
Exhibit 99.2

NYSE American: OSTX Biomarker - focused Conference Call April 30, 2026

Safe Harbor Statement This document contains forward - looking statements . In addition, from time to time, we or our representatives may make forward - looking statements orally or in writing . We base these forward - looking statements on our expectations and projections about future events, which we derive from the information currently available to us . Such forward - looking statements relate to future events or our future performance, including : our financial performance and projections ; our growth in revenue and earnings ; and our business prospects and opportunities . You can identify forward - looking statements by those that are not historical in nature, particularly those that use terminology such as “may,” “should,” “expects,” “anticipates,” “contemplates,” “estimates,” “believes,” “plans,” “projected,” “predicts,” “potential,” or “hopes” or the negative of these or similar terms . In evaluating these forward - looking statements, you should consider various factors, including : our ability to change the direction of the Company ; our ability to keep pace with new technology and changing market needs ; and the competitive environment of our business . These and other factors may cause our actual results to differ materially from any forward - looking statement . Forward - looking statements are only predictions . The forward - looking events discussed in this document and other statements made from time to time by us or our representatives, may not occur, and actual events and results may differ materially and are subject to risks, uncertainties and assumptions about us . We are not obligated to publicly update or revise any forward - looking statement, whether as a result of uncertainties and assumptions, the forward - looking events discussed in this document and other statements made from time to time by us or our representatives might not occur .

Agenda 1. Introduction – Harrison Seidner, PhD 2. Corporate Update – Paul Romness, MPH 3. Biotech Innovation Curve – Dr. Robert Langer 4. Clinical, Biomarker and Safety Data – Andrew Exley, MD, FRCP, FRCPath 5. Regulatory status by Jurisdiction (Europe, Australia, U.K. and U.S.) – David Brindley, PhD 6. Insights on developing treatments for rare pediatric cancers – Dr. Craig Eagle 7. Questions

Agenda 1. Introduction – Harrison Seidner, PhD 2. Corporate Update – Paul Romness, MPH 3. Biotech Innovation Curve – Dr. Robert Langer 4. Clinical, Biomarker and Safety Data – Andrew Exley, MD, FRCP, FRCPath 5. Regulatory status by Jurisdiction (Europe, Australia, U.K. and U.S.) – David Brindley, PhD 6. Insights on developing treatments for rare pediatric cancers – Dr. Craig Eagle 7. Questions

Agenda 1. Introduction – Harrison Seidner, PhD 2. Corporate Update – Paul Romness, MPH 3. Biotech Innovation Curve – Dr. Robert Langer 4. Clinical, Biomarker and Safety Data – Andrew Exley, MD, FRCP, FRCPath 5. Regulatory status by Jurisdiction (Europe, Australia, U.K. and U.S.) – David Brindley, PhD 6. Insights on developing treatments for rare pediatric cancers – Dr. Craig Eagle 7. Questions

Agenda 1. Introduction – Harrison Seidner, PhD 2. Corporate Update – Paul Romness, MPH 3. Biotech Innovation Curve – Dr. Robert Langer 4. Clinical, Biomarker and Safety Data – Andrew Exley, MD, FRCP, FRCPath 5. Regulatory status by Jurisdiction (Europe, Australia, U.K. and U.S.) – David Brindley, PhD 6. Insights on developing treatments for rare pediatric cancers – Dr. Craig Eagle 7. Questions

