Exhibit 99.1

Precision Designed Science for Cancer Patients NASDAQ: PDSB April 2026

Forward-Looking Statements This communication contains forward-looking
statements (including within the meaning of Section 21E of the United States Securities Exchange Act of 1934, as amended, and Section 27A of the United States Securities Act of 1933, as amended) concerning PDS Biotechnology Corporation (the
“Company”) and other matters. These statements may discuss goals, intentions and expectations as to future plans, trends, events, results of operations or financial condition, or otherwise, based on current beliefs of the Company’s
management, as well as assumptions made by, and information currently available to, management. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions,
and include words such as “may,” “will,” “should,” “would,” “expect,” “anticipate,” “plan,” “likely,” “believe,” “estimate,” “project,” “intend,” “forecast,” “guidance”, “outlook” and other similar expressions among others. Forward-looking
statements are based on current beliefs and assumptions that are subject to risks and uncertainties and are not guarantees of future performance. Actual results could differ materially from those contained in any forward-looking statement
as a result of various factors, including, without limitation: the Company’s ability to protect its intellectual property rights; the Company’s anticipated capital requirements, including the Company’s anticipated cash runway and the
Company’s current expectations regarding its plans for future equity financings; the Company’s dependence on additional financing to fund its operations and complete the development and commercialization of its product candidates, and the
risks that raising such additional capital may restrict the Company’s operations or require the Company to relinquish rights to the Company’s technologies or product candidates; the Company’s limited operating history in the Company’s
current line of business, which makes it difficult to evaluate the Company’s prospects, the Company’s business plan or the likelihood of the Company’s successful implementation of such business plan; the timing for the Company or its
partners to conduct clinical trials for PDS0101 (Versamune® HPV), PDS01ADC, PDS0103 (Versamune® MUC1) and other Versamune® based product candidates; the future success of such trials; the successful implementation of the Company’s research
