.5 | % | | 18.6 | % |
| Expected volatility | | 103.2 | % | | 118.6 | % |
| | | | |
| Discounted cash flow model: | | | | |
| Discount rate | | 18.5 | % | | 18.6 | % |
The company elected the fair value option for recognizing the $505 million December 2024 Promissory Note under ASC 825. Under the FVO election, the note was initially measured at estimated fair value upon issuance and is remeasured at estimated fair value on a recurring basis at each reporting period date. The estimated fair value of the convertible note was computed using the following unobservable inputs at the following dates:
| | | | | | | | | | | | | | | | | | | | | | |
| | | | Immediately After
the Amendment on
December 10, 2024 | | As of
December 31, 2024 | | As of
December 31, 2025 |
| | | | | | | | |
| Binomial lattice model: | | | | | | | | |
| Expected market yield | | | | 18.7 | % | | 19.3 | % | | 14.8 | % |
| Expected volatility | | | | 115.1 | % | | 125.6 | % | | 87.5 | % |
The change in the fair value of this note was as follows (in thousands):
| | | | | | | | |
| Fair value at issuance date, December 10, 2024 | | $ | 518,378 | |
| Interest payment | | (11,808) | |
| Change in fair value, including $1.2 million related to instrument-specific credit risk | | (44,693) | |
| Fair value, at December 31, 2024 | | 461,877 | |
| Change in fair value of related-party convertible note | | 42,773 | |
| Change in fair value of related-party convertible note related to instrument-specific credit risk | | (27,557) | |
| Fair value, at December 31, 2025 | | $ | 477,093 | |
ImmunityBio, Inc. | 66 | 2026 Proxy Statement
During the years ended December 31, 2025 and 2024, the company recorded gains of $27.6 million and $1.2 million, respectively, in change in fair value of related-party convertible note related to instrument-specific credit risk, on the consolidated statements of comprehensive loss.
The following tables summarize the interest expense for our related-party promissory notes during the years ended December 31, 2025, 2024 and 2023 (in thousands):
| | | | | | | | | | | | | | |
| | Year Ended
December 31, 2025 |
| | Interest
Expense | | Debt
Discount
Amortization |
| | | | |
| $505 million December 2024 Promissory Note | | $ | 60,886 | | | $ | — | |
| Total | | $ | 60,886 | | | $ | — | |
| | | | | | | | | | | | | | |
| | Year Ended
December 31, 2024 |
| | Interest
Expense | | Debt
Discount
Amortization |
| | | | |
| $505 million December 2024 Promissory Note | | $ | 3,585 | | | $ | — | |
| $505 million December 2023 Promissory Note Tranche 1 | | 15,281 | | | 8,454 | |
| $505 million December 2023 Promissory Note Tranche 2 | | 44,658 | | | 14,133 | |
| $30 million March 2023 Promissory Note | | 3,667 | | | — | |
| $200 million September 2023 Promissory Note | | 24,810 | | | — | |
| Total | | $ | 92,001 | | | $ | 22,587 | |
| | | | | | | | | | | | | | |
| | Year Ended
December 31, 2023 |
| | Interest
Expense | | Debt
Discount
Amortization |
| | | | |
| $300 million December 2021 Promissory Note (1) | | $ | 39,653 | | | $ | 27,967 | |
| $125 million August 2022 Promissory Note (1) | | 16,521 | | | 5,962 | |
| $50 million December 2022 Promissory Note (1) | | 6,609 | | | 478 | |
| $30 million March 2023 Promissory Note (1) | | 4,590 | | | — | |
| $30 million June 2023 Promissory Note | | 2,096 | | | 258 | |
| $200 million September 2023 Promissory Note | | 8,185 | | | 2,586 | |
| Related-Party Fixed-Rate Promissory Notes | | 8,799 | | | 5,145 | |
| Total | | $ | 86,453 | | | $ | 42,396 | |
_______________
| | | | | |
| (1) | Balances include the amortization of debt discount totaling $0.5 million recorded during the period from December 29, 2023 to December 31, 2023. The interest expense recorded during this period was $0.4 million. |
ImmunityBio, Inc. | 67 | 2026 Proxy Statement
The following table summarizes the estimated future contractual obligations of our related-party debt as of December 31, 2025 (in thousands):
| | | | | | | | | | | | | | | | | |
| Principal
Payments | Interest
Payments (1) | | Total |
| | | | | |
| 2026 | $ | — | | | $ | 58,887 | | | $ | 58,887 | |
| 2027 | 505,000 | | | 58,887 | | | 563,887 | |