• Major form of pediatric bone cancer • ~1,000 cases diagnosed annually in US • Primarily affects adolescents C young adults: 12 – 39 yrs • Localized disease • Chemotherapy C Surgery can sometimes achieve remission • Recurrent / Metastatic disease e.g. in Lungs • No FDA - approved treatment options for recurrent, resected lung metastases • Systematic reviews highlight dearth of new therapies (Gazouli 2021, Biermann 2025) • Some progress on disease mechanisms BUT • Prognosis is dire with little improvement over the last 30 years (Cole 2022) • Justifies use of historical control data Osteosarcoma: Clinical Outline
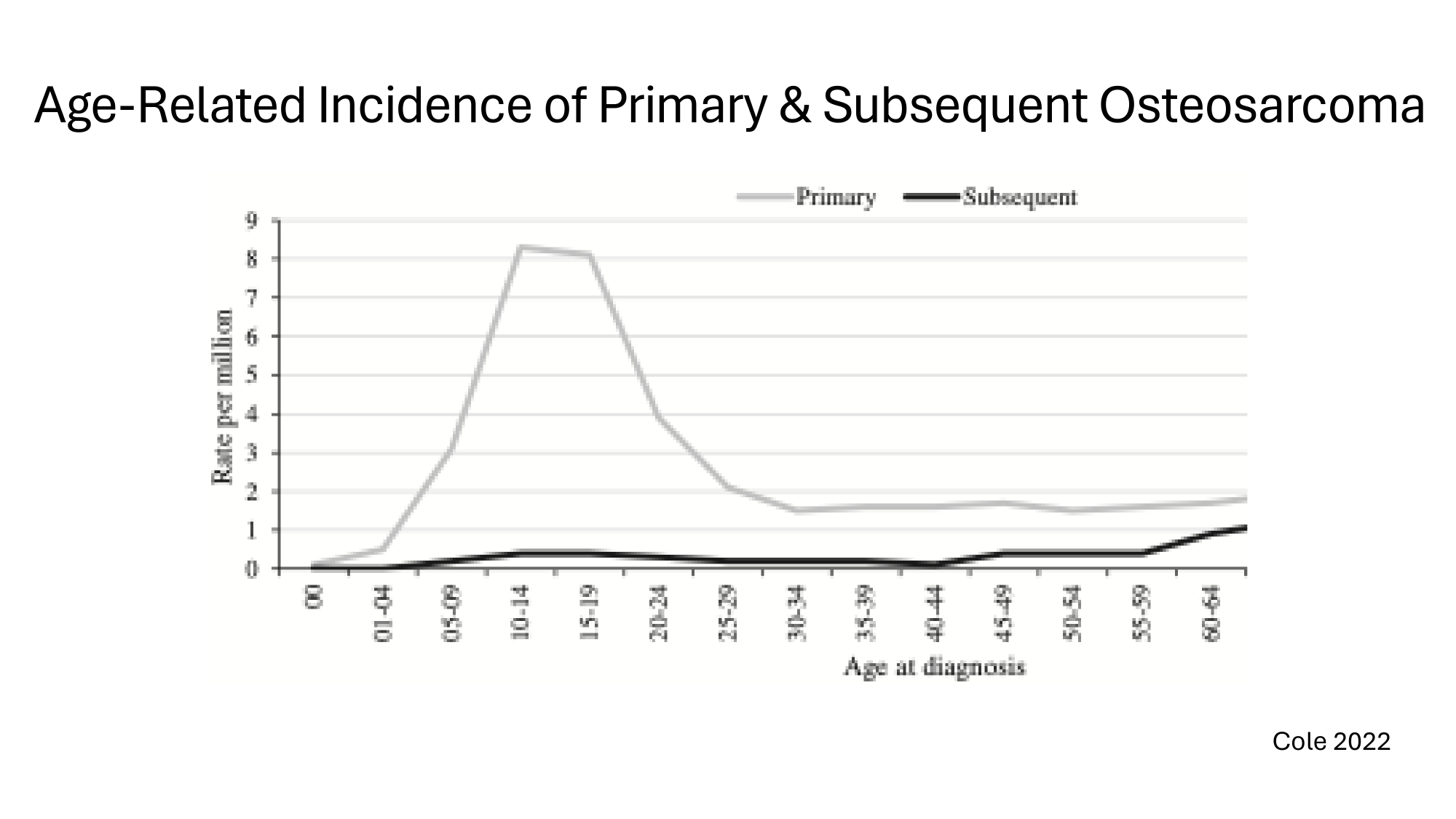
Age - Related Incidence of Primary s Subsequent Osteosarcoma Cole 2022
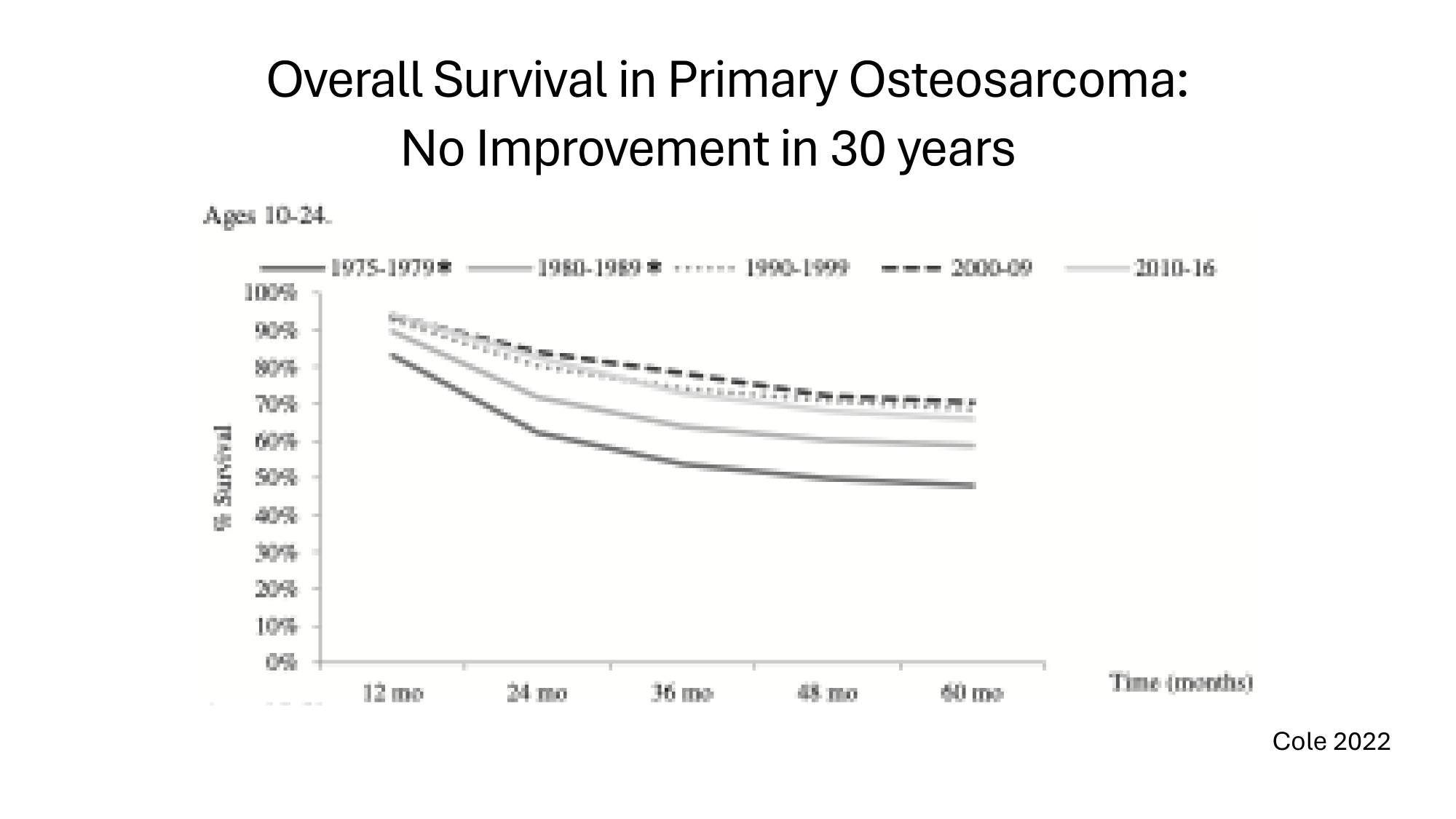
Overall Survival in Primary Osteosarcoma: No Improvement in 30 years Cole 2022
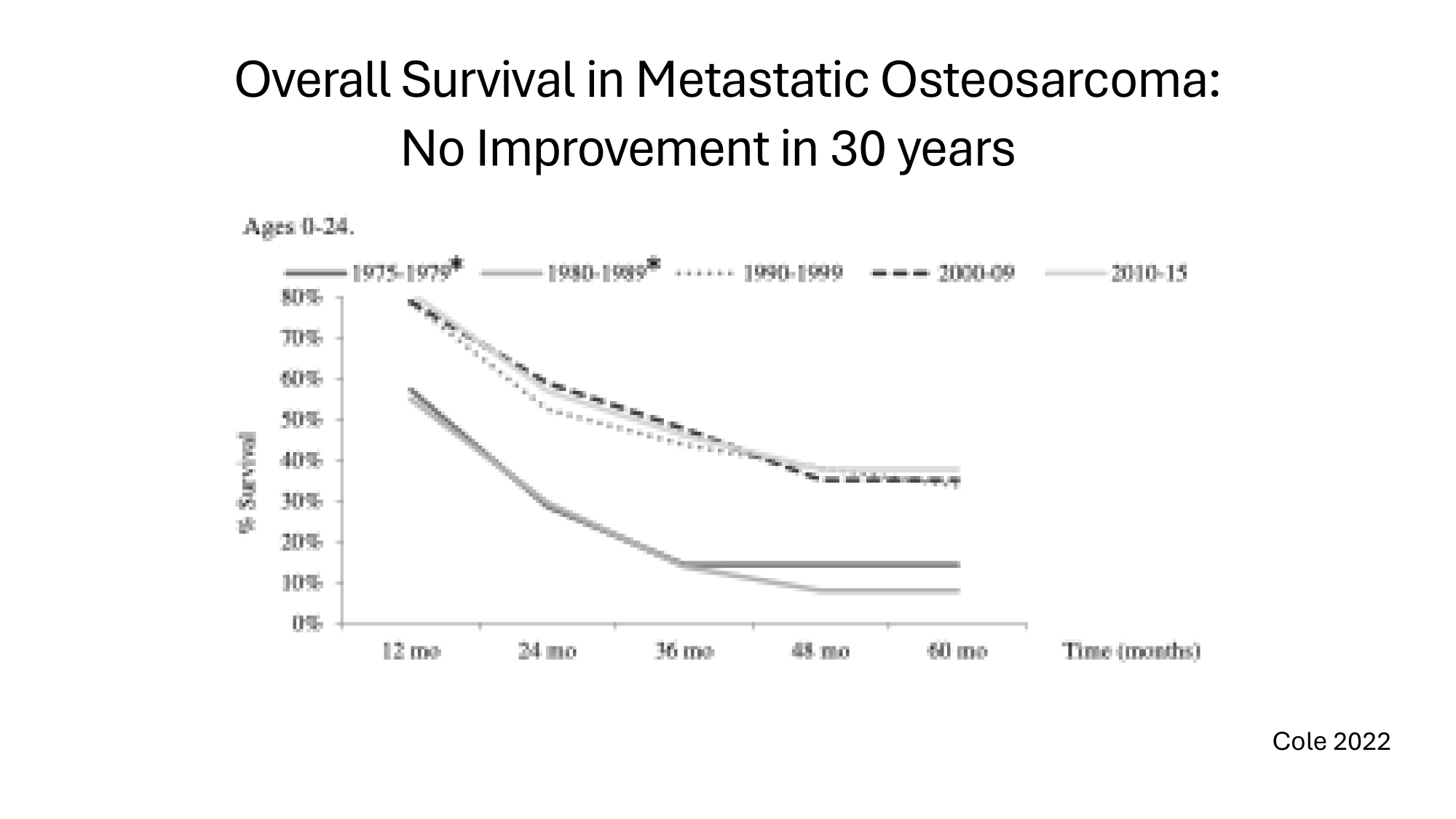
Overall Survival in Metastatic Osteosarcoma: No Improvement in 30 years Cole 2022
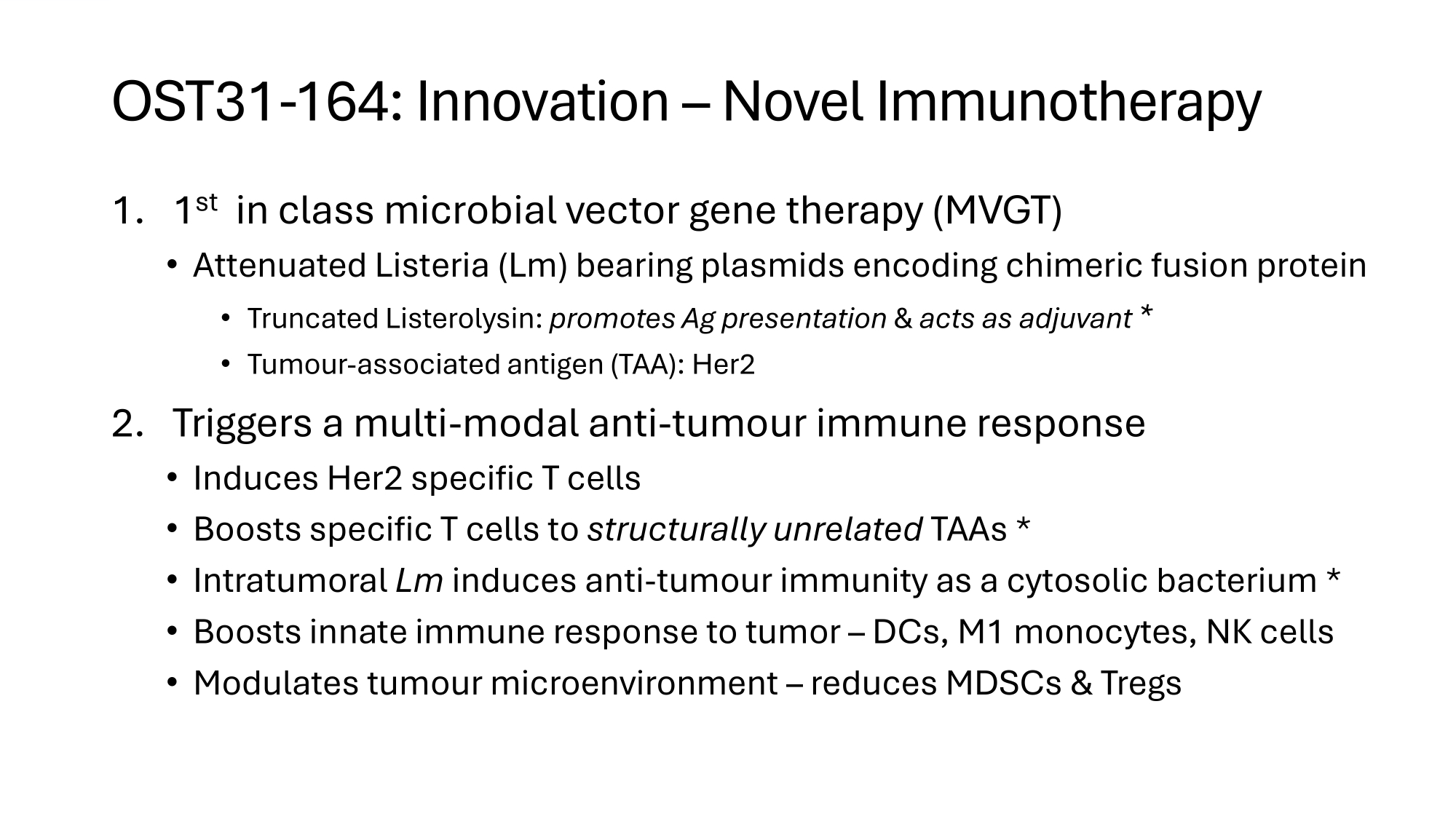
OST31 - 164: Innovation – Novel Immunotherapy 1. 1 st in class microbial vector gene therapy (MVGT) • Attenuated Listeria (Lm) bearing plasmids encoding chimeric fusion protein • Truncated Listerolysin: promotes Ag presentation C acts as adjuvant * • Tumour - associated antigen (TAA): Her2 2. Triggers a multi - modal anti - tumour immune response • Induces Her2 specific T cells • Boosts specific T cells to structurally unrelated TAAs * • Intratumoral Lm induces anti - tumour immunity as a cytosolic bacterium * • Boosts innate immune response to tumor – DCs, M1 monocytes, NK cells • Modulates tumour microenvironment – reduces MDSCs C Tregs
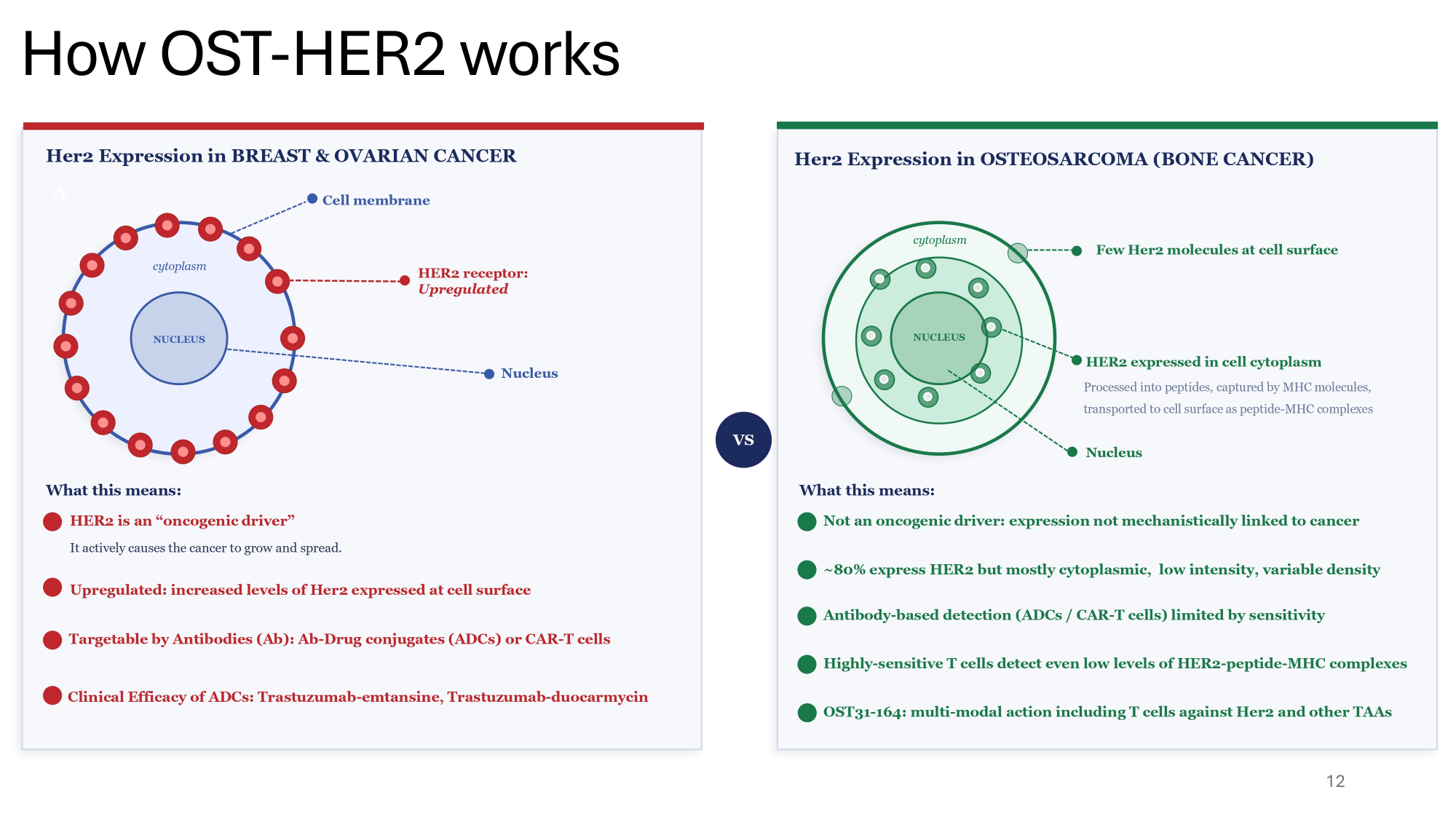