and development programs and collaborations, including any collaboration studies concerning PDS0101, PDS01ADC, PDS0103 and other Versamune® based product candidates and the Company’s interpretation of the results and findings of such
programs and collaborations and whether such results are sufficient to support the future success of the Company’s product candidates; the success, timing and cost of the Company’s or its partners’ ongoing clinical trials and anticipated
clinical trials for the Company’s current product candidates, including statements regarding response rates, the timing of initiation, pace of enrollment and completion of the trials (including the Company’s ability to fully fund its
disclosed clinical trials, which assumes no material changes to the Company’s currently projected expenses), futility analyses, presentations at conferences and data reported in an abstract, and receipt of interim or preliminary results
(including, without limitation, any preclinical results or data), which are not necessarily indicative of the final results of the Company’s ongoing clinical trials; any Company statements about its understanding of product candidates
mechanisms of action and interpretation of preclinical and early clinical results from its clinical development programs and any collaboration studies; the Company’s ability to continue as a going concern; and other factors, including
legislative, regulatory, political and economic developments not within the Company’s control. The foregoing review of important factors that could cause actual events to differ from expectations should not be construed as exhaustive and
should be read in conjunction with statements that are included herein and elsewhere, including the other risks, uncertainties, and other factors described under “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition
and Results of Operations” and elsewhere in the documents we file with the U.S. Securities and Exchange Commission. The forward-looking statements are made only as of the date of this press release and, except as required by applicable law,
the Company undertakes no obligation to revise or update any forward-looking statement, or to make any other forward-looking statements, whether as a result of new information, future events or otherwise. Versamune® is a registered