| Total | $ | 505,000 | | | $ | 117,774 | | | $ | 622,774 | |
_______________
| | | | | | | | | | | | | | |
| (1) | Interest payments on our promissory note are calculated based on Term SOFR plus the contractual spread per the loan agreement. The interest rate on our promissory note as of December 31, 2025 was 11.66%. |
Interest expense consists of the following (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2025 | | 2024 | | 2023 |
| | | | | |
| Interest expense on related-party notes payable | $ | 60,886 | | | $ | 92,001 | | | $ | 86,453 | |
| Amortization of related-party notes discounts | — | | | 22,587 | | | 42,396 | |
| Interest expense – related party | $ | 60,886 | | | $ | 114,588 | | | $ | 128,849 | |
In connection with the RIPA transaction, the outstanding related-party convertible note was subordinated to the RIPA payment obligations.
| | | | | | | | | | | | | | |
| Other Related-Party Agreements and Arrangements |
All references to consolidated financial statements in this section relate to the consolidated financial statements of ImmunityBio, Inc. and its subsidiaries that appear in Part II, Item 8. “Financial Statements and Supplementary Data” of our Annual Report for the fiscal year ended December 31, 2025 filed with the SEC on February 23, 2026.
Our Founder, Executive Chairman and Global Chief Scientific and Medical Officer also founded and has a controlling interest in NantWorks, which is a collection of companies in the healthcare and technology space. As described below, we have entered into arrangements with NantWorks, and certain affiliates of NantWorks, to facilitate the development of new immunotherapies for our product pipeline. Affiliates of NantWorks are also affiliates of the company due to the common control by and/or common ownership interest of our Founder, Executive Chairman, Global Chief Scientific and Medical Officer, and principal stockholder.
NantWorks, LLC
Shared Services Agreement
Under the amended and restated shared services agreement with NantWorks dated as of June 2016, but effective as of August 2015, NantWorks, a related party, provides corporate, general and administrative, certain research and development, and other support services to us, and we provide certain of such services to them. The receiving party is charged for the services at cost plus reasonable allocations of employee benefits, facilities, and other direct or fairly allocated indirect costs that relate to the employees providing the services. During the years ended December 31, 2025, 2024 and 2023, we recorded $2.1 million, $2.6 million and $3.3 million, respectively, in selling, general and administrative – related parties, and $0.8 million, $3.3 million, and $2.2 million, respectively, of expense reimbursements under this arrangement in research and development – related parties, on the consolidated statements of operations. These amounts exclude certain immaterial general and administrative expenses provided by third-party vendors directly for our benefit, which were reimbursed to NantWorks based on those vendors’ invoiced amounts without markup by NantWorks.
ImmunityBio, Inc. | 68 | 2026 Proxy Statement
As of December 31, 2025 and 2024, we had a payable of $0.2 million and a receivable of $0.2 million, respectively, for all agreements with NantWorks, which are included in due to/due from related parties, on the consolidated balance sheets. We also recorded $0.7 million and $3.0 million of prepaid expenses for various services that we expect will be passed through to the company from NantWorks as of December 31, 2025 and 2024, respectively, which are included in prepaid expenses – related parties, on the consolidated balance sheets.
Facility License Agreement
In 2015, we entered into a facility license agreement with NantWorks for office space in Culver City, California, which was converted to a research and development laboratory and a cGMP manufacturing facility. During the years ended December 31, 2025, 2024 and 2023, we recorded license fee expense for this facility totaling $3.6 million, $3.5 million, and $3.4 million, respectively, in research and development – related parties, on the consolidated statements of operations.