12 A Her2 Expression in BREAST & OVARIAN CANCER cytoplasm NUCLEUS Cell membrane HER2 receptor: Upregulated Nucleus What this means: HER2 is an “oncogenic driver” It actively causes the cancer to grow and spread. Upregulated: increased levels of Her2 expressed at cell surface Targetable by Antibodies (Ab): Ab - Drug conjugates (ADCs) or CAR - T cells Clinical Efficacy of ADCs: Trastuzumab - emtansine, Trastuzumab - duocarmycin VS Her2 Expression in OSTEOSARCOMA (BONE CANCER) cytoplasm Few Her2 molecules at cell surface NUCLEUS HER2 expressed in cell cytoplasm Processed into peptides, captured by MHC molecules, transported to cell surface as peptide - MHC complexes Nucleus What this means: Not an oncogenic driver: expression not mechanistically linked to cancer ~80% express HER2 but mostly cytoplasmic, low intensity, variable density Antibody - based detection (ADCs / CAR - T cells) limited by sensitivity Highly - sensitive T cells detect even low levels of HER2 - peptide - MHC complexes OST31 - 164: multi - modal action including T cells against Her2 and other TAAs How OST - HER2 works
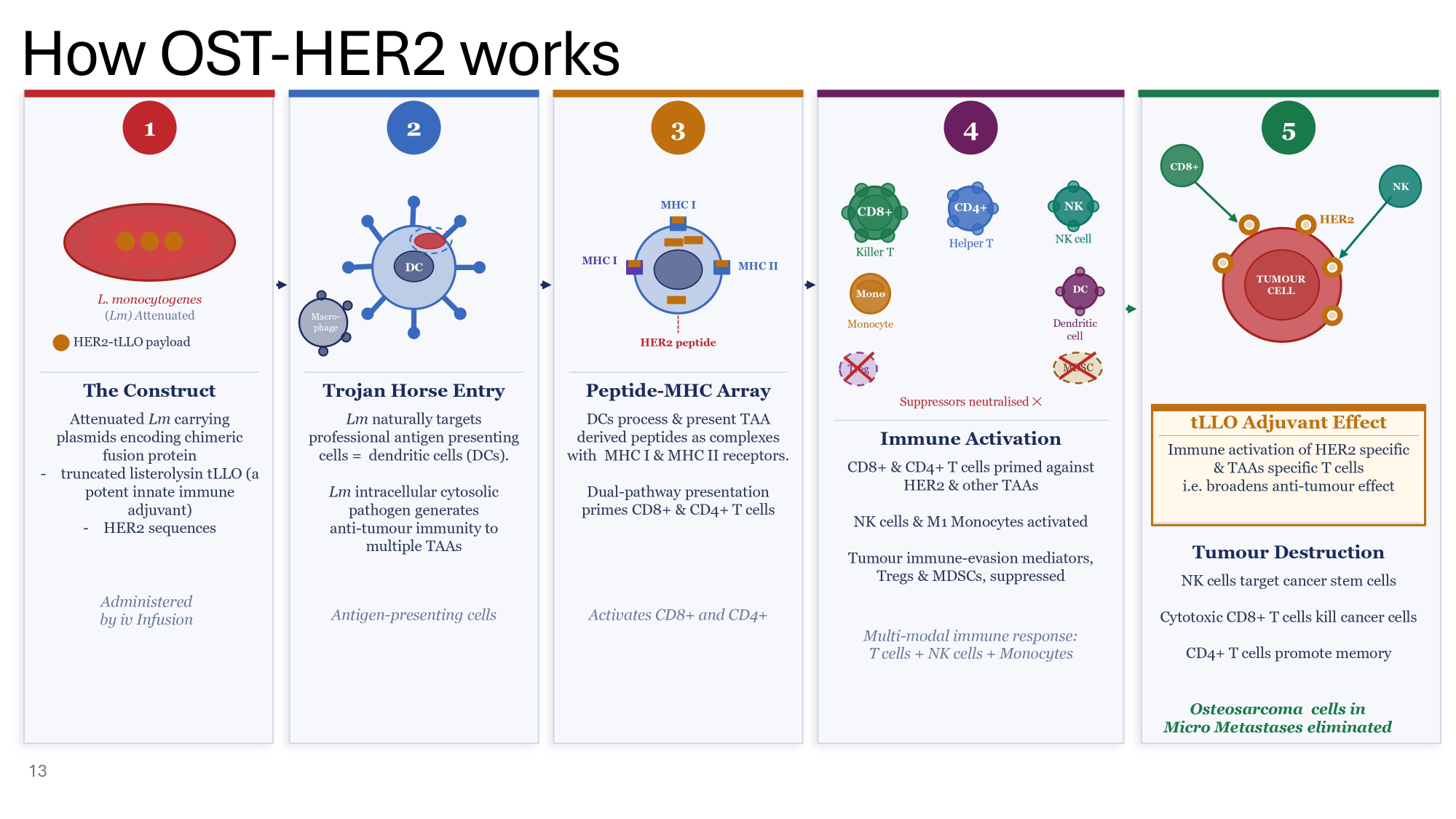
How OST - HER2 works 13 3 4 1 L. monocytogenes ( Lm) A ttenuated HER2 - tLLO payload The Construct Attenuated Lm carrying plasmids encoding chimeric fusion protein - truncated listerolysin tLLO (a potent innate immune adjuvant) - HER2 sequences Administered by iv Infusion 2 DC Macro - phage Trojan Horse Entry Lm naturally targets professional antigen presenting cells = dendritic cells (DCs). Lm intracellular cytosolic pathogen generates anti - tumour immunity to multiple TAAs Antigen - presenting cells MHC I MHC II MHC I HER2 peptide Peptide - MHC Array DCs process & present TAA derived peptides as complexes with MHC I & MHC II receptors. Dual - pathway presentation primes CD8+ & CD4+ T cells Activates CD8+ and CD4+ CD8+ Killer T CD4+ Helper T NK NK cell Mono Monocyte DC Dendritic cell Treg