trademark of PDS Biotechnology Corporation. KEYTRUDA® is a registered trademark of Merck Sharp and Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.

months 12-18 months for current standard of care1 Keytrud (pembrolizumab)
benchmark in First-line (1L) treatment for Recurrent/Metastatic Head and Neck Squamous Cell Carcinoma (1L R/M HNSCC) 77.4% disease control rate with PDS0101 in 1L R/M HNSCC Phase 3 VERSATILE-003 pivotal trial FDA alignment on
Progression-Free-Survival (PFS) as interim primary endpoint provides for accelerated pathway to approval Partnerships with leading cancer institutions MD Anderson Mayo Clinic National Cancer Institute Targeting HPV16+
Cancers REGISTRATIONAL TRIAL COMPELLING CLINICAL DATA POTENTIAL ACROSS CANCERS 4 clinical trials completed 39.3 median overall survival No head-to-head studies have been performed.
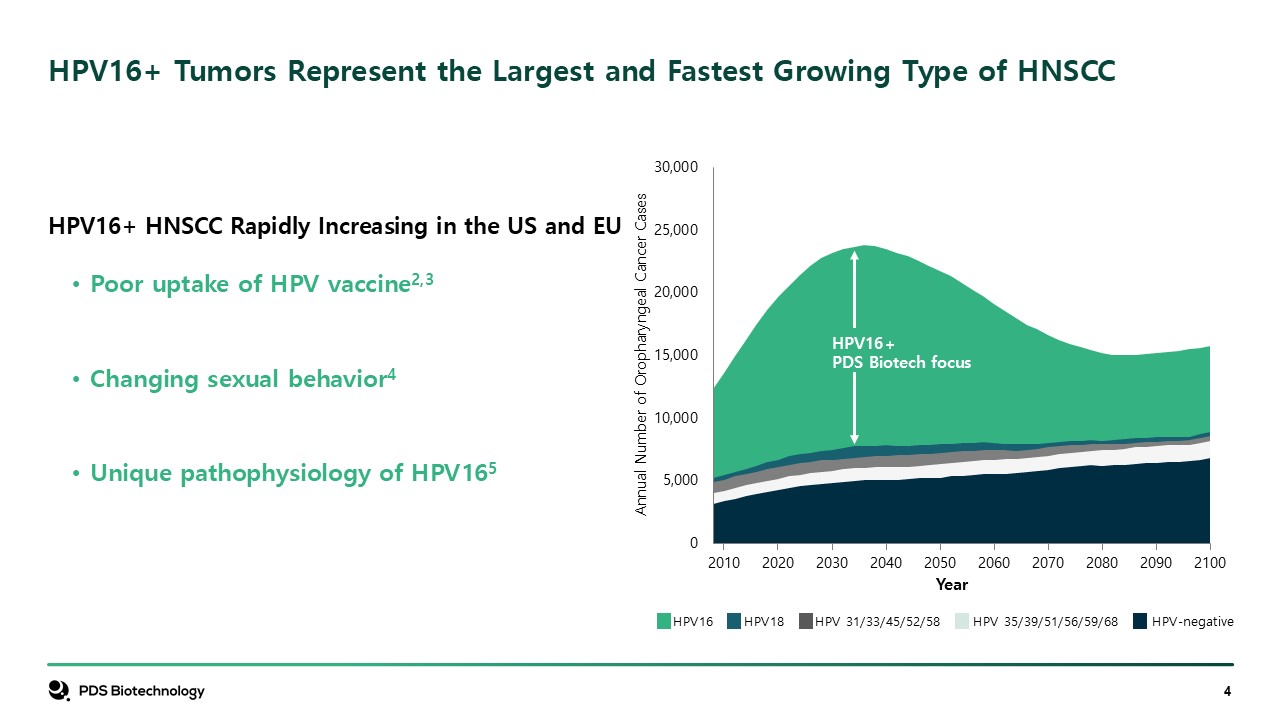
HPV16 HPV18 HPV 31/33/45/52/58 HPV 35/39/51/56/59/68 HPV-negative HPV16+
Tumors Represent the Largest and Fastest Growing Type of HNSCC HPV16+ HNSCC Rapidly Increasing in the US and EU Poor uptake of HPV vaccine2,3 Changing sexual behavior4 Unique pathophysiology of HPV165 Annual Number of Oropharyngeal
Cancer Cases HPV16+ PDS Biotech focus

PUBLISHED EVIDENCE Two head-to-head studies demonstrate that HPV16-positive
patients have statistically worse survival compared to other patient groups: Early-stage HNSCC: HPV16+ patients had worse survival than other HPV+ (P16+) patients6 Advanced oral cancer: HPV16+ patients had worse survival than HPV-negative
patients7 Keytruda® (pembrolizumab) + chemotherapy median survival1 12–18 months in recurrent/metastatic setting HPV16+ Patients Have No Targeted Therapies and Worse Clinical Outcomes CURRENT STANDARD OF CARE 2 1 A targeted approach
for HPV16+ patients represents a significant opportunity
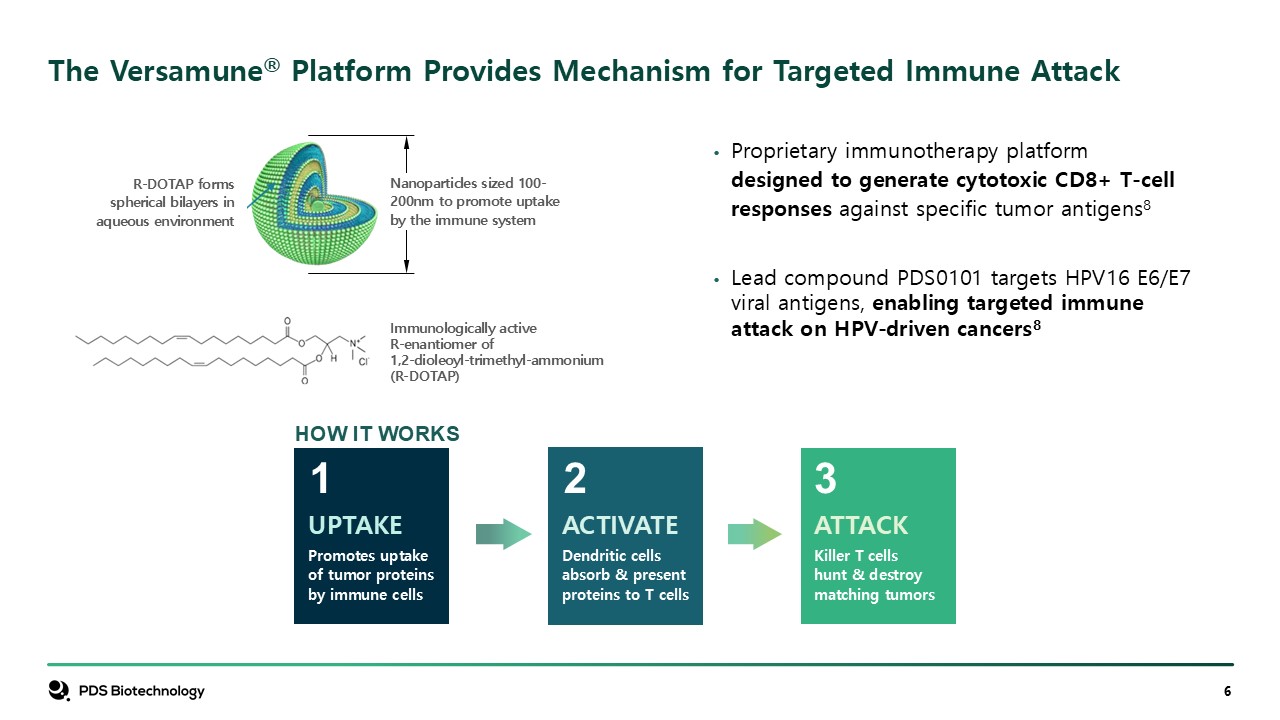
HOW IT WORKS 1 2 3 ATTACK Killer T cells hunt & destroy matching
tumors Immunologically active R-enantiomer of1,2-dioleoyl-trimethyl-ammonium (R-DOTAP) R-DOTAP forms spherical bilayers in aqueous environment The Versamune® Platform Provides Mechanism for Targeted Immune Attack Proprietary
immunotherapy platform designed to generate cytotoxic CD8+ T-cell responses against specific tumor antigens8 Lead compound PDS0101 targets HPV16 E6/E7 viral antigens, enabling targeted immune attack on HPV-driven cancers8 Nanoparticles