Related-Party Leases
The company leases certain assets under operating leases with certain entities controlled by NantWorks and its affiliates or subsidiaries. Right-of-use assets for related-party operating leases totaled $13.2 million and $16.0 million, and the associated operating lease liabilities totaled $17.0 million and $20.4 million, as of December 31, 2025 and 2024, respectively. The company recorded operating lease expense totaling $4.6 million, $4.6 million and $4.5 million for the years ended December 31, 2025, 2024 and 2023, respectively. Related-party leases were classified as either selling, general and administrative – related parties or research and development – related parties, on the consolidated statements of operations, depending on the use of the facility.
Immuno-Oncology Clinic, Inc.
We have entered into multiple agreements with the Clinic to conduct clinical trials related to certain of our product candidates. The Clinic is a related party as it is owned by an officer of the company and NantWorks manages the administrative operations of the Clinic.
During the years ended December 31, 2025, 2024 and 2023, we recorded $2.9 million, $2.6 million, and $2.2 million respectively, related to clinical trial and transition services provided by the Clinic in research and development – related parties, on the consolidated statements of operations. As of December 31, 2025 and 2024, we owed the Clinic a $0.1 million and an immaterial amount, respectively, which are included in due to related parties, on the consolidated balance sheets.
| | | | | | | | | | | | | | |
| Indemnification Agreements |
We have entered into indemnification agreements with each of our directors and executive officers. The indemnification agreements and our Certificate of Incorporation and Bylaws require us to indemnify our directors, executive officers and certain controlling persons to the fullest extent permitted by Delaware law.
ImmunityBio, Inc. | 69 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
Stockholder Proposals for the 2027 Annual Meeting |
Proposals Pursuant to Rule 14a-8. Stockholders may present proper proposals for inclusion in our proxy statement and for consideration at the 2027 annual meeting of stockholders by submitting their proposals in writing to our Corporate Secretary at our principal executive offices so that they are received no later than the close of business (5:30 p.m. Pacific Time) on December 30, 2026. In addition, stockholder proposals must comply with the requirements of Rule 14a-8 under the Exchange Act regarding the inclusion of stockholder proposals in company-sponsored proxy materials. Pursuant to the rules promulgated by the SEC, simply submitting a proposal does not guarantee that it will be included.
Director Nominations Pursuant to our Bylaws. It is the policy of the Governance Committee to consider recommendations for candidates to the Board from stockholders holding no less than 1% of the outstanding shares of our common stock continuously for at least 12 months prior to the date of the submission of the recommendation or nomination.
A stockholder that wishes to recommend a candidate for consideration by the Governance Committee as a potential candidate for director must direct the recommendation in writing to our Corporate Secretary at our principal executive offices, and must include the candidate’s name, home and business contact information, detailed biographical data, relevant qualifications, class and number of shares of the company’s common stock that are held by the nominee, a signed letter from the candidate confirming willingness to serve, information regarding any relationships between us and the candidate and evidence of the recommending stockholder’s ownership of our common stock. Such recommendation must also include a statement from the recommending stockholder in support of the candidate, particularly within the context of the criteria for board membership, including issues of character, integrity, judgment, and diversity of experience, independence, area of expertise, corporate experience, potential conflicts of interest, other commitments and the like and personal references. The Governance Committee will consider the recommendation but will not be obligated to take any further action with respect to the recommendation.
Advance Notice Procedures for 2027 Annual Meeting. In order to be brought before our 2027 annual meeting of stockholders, the stockholder must have given timely notice of such proposal or nomination, in proper written form. To be timely for our 2027 annual meeting of stockholders, a stockholder’s notice of a matter that the stockholder wishes to present, or the person or persons the stockholder wishes to nominate as a director, must be delivered to our Corporate Secretary at our principal executive offices not less than 45 days and not more than 75 days before the one-year anniversary of the date on which we first mailed our proxy materials or a notice of availability of proxy materials (whichever is earlier) for the preceding year’s annual meeting. As a result, any written notice given by a stockholder pursuant to these provisions of our Bylaws must be received by our Corporate Secretary at our principal executive offices:
•Not earlier than February 13, 2027 and
•Not later than March 15, 2027.