MDSC Suppressors neutralised ظ Immune Activation CD8+ & CD4+ T cells primed against HER2 & other TAAs NK cells & M1 Monocytes activated Tumour immune - evasion mediators, Tregs & MDSCs, suppressed Multi - modal immune response: T cells + NK cells + Monocytes tLLO Adjuvant Effect Immune activation of HER2 specific & TAAs specific T cells i.e. broadens anti - tumour effect 5 CD8+ NK HER2 TUMOUR CELL Tumour Destruction NK cells target cancer stem cells Cytotoxic CD8+ T cells kill cancer cells CD4+ T cells promote memory Osteosarcoma cells in Micro Metastases eliminated
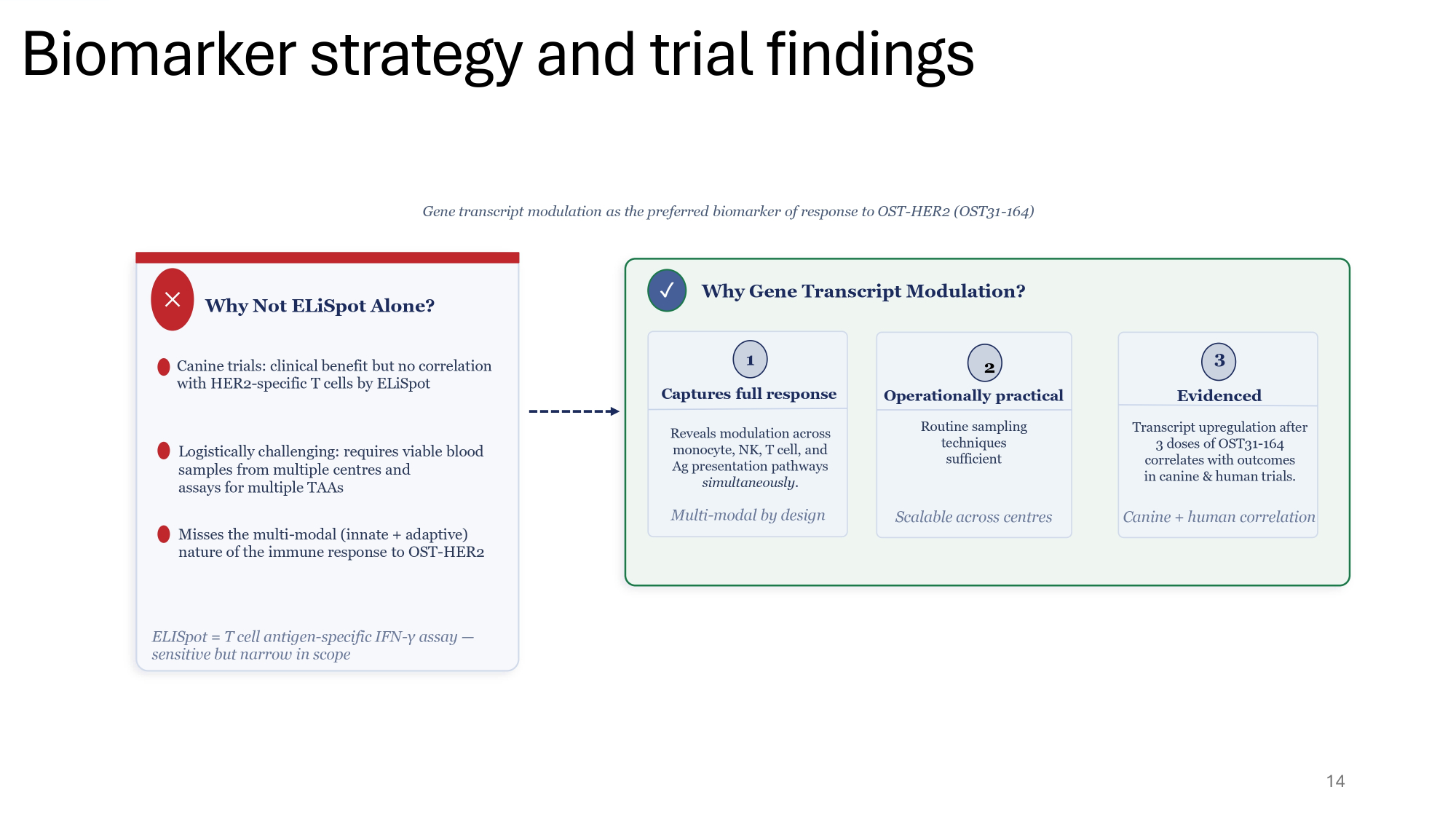
14 Gene transcript modulation as the preferred biomarker of response to OST - HER2 (OST31 - 164) ظ Why Not ELiSpot Alone? Canine trials: clinical benefit but no correlation with HER2 - specific T cells by ELiSpot Logistically challenging: requires viable blood samples from multiple centres and assays for multiple TAAs Misses the multi - modal (innate + adaptive) nature of the immune response to OST - HER2 ELISpot = T cell antigen - specific IFN - γ assay — sensitive but narrow in scope Reveals modulation across monocyte, NK, T cell, and Ag presentation pathways simultaneously . Multi - modal by design 1 Captures full response Routine sampling techniques sufficient Scalable across centres Transcript upregulation after 3 doses of OST31 - 164 correlates with outcomes in canine & human trials. Canine + human correlation 3 Evidenced 2 Operationally practical Why Gene Transcript Modulation? ض Biomarker strategy and trial findings
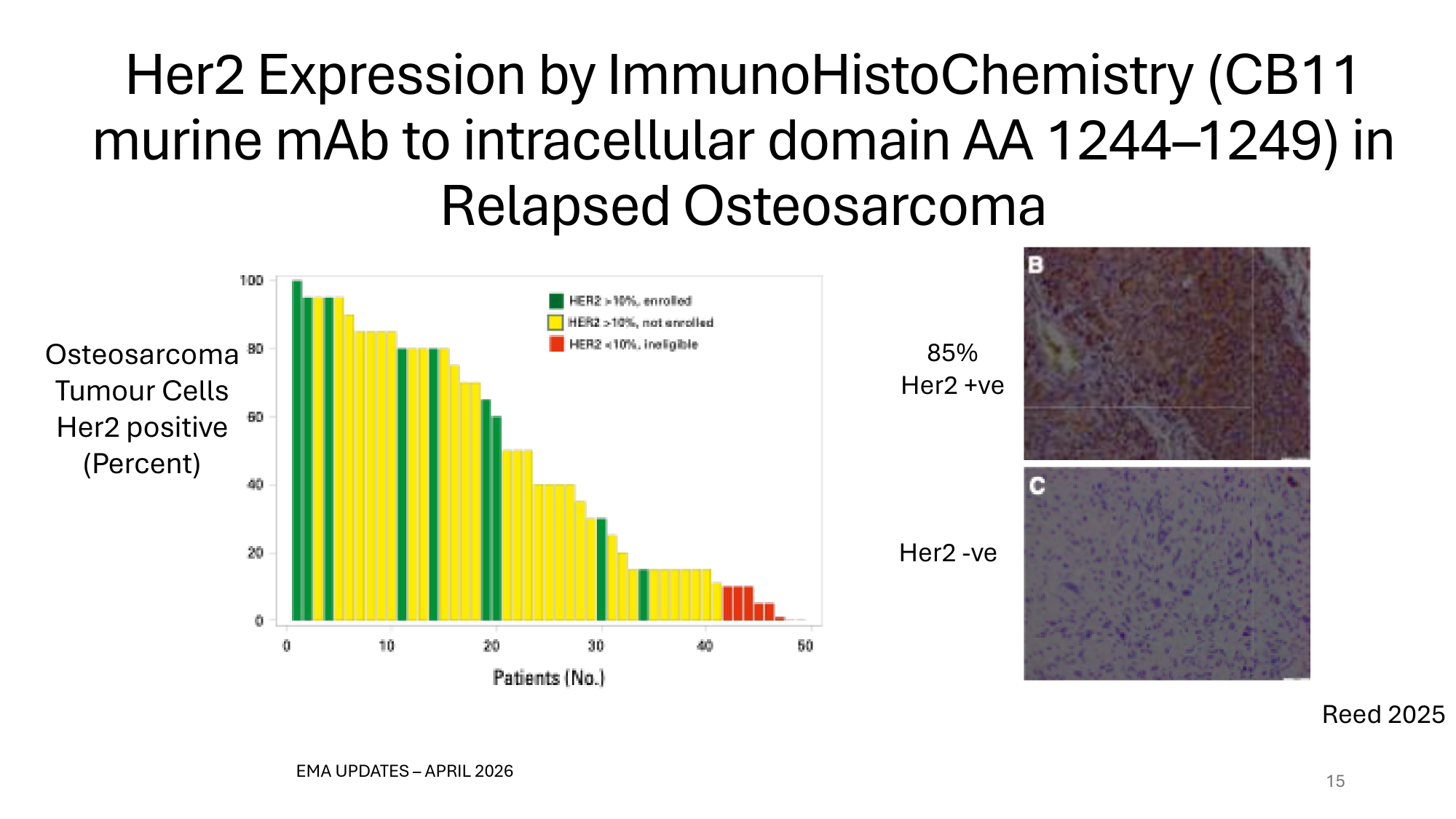
Her2 Expression by ImmunoHistoChemistry (CB11 murine mAb to intracellular domain AA 1244 – 1246) in Relapsed Osteosarcoma 15 Osteosarcoma Tumour Cells Her2 positive (Percent) EMA UPDATES – APRIL 2026 85% Her2 +ve Her2 - ve Reed 2025