sized 100-200nm to promote uptake by the immune system ACTIVATE Dendritic cells absorb & present proteins to T cells UPTAKE Promotes uptake of tumor proteins by immune cells
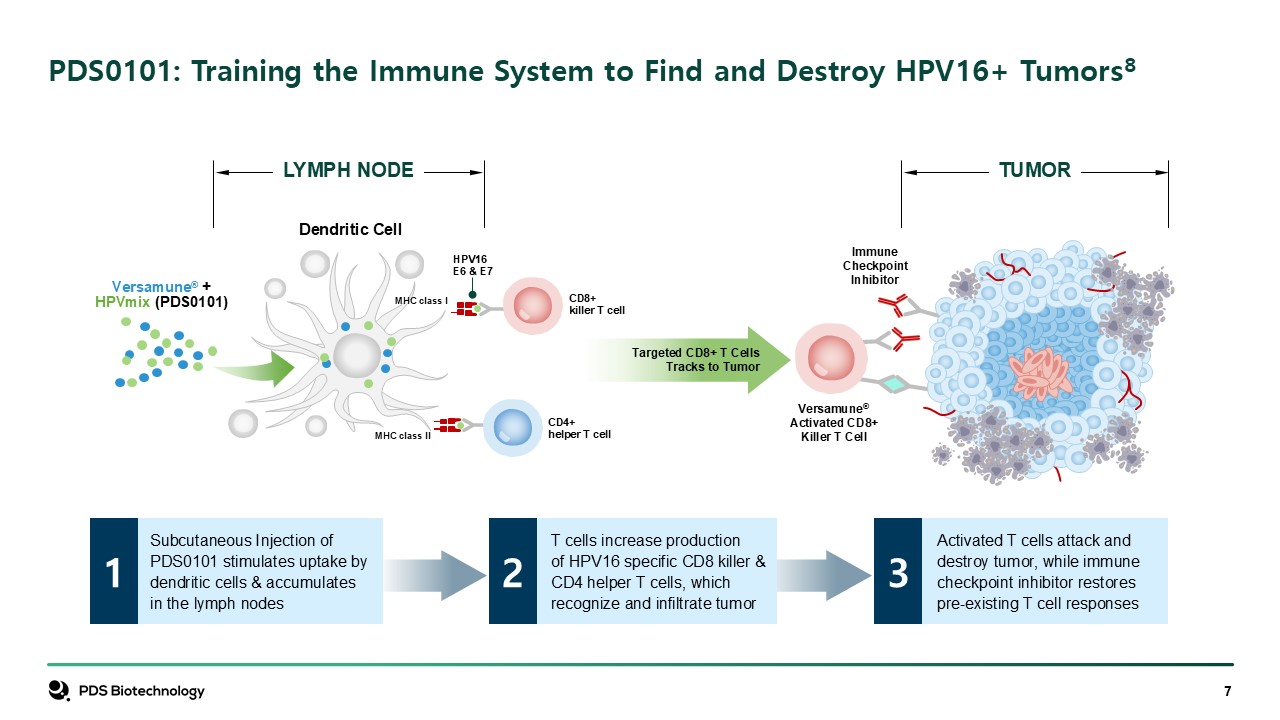
PDS0101: Training the Immune System to Find and Destroy HPV16+
Tumors8 CD4+helper T cell CD8+killer T cell HPV16 E6 & E7 Versamune®Activated CD8+Killer T Cell ImmuneCheckpointInhibitor Versamune® +HPVmix (PDS0101) Targeted CD8+ T CellsTracks to Tumor Dendritic Cell MHC class II MHC class
I LYMPH NODE TUMOR Subcutaneous Injection of PDS0101 stimulates uptake by dendritic cells & accumulates in the lymph nodes Activated T cells attack anddestroy tumor, while immune checkpoint inhibitor restores pre-existing T cell
responses 3 T cells increase production of HPV16 specific CD8 killer & CD4 helper T cells, which recognize and infiltrate tumor 2 1
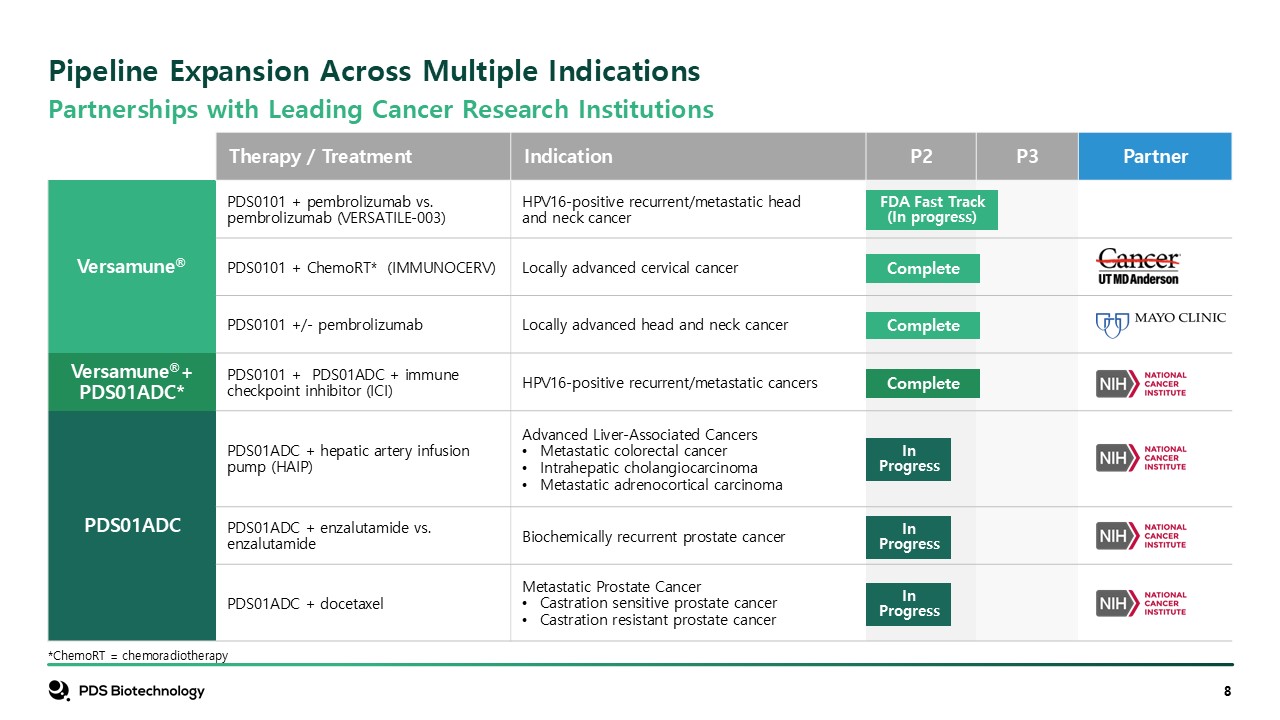
Therapy / Treatment Indication P2 P3 Partner Versamune® PDS0101 +
pembrolizumab vs. pembrolizumab (VERSATILE-003) HPV16-positive recurrent/metastatic head and neck cancer PDS0101 + ChemoRT* (IMMUNOCERV) Locally advanced cervical cancer PDS0101 +/- pembrolizumab Locally advanced head and neck
cancer Versamune®+PDS01ADC* PDS0101 + PDS01ADC + immune checkpoint inhibitor (ICI) HPV16-positive recurrent/metastatic cancers PDS01ADC PDS01ADC + hepatic artery infusion pump (HAIP) Advanced Liver-Associated Cancers Metastatic
colorectal cancer Intrahepatic cholangiocarcinoma Metastatic adrenocortical carcinoma PDS01ADC + enzalutamide vs. enzalutamide Biochemically recurrent prostate cancer PDS01ADC + docetaxel Metastatic Prostate Cancer Castration
sensitive prostate cancer Castration resistant prostate cancer FDA Fast Track (In progress) Complete Complete Complete In Progress In Progress In Progress Pipeline Expansion Across Multiple Indications *ChemoRT =
chemoradiotherapy Partnerships with Leading Cancer Research Institutions
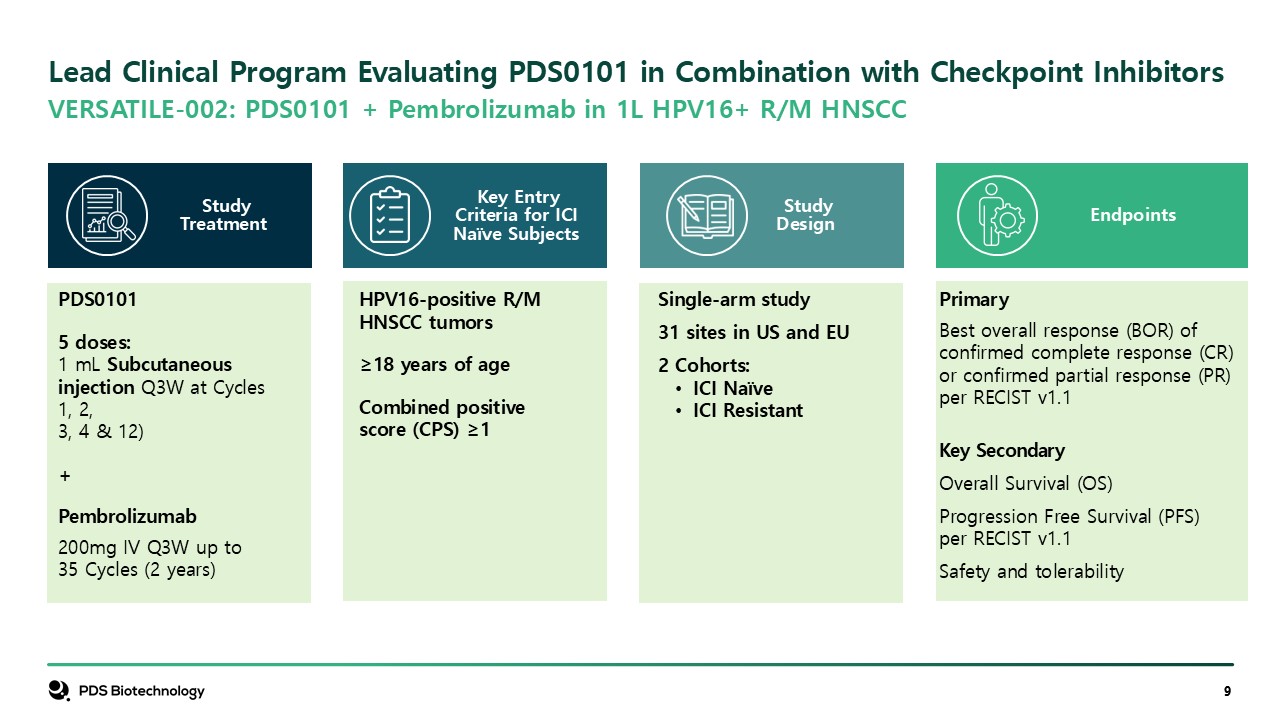
Endpoints Single-arm study 31 sites in US and EU 2 Cohorts: ICI
Naïve ICI Resistant HPV16-positive R/M HNSCC tumors ≥18 years of age Combined positive score (CPS) ≥1 PDS0101 5 doses: 1 mL Subcutaneous injection Q3W at Cycles 1, 2, 3, 4 & 12) + Pembrolizumab 200mg IV Q3W up to 35 Cycles (2
years) Primary Best overall response (BOR) of confirmed complete response (CR) or confirmed partial response (PR) per RECIST v1.1 Key Secondary Overall Survival (OS) Progression Free Survival (PFS) per RECIST v1.1 Safety and