In the event that we hold our 2027 annual meeting of stockholders more than 30 days before or more than 60 days after the one-year anniversary date of the 2026 Annual Meeting, then such written notice must be received no earlier than the close of business on the 120th day prior to such annual meeting and no later than the close of business on the later of the following two dates:
•The 90th day prior to such annual meeting, or
•The 10th day following the day on which public announcement of the date of such meeting is first made.
To be in proper written form, a stockholder’s notice and/or proposals must include the specified information concerning the proposal or nominee as described in our Bylaws. We reserve the right to reject, rule out of order, or take other appropriate action with respect to any proposal that does not comply with these and other applicable requirements. Notices and/or proposals should be addressed to our Corporate Secretary at our principal executive offices.
ImmunityBio, Inc. | 70 | 2026 Proxy Statement
In addition, stockholders who intend to solicit proxies in support of director nominees other than our nominees must also provide notice that sets forth the information required by Rule 14a-19 of the Exchange Act no later than 60 calendar days prior to the anniversary date of the previous year’s annual meeting. For any such director nominee to be included on our proxy card for next year’s annual meeting, the Corporate Secretary must receive notice under SEC Rule 14a-19 no later than April 12, 2027. Please note that the notice requirement under SEC Rule 14a-19 is in addition to the applicable notice requirements under the advance notice provisions of our Bylaws described above.
| | |
Availability of Amended and Restated Bylaws |
A copy of our Bylaws may be obtained by accessing the company’s filings on the SEC’s website at sec.gov. You may also contact our Corporate Secretary at our principal executive offices for a copy of the relevant bylaw provisions regarding the requirements for making stockholder proposals and nominating director candidates.
| | |
Householding of Proxy Materials |
Beneficial owners of common stock who share a single address will receive only one copy of the Notice of Internet Availability of Proxy Materials or the proxy materials, as the case may be, unless their broker, bank, or nominee has received contrary instructions from any beneficial owner at that address. This practice, known as “householding,” is designed to reduce printing and mailing costs. If any beneficial stockholder(s) sharing a single address wish to discontinue householding and receive a separate copy of the Notice of Internet Availability of Proxy Materials or the proxy materials, we will have a separate copy promptly delivered to them upon their written or oral request. To make the request, they may contact Broadridge, either by calling 866-540-7095, or by writing to Broadridge, Householding Department, 51 Mercedes Way, Edgewood, NY 11717, and including their name, the name of their broker, or other nominee, and their account number(s). Beneficial owners may also contact Broadridge if they received multiple copies of the proxy materials and prefer to receive a single copy in the future.
| | |
No Incorporation by Reference |
The reports of the Compensation and Audit Committees shall not be deemed to be “soliciting material” or to be “filed” with the SEC or subject to Regulation 14A promulgated by the SEC or Section 18 of the Exchange Act, and shall not be deemed incorporated by reference into any prior or subsequent filing by the company under the Securities Act, except to the extent the company specifically requests that the information be treated as soliciting material or specifically incorporates it by reference.
We maintain a website at ir.immunitybio.com. Information contained on, or that can be accessed through, our website is not intended to be incorporated by reference into this proxy statement, and references to our website address in this proxy statement are inactive textual references only.
This proxy statement contains statements regarding future individual and company performance targets and company performance goals. These targets and company performance goals are disclosed in the limited context of our compensation programs and should not be understood to be statements of management’s expectations or estimates of results or other guidance. We specifically caution investors not to apply these statements to other contexts.
ImmunityBio, Inc. | 71 | 2026 Proxy Statement
| | | | | | | | | | | | | | |
| Forward-Looking Statements |
This Proxy Statement contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. These forward-looking statements reflect the current views of our management with respect to, among other things, our operations, our business strategy and plans, our objectives and initiatives, our financial performance, our industry, and our business. Forward-looking statements include all statements that are not historical facts. In some cases, you can identify these forward-looking statements by the use of words such as “outlook,” “believe(s),” “expect(s),” “potential,” “continue(s),” “may,” “will,” “should,” “could,” “would,” “seek(s),” “predict(s),” “intend(s),” “trends,” “plan(s),” “estimate(s),” “anticipates,” “projection,” “will likely result” and or the negative version of these words or other comparable words of a future or forward-looking nature, although not all forward-looking statements contain these words. Such forward-looking statements are based upon various estimates and assumptions, as well as information known to us as of the date hereof and are subject to various risks and uncertainties. Accordingly, there are or will be important factors that could cause actual outcomes or results to differ materially from those indicated in these statements and you should not rely upon forward-looking statements as predictions of future events. These risks and uncertainties include, but are not limited to, those described under the caption “Risk Factors” in our Annual Report on Form 10-K for the fiscal year ended December 31, 2025, and our other SEC filings, which are available on our investor relations website at ir.immunitybio.com and on the SEC website at sec.gov. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, performance, or achievements. We undertake no obligation to publicly update or review any forward-looking statements, whether as a result of new information, future developments or otherwise, except as required by law.