OST31 - 164: Proof of Principle: Comparative Oncology 3. NIH supported collaboration with comparative oncology experts demonstrates efficacy of OST31 - 164 in spontaneous osteosarcoma in canines

Canine Osteosarcoma: Parallels with Human Osteosarcoma I. Spontaneous onset in immunocompetent dogs: rate >10x human rate II. Many clinical, biological, and molecular features in common i. Primary lesion: typically, high - grade tumours in long bones ii. Standard treatment: chemotherapy C radical surgery iii. Disease course: metastatic disease often in the lungs iv. Highly rearranged genomes often affecting TP53, CDKN2A, RB1 genes Similar molecular pathway alterations of prognostic significance • TH 1 C TH 2 immune cell signalling, Interferon signalling, Inflammatory responses III. IV. Share 3 distinct TME subtypes – independent predictors of PFS • Immune Enriched [IE]; IE dense extracellular matrix - like; Immune Desert Makielski 2019, Mannheimer 2023, Mason 2025, Patkar 2024

Immunological responses and clinical outcomes in dogs with osteosarcoma receiving SOC + ADXS31 - 164 Mason NJ 2025
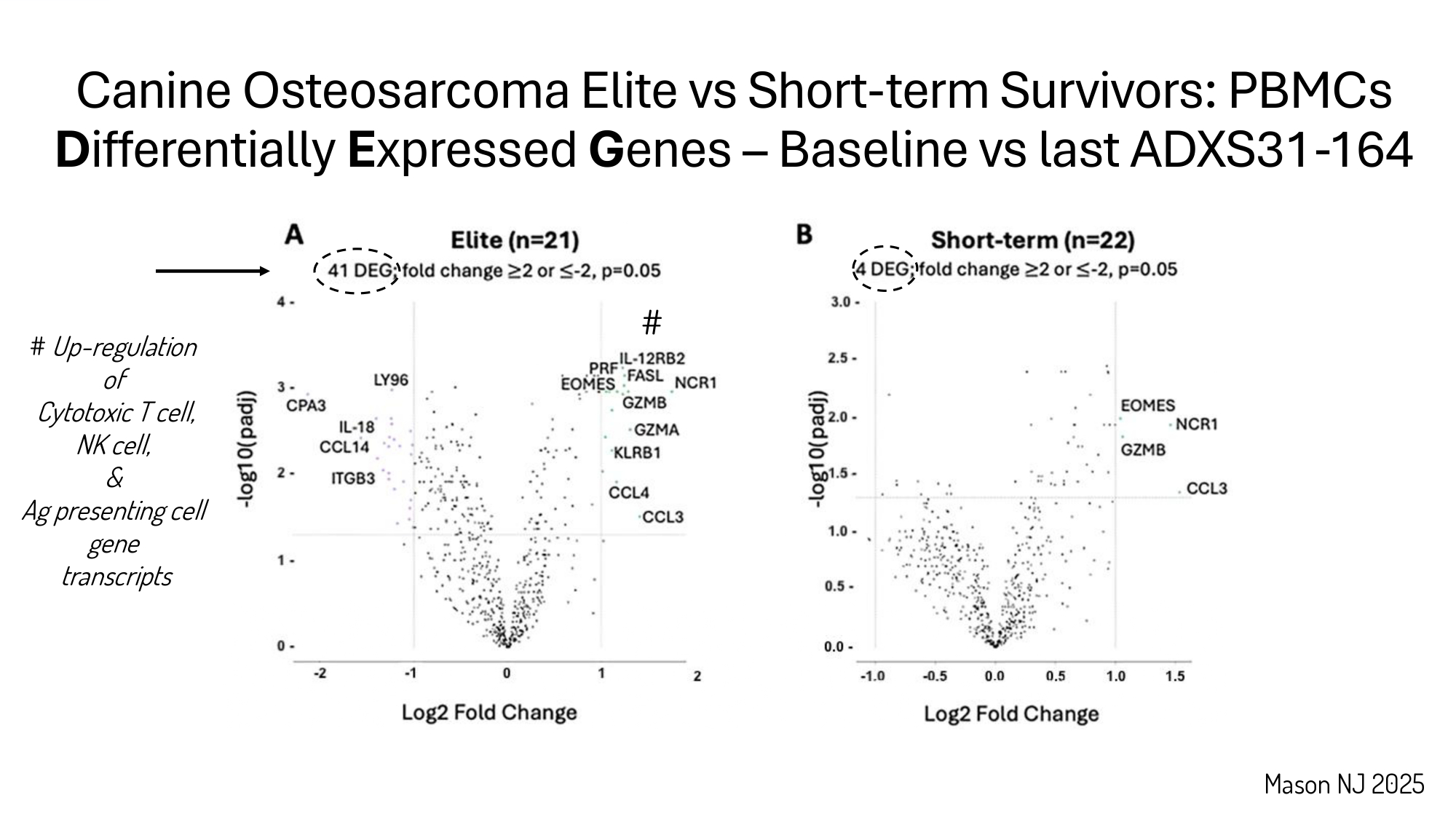
Mason NJ 2025 # Up - regulation of Cytotoxic T cell, NK cell, & Ag presenting cell gene transcripts Canine Osteosarcoma Elite vs Short - term Survivors: PBMCs D ifferentially E xpressed G enes – Baseline vs last ADXS31 - 164 #
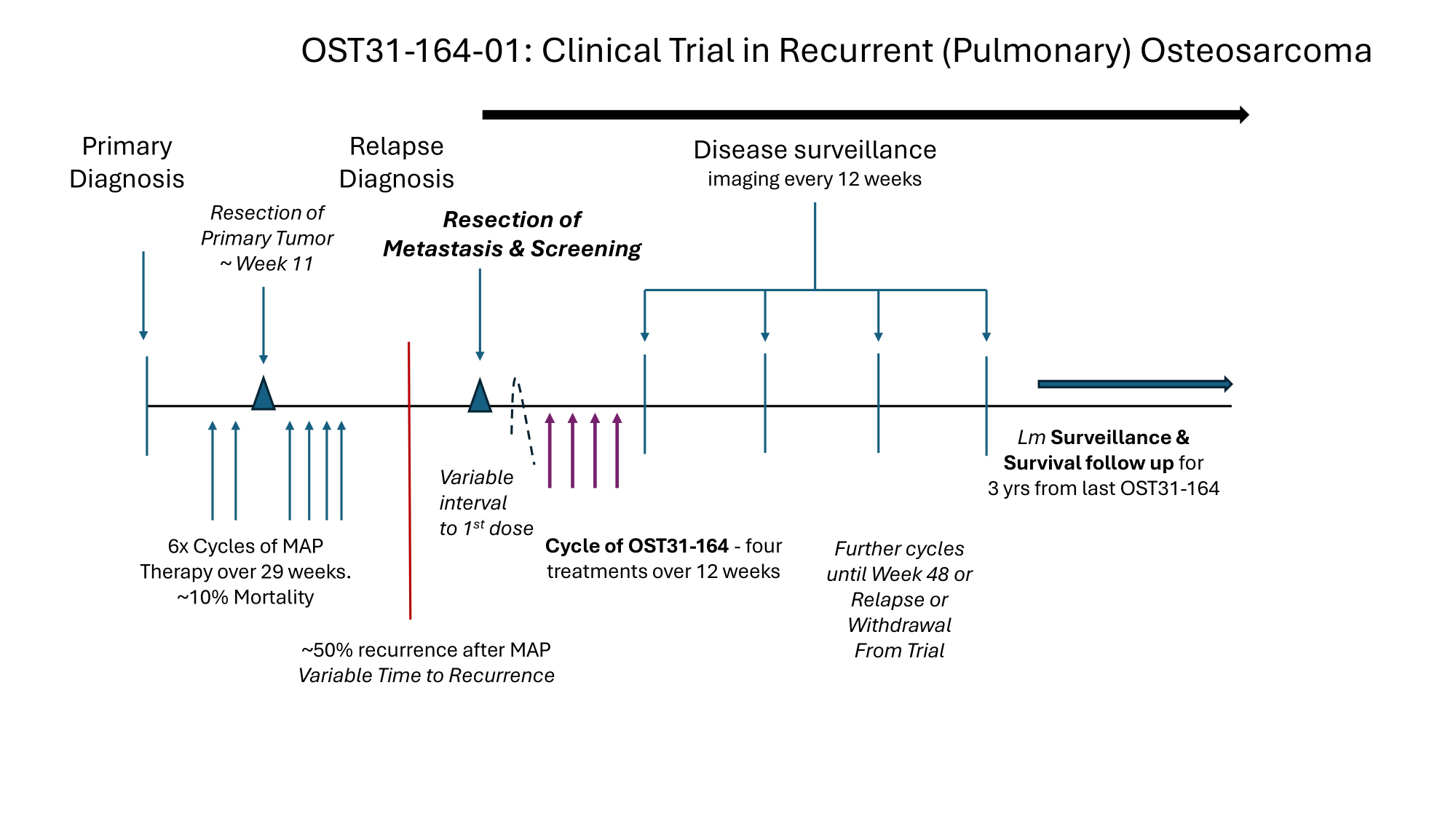
Primary Diagnosis Resection of Primary Tumor ~ Week 11 Cycle of OST31 - 164 - four treatments over 12 weeks Disease surveillance imaging every 12 weeks 6x Cycles of MAP Therapy over 29 weeks. ~10% Mortality ~50% recurrence after MAP Variable Time to Recurrence Lm Surveillance s Survival follow up for 3 yrs from last OST31 - 164 Relapse Diagnosis Resection of Metastasis & Screening Further cycles until Week 48 or Relapse or Withdrawal From Trial Variable interval to 1 st dose OST31 - 164 - 01: Clinical Trial in Recurrent (Pulmonary) Osteosarcoma