tolerability StudyDesign Key Entry Criteria for ICI Naïve Subjects Study Treatment Lead Clinical Program Evaluating PDS0101 in Combination with Checkpoint Inhibitors VERSATILE-002: PDS0101 + Pembrolizumab in 1L HPV16+ R/M HNSCC
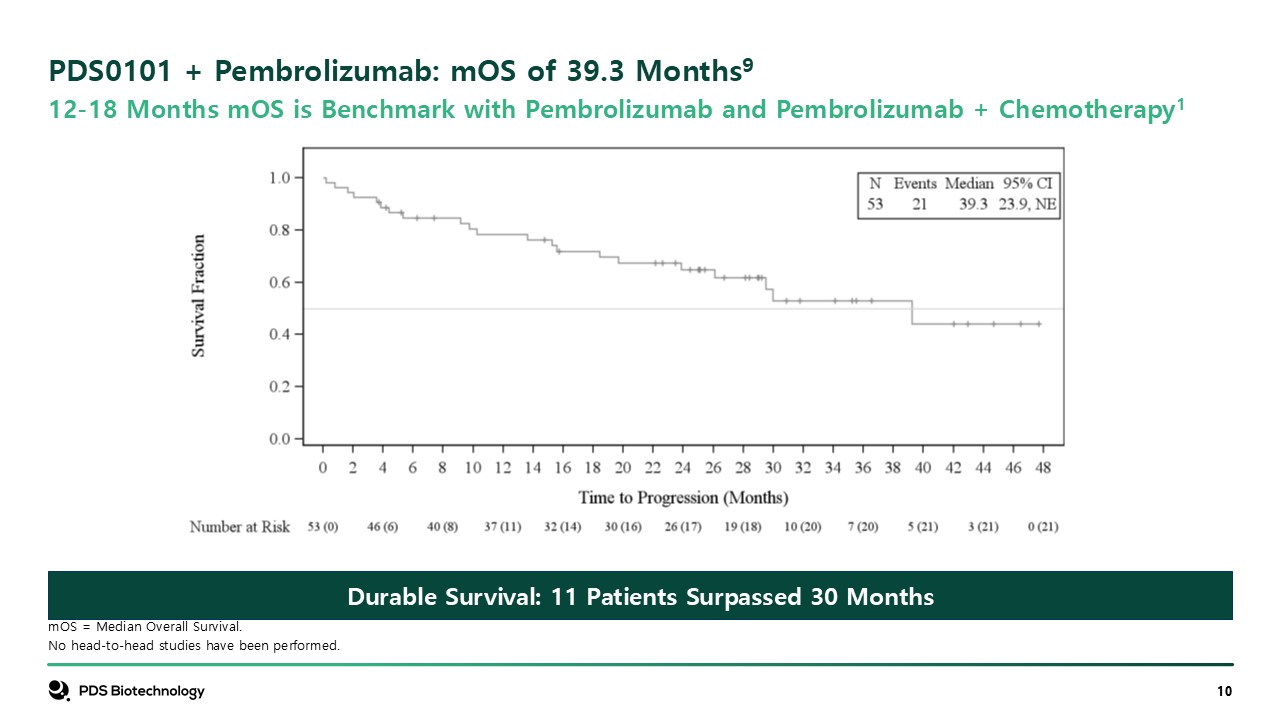
PDS0101 + Pembrolizumab: mOS of 39.3 Months9 mOS = Median Overall
Survival. No head-to-head studies have been performed. 12-18 Months mOS is Benchmark with Pembrolizumab and Pembrolizumab + Chemotherapy1 Durable Survival: 11 Patients Surpassed 30 Months
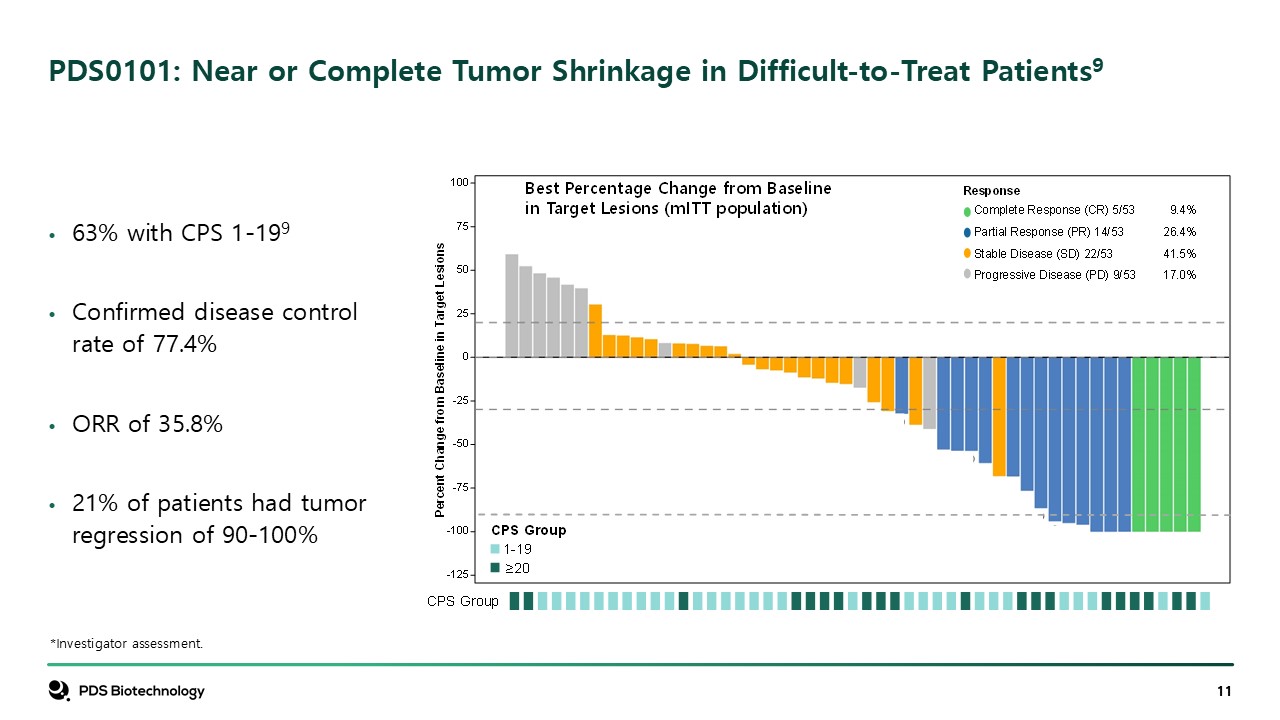
PDS0101: Near or Complete Tumor Shrinkage in Difficult-to-Treat
Patients9 *Investigator assessment. 63% with CPS 1-199 Confirmed disease control rate of 77.4% ORR of 35.8% 21% of patients had tumor regression of 90-100%
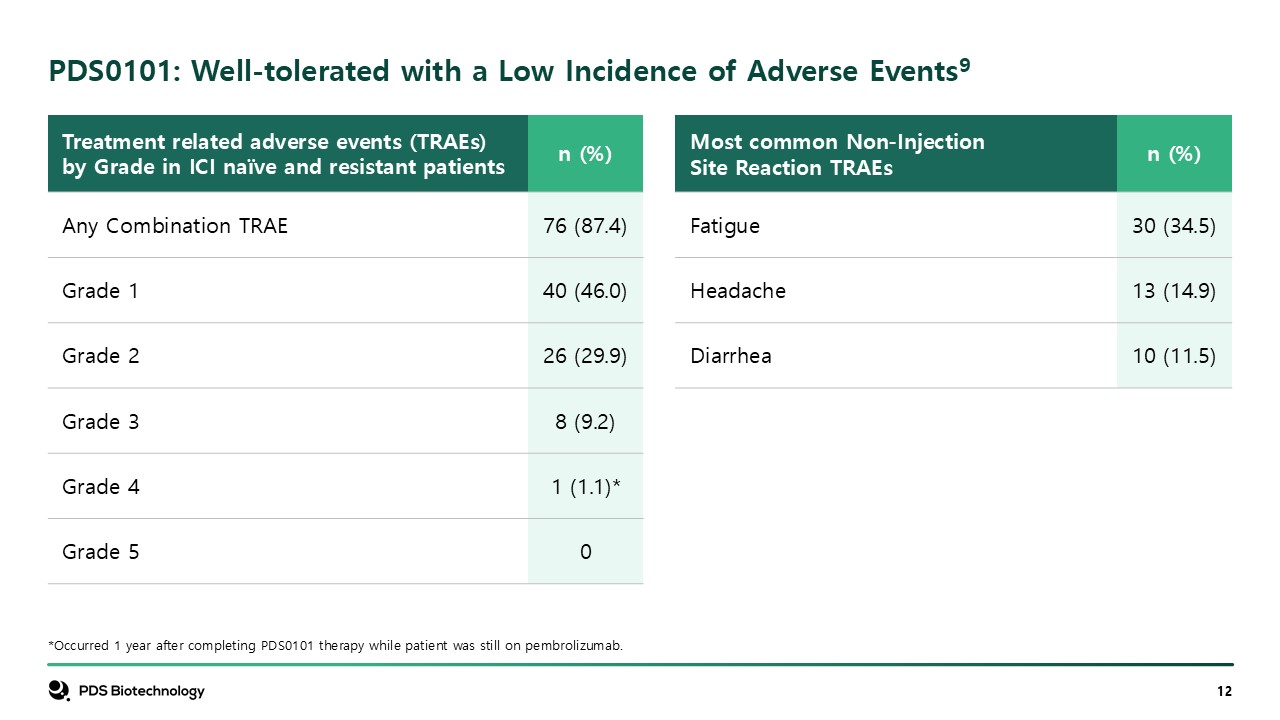
PDS0101: Well-tolerated with a Low Incidence of Adverse Events9 *Occurred 1
year after completing PDS0101 therapy while patient was still on pembrolizumab. Treatment related adverse events (TRAEs) by Grade in ICI naïve and resistant patients n (%) Any Combination TRAE 76 (87.4) Grade 1 40 (46.0) Grade
2 26 (29.9) Grade 3 8 (9.2) Grade 4 1 (1.1)* Grade 5 0 Most common Non-Injection Site Reaction TRAEs n (%) Fatigue 30 (34.5) Headache 13 (14.9) Diarrhea 10 (11.5)
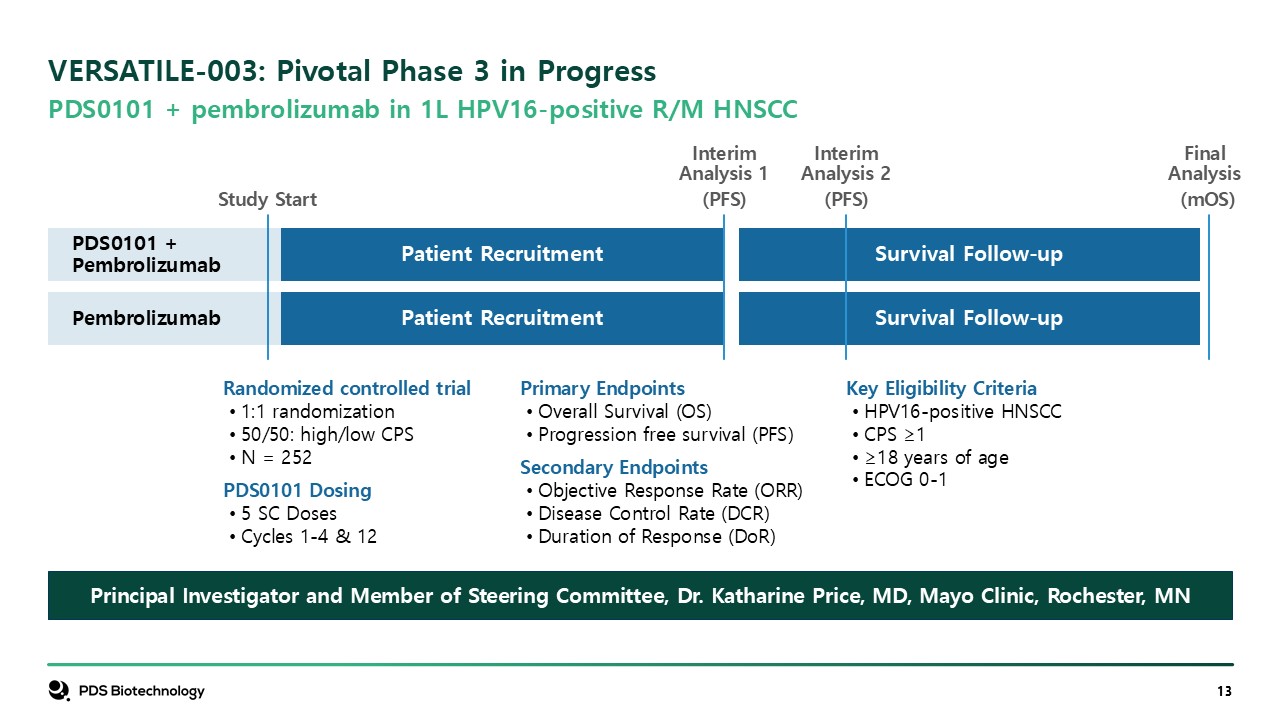
VERSATILE-003: Pivotal Phase 3 in Progress PDS0101 + pembrolizumab in 1L
HPV16-positive R/M HNSCC Key Eligibility Criteria HPV16-positive HNSCC CPS ≥1 ≥18 years of age ECOG 0-1 Primary Endpoints Overall Survival (OS) Progression free survival (PFS) Secondary Endpoints Objective Response Rate
(ORR) Disease Control Rate (DCR) Duration of Response (DoR) Randomized controlled trial 1:1 randomization 50/50: high/low CPS N = 252 PDS0101 Dosing 5 SC Doses Cycles 1-4 & 12 Principal Investigator and Member of Steering
Committee, Dr. Katharine Price, MD, Mayo Clinic, Rochester, MN Patient Recruitment InterimAnalysis 1 (PFS) Study Start Patient Recruitment Survival Follow-up Survival Follow-up Final Analysis (mOS) InterimAnalysis
2 (PFS) PDS0101 + Pembrolizumab Pembrolizumab

Well-Positioned in 1L HPV16+ R/M HNSCC No head-to-head studies have been
performed. Combinations with pembrolizumab PDS0101: Only Subcutaneous Combination Therapy in Late-stage Development for 1L HPV16+ HNSCC Genmab10 Bicara11 BioNTech12 PDS Biotech Median OS Not disclosed 21.3 months 22.6 months 39.3