The Board knows of no matters other than those listed in the attached Notice of Annual Meeting of Stockholders that are likely to be brought before the 2026 Annual Meeting. If any other matter properly comes before the 2026 Annual Meeting, the persons named on the enclosed proxy card will vote the proxy in accordance with their best judgment on such matter.
| | |
| By Order of the Board of Directors, |
|
|
|
| Philip LoScalzo |
| Corporate Secretary |
April 29, 2026
ImmunityBio, Inc. | 72 | 2026 Proxy Statement
ImmunityBio, Inc.
Reconciliation of GAAP to Non-GAAP Measure
The following table presents the reconciliation of Adjusted EBITDA to its nearest GAAP measure (in thousands; unaudited):
| | | | | | | | |
| | Year Ended
December 31,
2025 |
| |
| | |
Loss from operations (1) | | $ | (256,027) | |
| Add back: | | |
| Depreciation and amortization | | 15,525 | |
| Stock-based compensation expense | | 36,809 | |
| Loss due to write-off of fixed assets | | 14,018 | |
| Adjusted EBITDA | | $ | (189,675) | |
_______________
(1)Loss from operations excludes interest, income tax benefit, and non-cash other income (expense), net items, including change in fair value of warrant and derivative liabilities, and related-party convertible note.
ImmunityBio, Inc. | A-1 | 2026 Proxy Statement

Signature [PLEASE SIGN WITHIN BOX] Date Signature (Joint Owners) Date TO VOTE, MARK BLOCKS BELOW IN BLUE OR BLACK INK AS FOLLOWS: KEEP THIS PORTION FOR YOUR RECORDS DETACH AND RETURN THIS PORTION ONLYTHIS PROXY CARD IS VALID ONLY WHEN SIGNED AND DATED. V92080-P51159 1a.	 Patrick Soon-Shiong, M.D. 1b.	 Cheryl L. Cohen 1c.	 Richard Adcock 1d.	 Michael D. Blaszyk 1e.	 Wesley Clark 1f.	 Linda Maxwell, M.D. 1g.	 Christobel Selecky 1h.	 Barry J. Simon, M.D. 1i.	 Bruce Wendel For Against Abstain !! !! !! !! !! !! !! !! ! !! IMMUNITYBIO, INC. 3530 JOHN HOPKINS COURT SAN DIEGO, CA 92121 VOTE BY INTERNET Before The Meeting - Go to www.proxyvote.com or scan the QR Barcode above Use the Internet to transmit your voting instructions and for electronic delivery of information up until 11:59 p.m. Eastern Time on June 8, 2026. Have your proxy card in hand when you access the web site and follow the instructions to obtain your records and to create an electronic voting instruction form. During The Meeting - Go to www.virtualshareholdermeeting.com/IBRX2026 You may attend the meeting via the Internet and vote during the meeting. Have the information that is printed in the box marked by the arrow available and follow the instructions. VOTE BY PHONE - 1-800-690-6903 Use any touch-tone telephone to transmit your voting instructions up until 11:59 p.m. Eastern Time on June 8, 2026. Have your proxy card in hand when you call and then follow the instructions. VOTE BY MAIL Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717. 2.	 To ratify the selection of Deloitte & Touche LLP as ImmunityBio’s independent registered public accounting firm for the fiscal year ending December 31, 2026. IMMUNITYBIO, INC. The Board of Directors recommends you vote FOR the following: The Board of Directors recommends you vote FOR the following proposal: NOTE: In their discretion, the proxies are authorized to vote upon such other business as may properly come before the Annual Meeting. 	 Nominees: 1.	 Election of Directors For Withhold Please sign exactly as your name(s) appear(s) hereon. When signing as attorney, executor, administrator, or other fiduciary, please give full title as such. Joint owners should each sign personally. All holders must sign. If a corporation or partnership, please sign in full corporate or partnership name by authorized officer. !! SCAN TO VIEW MATERIALS & VOTEw