OST31 - 164: Innovation – Historic Controls 4. Justification for use of Historic Controls i. Lack of improvement in OS (Cole 2022) i. Reduces risk of bias thru improvements in Standard Of Care (SOC) ii. Systematic identification of published trials iii. Systematic screening for comparable populations iv. Conservative approach : treatment effect of chemotherapy +/ - IND in controls v. Systematic extraction of data from comparable populations

Rationale for EFS vs Objective Response Rate (ORR) I. Radiographic response may not reflect critical cellular effect II. Surgical resection is standard approach to pulmonary recurrence i.e. no lesion to determine ORR III. Likely difference in drug activity in microscopic vs macroscopic disease IV. Propose EFS vs historical benchmark A. Measurable, unresectable osteosarcoma • EFS < 4 months vs >4 months = disease control failure v success B. Completely resected osteosarcoma • EFS < 12 months vs >12 months Lagmay 2016

Rationale for Preferring Overall Survival to Event Free Survival in the Assessment of Immunotherapies for Osteosarcoma 1. Patients value Overall survival over Event Free Survival (Tregear 2024) 2. Regulators prefer Overall Survival as a Hard Endpoint 3. Delayed effect of immunotherapies reduces discriminating power of 12month Event Free Survival
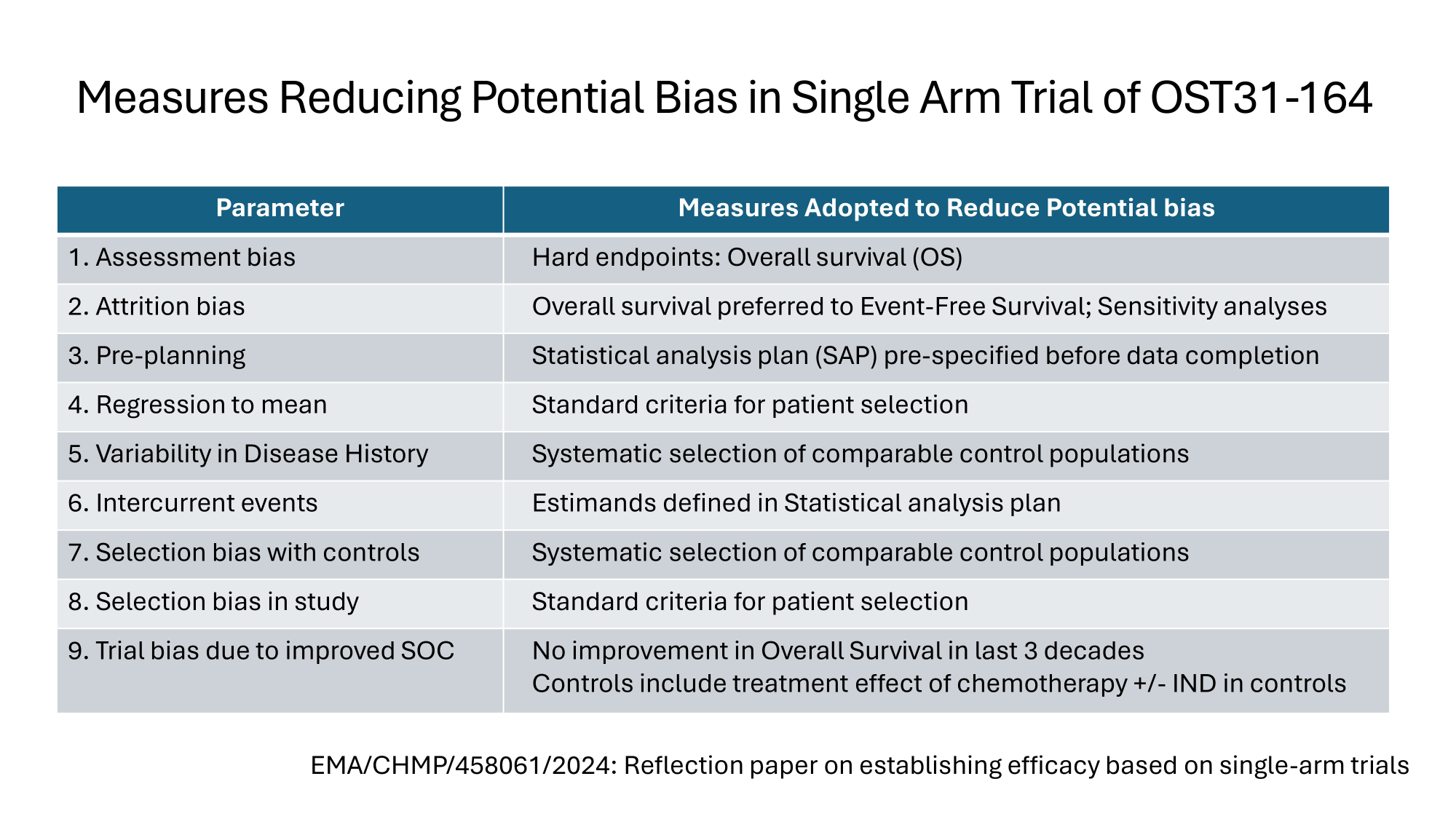