months 95% CI (23.9, NE) Population All comers HPV-negative HPV16-positive HPV16-positive Administration Convenience IV Q2W until PD or toxicity IV QW (D1, D8, D15) IV Q1W 8X then Q3W for 24mos 5 subcutaneous injections of
PDS0101 Cycles 1-4 Q3W & then Cycle 12
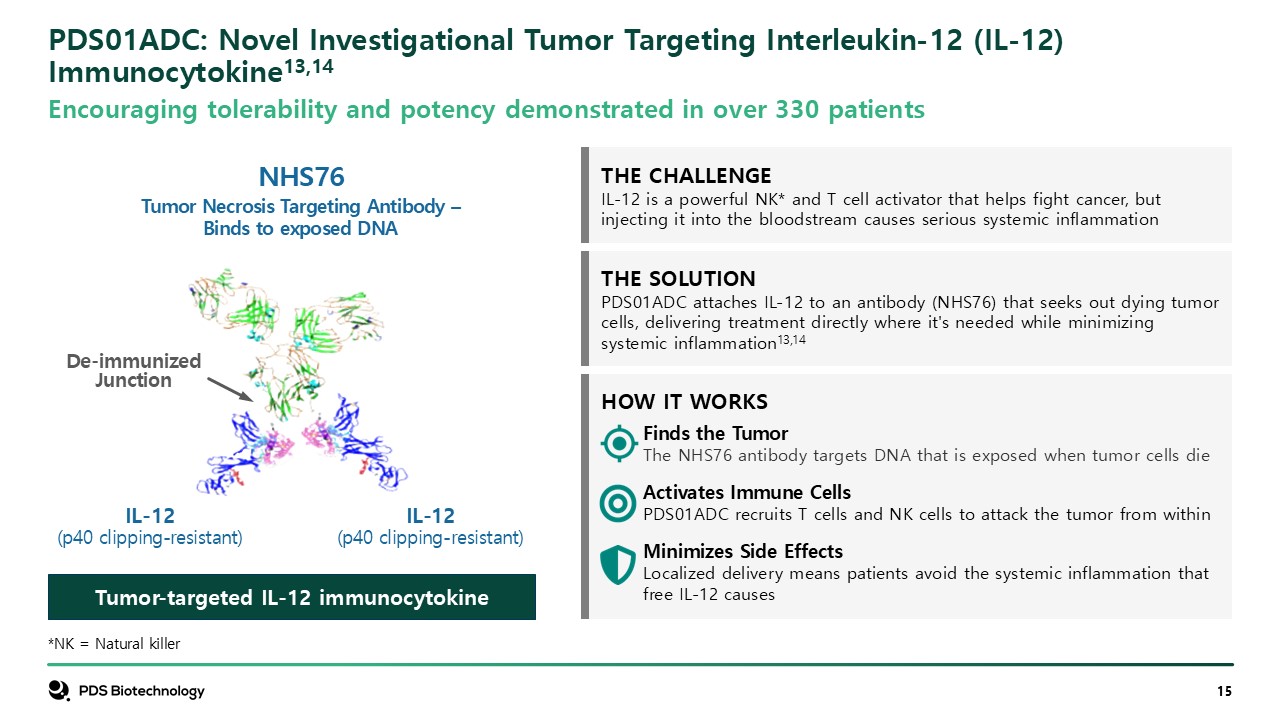
Tumor-targeted IL-12 immunocytokine THE SOLUTION PDS01ADC attaches IL-12 to
an antibody (NHS76) that seeks out dying tumor cells, delivering treatment directly where it's needed while minimizing systemic inflammation13,14 HOW IT WORKS Finds the TumorThe NHS76 antibody targets DNA that is exposed when tumor cells
die Activates Immune CellsPDS01ADC recruits T cells and NK cells to attack the tumor from within Minimizes Side EffectsLocalized delivery means patients avoid the systemic inflammation that free IL-12 causes THE CHALLENGE IL-12 is a
powerful NK* and T cell activator that helps fight cancer, but injecting it into the bloodstream causes serious systemic inflammation NHS76 Tumor Necrosis Targeting Antibody – Binds to exposed DNA De-immunized Junction PDS01ADC: Novel
Investigational Tumor Targeting Interleukin-12 (IL-12) Immunocytokine13,14 *NK = Natural killer Encouraging tolerability and potency demonstrated in over 330 patients IL-12 (p40 clipping-resistant) IL-12 (p40 clipping-resistant)
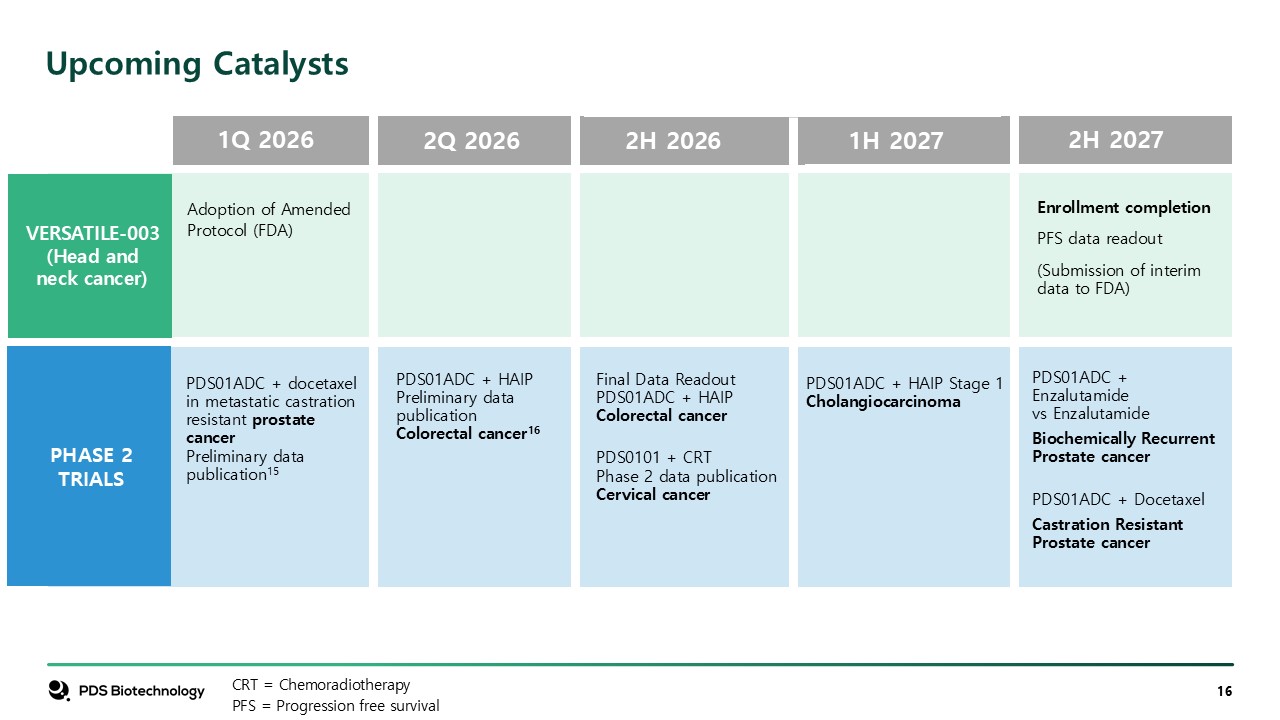
Enrollment completion PFS data readout (Submission of interim data to
FDA) PDS01ADC + HAIP Stage 1 Cholangiocarcinoma PDS01ADC + Enzalutamide vs Enzalutamide Biochemically Recurrent Prostate cancer PDS01ADC + Docetaxel Castration Resistant Prostate cancer Final Data ReadoutPDS01ADC + HAIP Colorectal
cancer PDS0101 + CRT Phase 2 data publication Cervical cancer VERSATILE-003 (Head and neck cancer) 2Q 2026 PDS01ADC + HAIPPreliminary data publicationColorectal cancer16 Upcoming Catalysts CRT = Chemoradiotherapy PFS = Progression
free survival PHASE 2 TRIALS 2H 2026 1H 2027 2H 2027 1Q 2026 PDS01ADC + docetaxel in metastatic castration resistant prostate cancerPreliminary data publication15 Adoption of Amended Protocol (FDA)

COVERAGE Composition and method claims protecting both platform technology
and product candidates 13 Patent Families PDS0101 EXCLUSIVITY 2041/2042 Global IP Portfolio Covers Key Commercial Markets 8 North America Asia Pacific Europe Middle
East USA Canada Mexico EU/UK Israel China Japan Australia KEYMARKETS