V92081-P51159 Important Notice Regarding the Availability of Proxy Materials for the Annual Meeting: The Notice and Proxy Statement and 2025 Annual Report are available at www.proxyvote.com. IMMUNITYBIO, INC. Annual Meeting Of Stockholders June 9, 2026 10:30 A.M., Pacific Time This Proxy Is Solicited On Behalf Of The Board Of Directors We have designated each of Richard Adcock, our Chief Executive Officer and President, Philip LoScalzo, our General Counsel and Corporate Secretary, and David Sachs, our Chief Financial Officer, to serve as proxies for the Annua l Meet ing, each wi th the power to appoint h i s subst i tute , and hereby author izes them to represent and to vote, as designated on the reverse side of this ballot, all of the shares of Common Stock of IMMUNITYBIO, INC. that the stockholders are entitled to vote at the Annual Meeting of Stockholders to be held at 10:30 A.M., Pacific Time, on Tuesday, June 9, 2026, virtually at www.virtualshareholdermeeting.com/IBRX2026, and any adjournment or postponement thereof. This proxy, when properly executed, will be voted in the manner directed herein. If no such direction is made, this proxy will be voted FOR the individuals named in Proposal 1 and FOR Proposal 2. Continued and to be signed on reverse side

Your Vote Counts! Smartphone users Point your camera here and vote without entering a control number For complete information and to vote, visit www.ProxyVote.com Control # V92092-P51159 *Please check the meeting materials for any special requirements for meeting attendance. IMMUNITYBIO, INC. 3530 JOHN HOPKINS COURT SAN DIEGO, CA 92121 IMMUNITYBIO, INC. 2026 Annual Meeting Vote by June 8, 2026 11:59 PM Eastern Time You invested in IMMUNITYBIO, INC. and it’s time to vote! You have the right to vote on proposals being presented at the Annual Meeting. This is an important notice regarding the availability of proxy materials for the stockholder meeting to be held on June 9, 2026. Vote Virtually at the Meeting* June 9, 2026 10:30 A.M., Pacific Time Virtually at: www.virtualshareholdermeeting.com/IBRX2026 Get informed before you vote View the Notice and Proxy Statement and 2025 Annual Report online OR you can receive a free paper or email copy of the material(s) by requesting prior to May 26, 2026. If you would like to request a copy of the material(s) for this and/or future stockholder meetings, you may (1) visit www.ProxyVote.com, (2) call 1-800-579-1639 or (3) send an email to sendmaterial@proxyvote.com. If sending an email, please include your control number (indicated below) in the subject line. Unless requested, you will not otherwise receive a paper or email copy.
Vote at www.ProxyVote.com Prefer to receive an email instead? While voting on www.ProxyVote.com, be sure to click “Delivery Settings”. Voting Items Board Recommends V92093-P51159 1.	 Election of Directors 	 Nominees: 1a.	 Patrick Soon-Shiong, M.D. For 1b.	 Cheryl L. Cohen For 1c.	 Richard Adcock For 1d.	 Michael D. Blaszyk For 1e.	 Wesley Clark For 1f.	 Linda Maxwell, M.D. For 1g.	 Christobel Selecky For 1h.	 Barry J. Simon, M.D. For 1i.	 Bruce Wendel For 2.	 To ratify the selection of Deloitte & Touche LLP as ImmunityBio’s independent registered public accounting firm for the fiscal year ending December 31, 2026. For NOTE: In their discretion, the proxies are authorized to vote upon such other business as may properly come before the Annual Meeting. THIS IS NOT A VOTABLE BALLOT This is an overview of the proposals being presented at the upcoming stockholder meeting. Please follow the instructions on the reverse side to vote these important matters.