Measures Reducing Potential Bias in Single Arm Trial of OST31 - 164 Measures Adopted to Reduce Potential bias Parameter Hard endpoints: Overall survival (OS) 1. Assessment bias Overall survival preferred to Event - Free Survival; Sensitivity analyses 2. Attrition bias Statistical analysis plan (SAP) pre - specified before data completion 3. Pre - planning Standard criteria for patient selection 4. Regression to mean Systematic selection of comparable control populations 5. Variability in Disease History Estimands defined in Statistical analysis plan 6. Intercurrent events Systematic selection of comparable control populations 7. Selection bias with controls Standard criteria for patient selection 8. Selection bias in study No improvement in Overall Survival in last 3 decades Controls include treatment effect of chemotherapy +/ - IND in controls 9. Trial bias due to improved SOC EMA/CHMP/458061/2024: Reflection paper on establishing efficacy based on single - arm trials
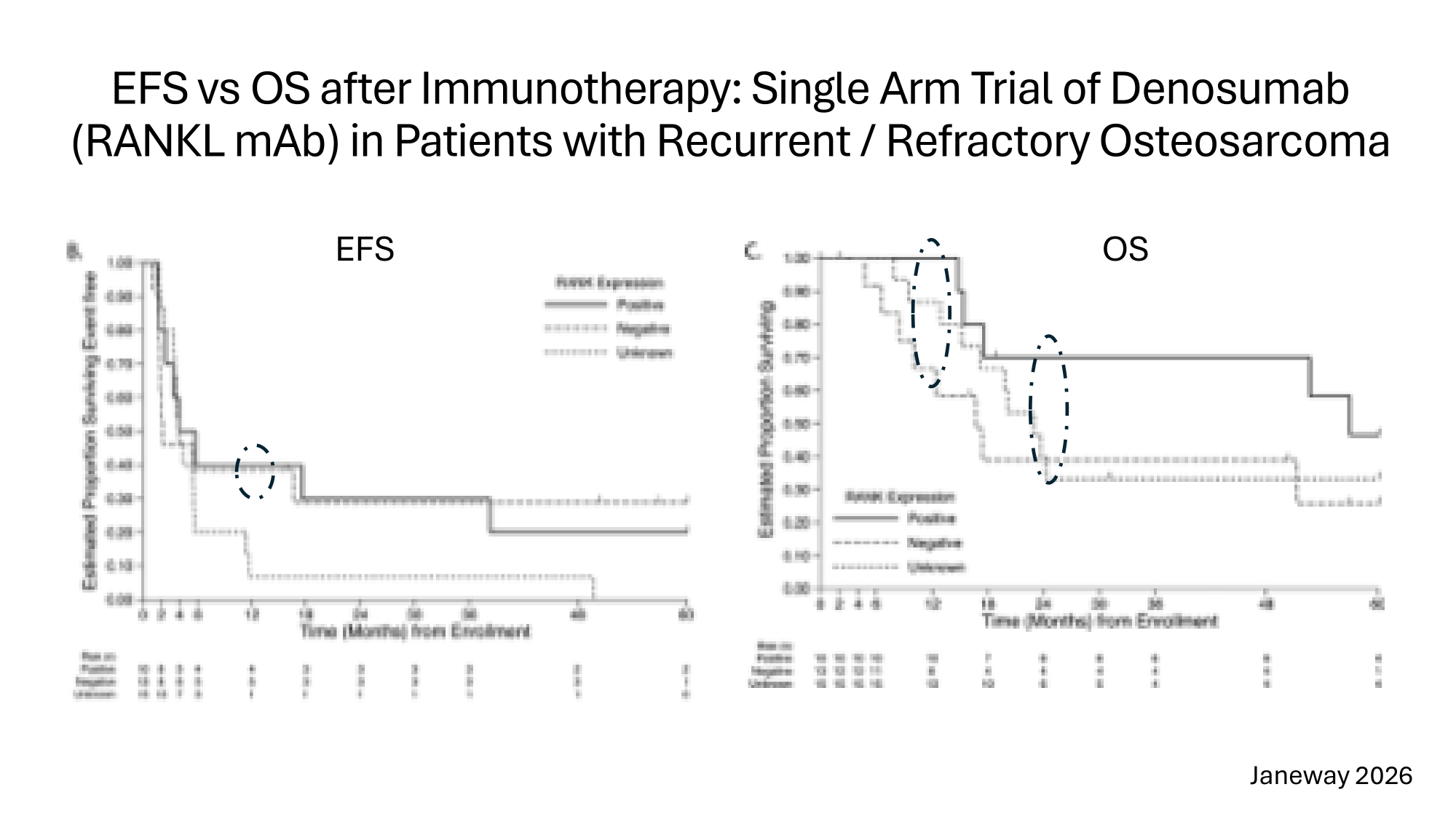
EFS vs OS after Immunotherapy: Single Arm Trial of Denosumab (RANKL mAb) in Patients with Recurrent / Refractory Osteosarcoma EFS OS Janeway 2026
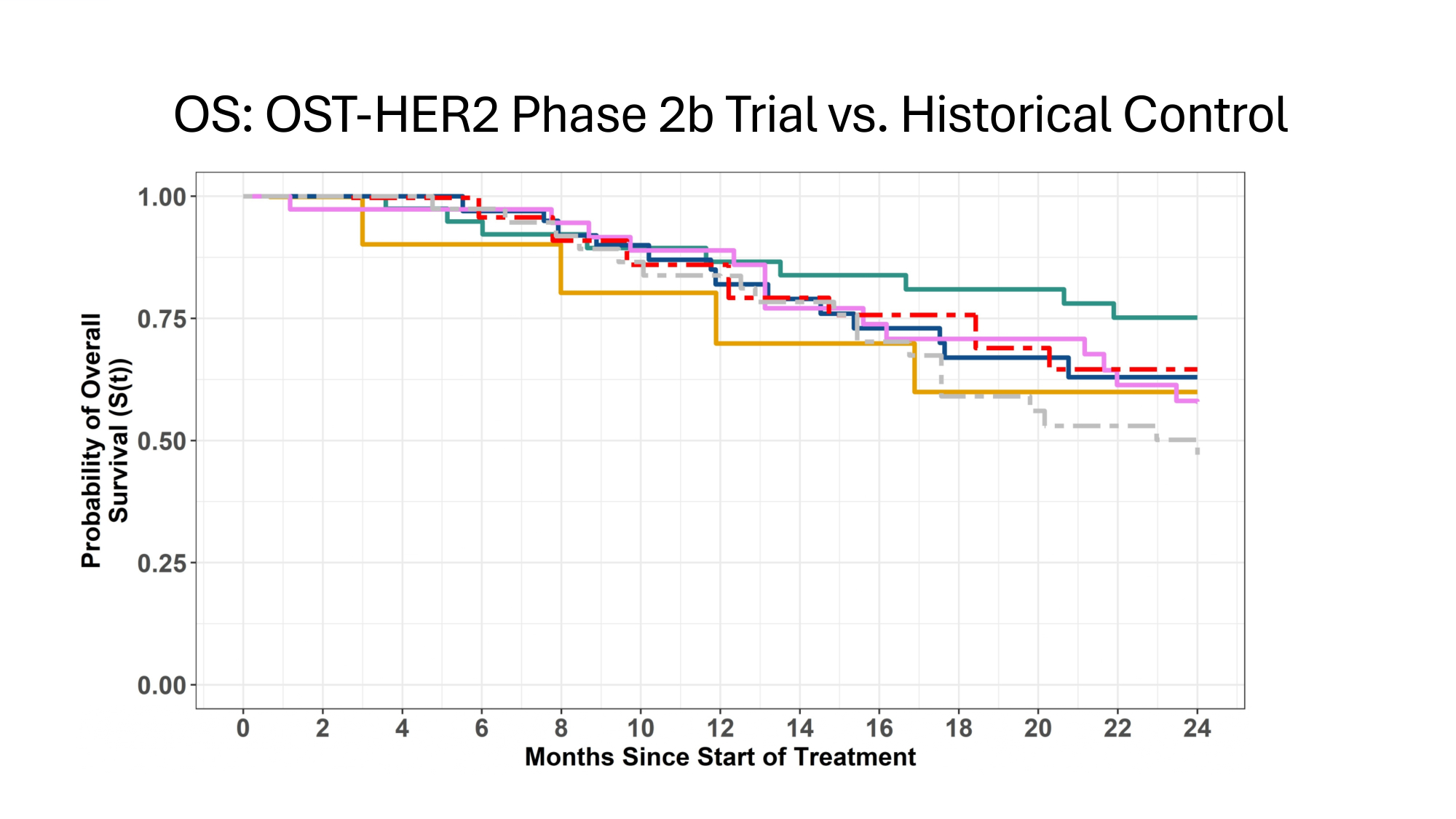
OS: OST - HER2 Phase 2b Trial vs. Historical Control
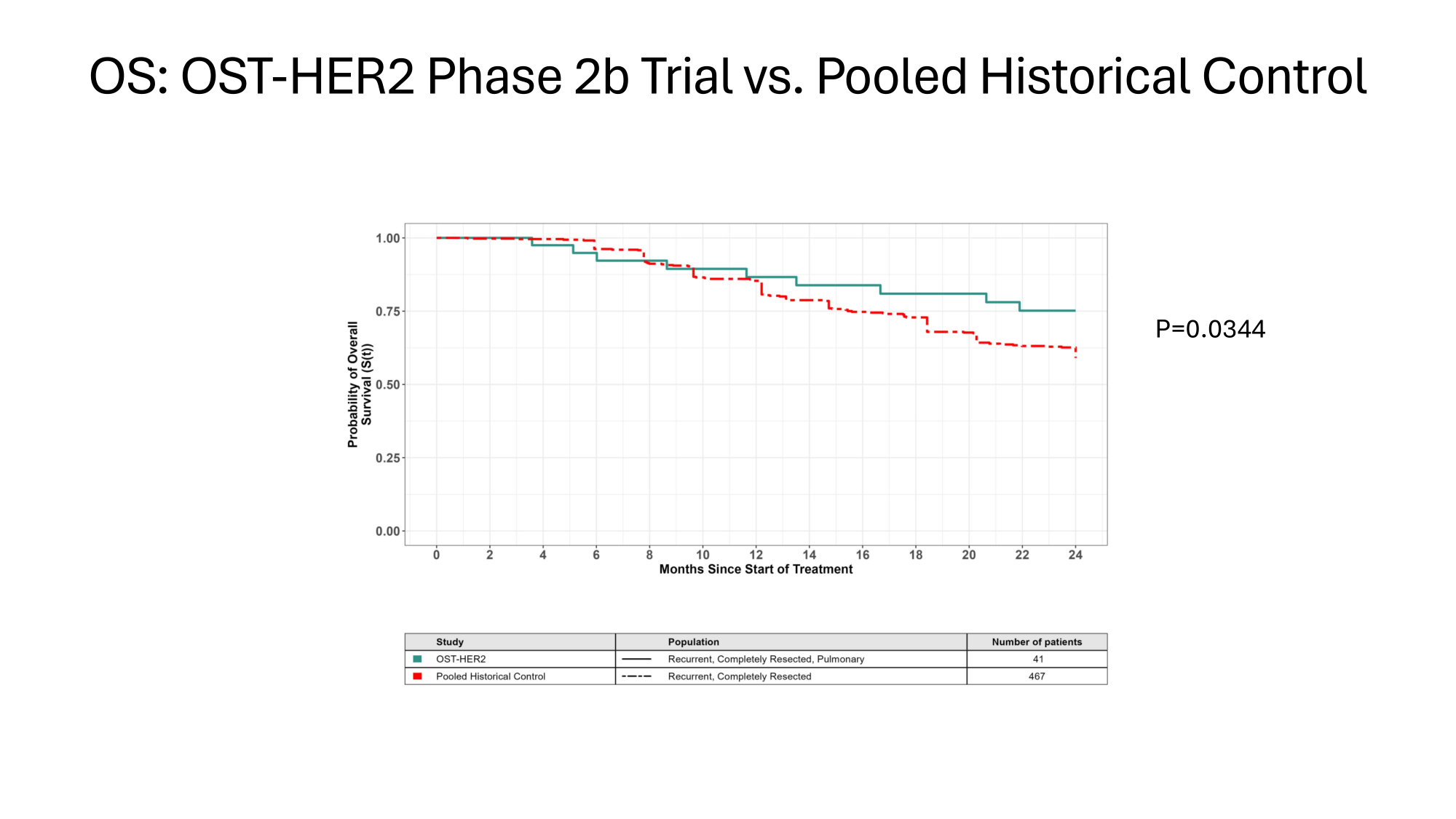
OS: OST - HER2 Phase 2b Trial vs. Pooled Historical Control P=0.0344