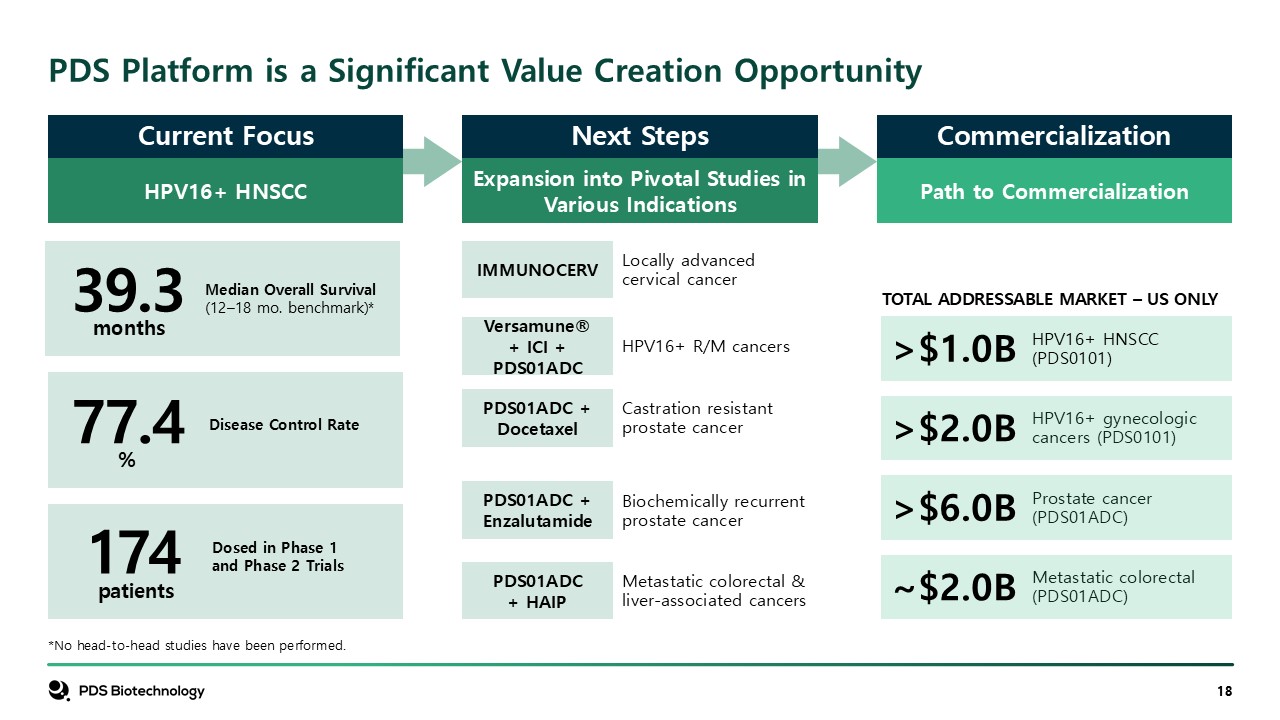
HPV16+ HNSCC PDS Platform is a Significant Value Creation
Opportunity Locally advanced cervical cancer Castration resistant prostate cancer HPV16+ R/M cancers Biochemically recurrent prostate cancer Metastatic colorectal & liver-associated cancers >$1.0B TOTAL ADDRESSABLE MARKET –
US ONLY HPV16+ HNSCC (PDS0101) HPV16+ gynecologic cancers (PDS0101) Prostate cancer (PDS01ADC) Metastatic colorectal (PDS01ADC) Current Focus Next Steps Commercialization Types Expansion into Pivotal Studies in Various
Indications Path to Commercialization PDS01ADC + Docetaxel IMMUNOCERV Versamune® + ICI + PDS01ADC PDS01ADC + Enzalutamide PDS01ADC + HAIP >$2.0B >$6.0B ~$2.0B 39.3 77.4 174 months % Median Overall Survival (12–18 mo.
benchmark)* Disease Control Rate Dosed in Phase 1 and Phase 2 Trials *No head-to-head studies have been performed. patients

The only investigational therapy in Phase 3 with 30+ months median survival in
1L HPV16+ R/M HNSCC $1.2 - 1.8B The PDS Opportunity World-Class Research PartnersNCI, Mayo Clinic, MD Anderson and leading cancer centers with studies externally funded, independently validated Global IP Protection13 patent families
across 8 markets; PDS0101 exclusivity through 2041/2042 Registrational TrialPhase 3 VERSATILE-003 underway; FDA-aligned on primary endpoint for accelerated approval Encouraging Data Supports Broader DevelopmentPDS01ADC programs in
colorectal and prostate cancer progressing toward potential advancement US Market Potential* *Company estimates based on external market research

Thank You NASDAQ: PDSB April 2026

References Harrington, KJ, Burtness B, Greil R, et al. Keytruda With or
Without Chemotherapy in Recurrent or Metastatic Head and Neck Squamous Cell Carcinoma: Updated Results of the Phase III KEYNOTE-048 Study. J Clin Oncol. 2022;41:790-802. https://doi.org/10.1200/JCO.21.02508. Damgacioglu H, Sonawane K,
Chhatwal J, et al. Long-term impact of HPV vaccination and COVID-19 pandemic on oropharyngeal cancer incidence and burden among men in the USA: A modeling Study. The Lancet Regional Health – Americas. 2022;8:100143. Tabatabaeian H et al,
Navigating therapeutic strategies: HPV classification in head and neck cancer, British Journal of Cancer. (2024) 131: 220-230. Landy R, et al JNCI: Upper age limits for US male human papillomavirus vaccination for oropharyngeal cancer