Isolation of Treatment Effect • Clinically significant improvement in Overall Survival C EFS I. Highly significant link between Overall Survival C Immune Response Gene Signature (IRGS) after 3 rd dose of OST31 - 164 II. IRGS includes cytotoxic effectors (NK/T cells) C T cell memory, and modulators of Tumour MicroEnvironment (M1/M2, MDSCs) III. IRGS maps directly to the Mechanism of Action of OST31 - 164 IV. Clinical trial extends findings in parallel population of spontaneous osteosarcoma in canines: multi - modal immune response links Overall Survival to Mechanism of Action

Immune Response Gene Signatures after 3 rd Dose of OST31 - 164 Link Mechanism of Action to OS in Canine s Human Osteosarcoma • Cox Proportional Hazards Model • Canine IRGS (optimized): p=10e - 8 for Overall Survival • Beneficial: Ag presentation, NK cell activation, T cell activation • Detrimental: Type 1 IFN; M2 polarization; Myeloid Derived Suppressor Cells • Human IRGS (optimized): p=10e - 4 for Overall Survival • Beneficial: Ag presentation; NK cell activation; T cell activation/differentiation; M1 polarization • Detrimental: M2 polarization; failure to generate central memory CD8+ T cells
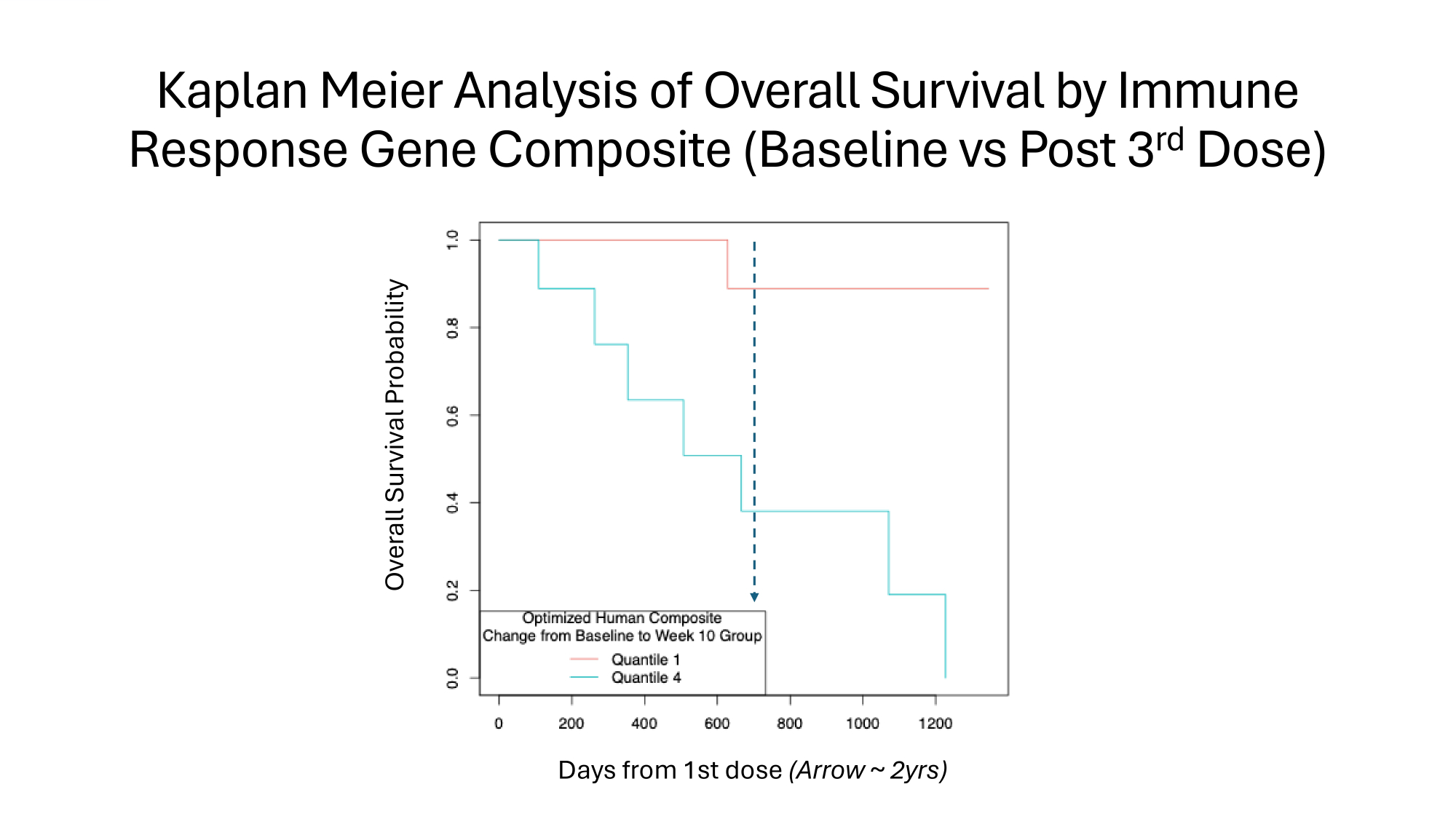
Kaplan Meier Analysis of Overall Survival by Immune Response Gene Composite (Baseline vs Post 3 rd Dose) Overall Survival Probability Days from 1st dose (Arrow ~ 2yrs)

Safety Profile III. I. Infusion related cytokine release syndrome • Frequency C severity reduced by pre - infusion regimen; no Rx discontinued • IV fluids, antihistamine, NSAID, antiemetic, H2 - receptor antagonist Cytokine release syndrome: maximum grade 2 intensity, 1 st cycle II. Infection risk • Single episode of asymptomatic bacteraemia in subject with indwelling infusion port; responsive to antibiotics C removal of port On - Target Off Tumor Effects • Her2 widely expressed at low level including skeletal C cardiac muscle • 1 episode of ?rhabdomyolysis: grade 3 ( haematuria + raised CK) in body builder • No signal in canine spontaneous osteosarcoma despite intensive monitoring

Agenda 1. Introduction – Harrison Seidner, PhD 2. Corporate Update – Paul Romness, MPH 3. Biotech Innovation Curve – Dr. Robert Langer 4. Clinical, Biomarker and Safety Data – Andrew Exley, MD, FRCP, FRCPath 5. Regulatory status by Jurisdiction (Europe, Australia, U.K. and U.S.) – David Brindley, PhD 6. Insights on developing treatments for rare pediatric cancers – Dr. Craig Eagle 7. Questions

Agenda 1. Introduction – Harrison Seidner, PhD 2. Corporate Update – Paul Romness, MPH 3. Biotech Innovation Curve – Dr. Robert Langer 4. Clinical, Biomarker and Safety Data – Andrew Exley, MD, FRCP, FRCPath 5. Regulatory status by Jurisdiction (Europe, Australia, U.K. and U.S.) – David Brindley, PhD 6. Insights on developing treatments for rare pediatric cancers – Dr. Craig Eagle 7. Questions

Agenda 1. Introduction – Harrison Seidner, PhD 2. Corporate Update – Paul Romness, MPH 3. Biotech Innovation Curve – Dr. Robert Langer 4. Clinical, Biomarker and Safety Data – Andrew Exley, MD, FRCP, FRCPath 5. Regulatory status by Jurisdiction (Europe, Australia, U.K. and U.S.) – David Brindley, PhD 6. Insights on developing treatments for rare pediatric cancers – Dr. Craig Eagle 7. Questions

Thank you for attending! OS Therapies, Inc. (NYSE American: OSTX)