prevention: a microsimulation-based modeling study; Journal of the National Cancer Institute, 2023, 115(4), 429–436. Luo X et al; HPV16 drives cancer immune escape via NLRX1-mediated degradation of STING; J Clin Invest.
2020;130(4):1635–1652. Ziai H. et al; Does HPV Subtype Predict Outcomes in Head and Neck Cancers?; International Journal of Otolaryngology; Volume 2021, Article ID 6672373; https://doi.org/10.1155/2021/6672373. Lee L et al; Human
Papillomavirus-16 Infection in Advanced Oral Cavity Cancer Patients Is Related to an Increased Risk of Distant Metastases and Poor Survival; PLOS One; July 2012, Volume 7, Issue 7, e40767. Gandhapudi SK, Ward M, Bush JPC, Bedu-Addo F, Conn
G, Woodward JG. Antigen Priming with Enantiospecific Cationic Lipid Nanoparticles Induces Potent Antitumor CTL Responses through Novel Induction of a Type I IFN Response. J Immunol. 2019;202:3524-3536 Weiss J et al. VERSATILE-002: Overall
Survival of HPV16 Positive Recurrent/Metastatic Head and Neck Squamous Cell Carcinoma Patients Treated with T Cell Stimulating Immunotherapy PDS0101 and Keytruda. Poster Presented: ASCO Congress 2025; June 2, 2025. Van Herpen CML et al,
Petosemtamab (MCLA-158) with pembrolizumab as first line (1L) treatment of PD-L1+ recurrent/metastatic (r/m) head and neck squamous cell carcinoma (HNSCC): Phase 2 Trial, ASCO 2025 Cortese T, Ficerafusp Alfa/Pembrolizumab Receives FDA BTD
in Frontline HNSCC, Cancer Network, October 14, 2025 Saba NF et al, Exploratory analysis of antitumor activity and translational results from the safety run-in of AHEAD-MERIT, a Phase 2 trial of first-line pembrolizumab plus the
fixed-antigen cancer vaccine BNT113 in advanced HPV16+ HNSCC, ESMO 2024 Minnar CM et al (2024) Preclinical and clinical studies of a tumor targeting IL-12 immunocytokine. Front. Oncol. 13:1321318.doi: 10.3389/fonc.2023.1321318 Greiner JW
et al NHS-IL12, a Tumor-Targeting Immunocytokine; ImmunoTargets and Therapy 2021:10 155–169 Abel ML et al, Abstract PR008: Docetaxel and the Tumor Targeting Interleukin-12 (IL-12) PDS01ADC in Patients with Metastatic Castration Resistant
Prostate Cancer (mCRPC), Cancer Research, Vol. 86, Issue 2 Supplement, 15 January 2026 Eade AV et al; Tumor-Targeted IL-12 (PDS01ADC) with Hepatic Artery Infusion Pump Therapy for Colorectal Liver Metastases: Interim Analysis of a
Non-randomized Phase II Tria; JCO Oncol Adv 3, e2500173 (2026) Vol. 3 #1