DEF 14A
0001974640falseDEF 14A00019746402024-01-012024-12-310001974640ecd:YrEndFrValOfEqtyAwrdsGrntdInCvrdYrOutsdngAndUnvstdMemberecd:PeoMember2025-01-012025-12-310001974640apge:CarlDambkowskiMDMember2026-01-020001974640ecd:ChngInFrValAsOfVstngDtOfPrrYrEqtyAwrdsVstdInCvrdYrMemberecd:PeoMember2025-01-012025-12-310001974640ecd:NonPeoNeoMemberecd:EqtyAwrdsInSummryCompstnTblForAplblYrMember2025-01-012025-12-310001974640apge:MichaelHendersonMDMember2026-01-0200019746402025-01-012025-12-3100019746402023-01-012023-12-310001974640ecd:ChngInFrValOfOutsdngAndUnvstdEqtyAwrdsGrntdInPrrYrsMemberecd:PeoMember2025-01-012025-12-310001974640ecd:NonPeoNeoMemberecd:ChngInFrValOfOutsdngAndUnvstdEqtyAwrdsGrntdInPrrYrsMember2025-01-012025-12-310001974640ecd:NonPeoNeoMemberecd:YrEndFrValOfEqtyAwrdsGrntdInCvrdYrOutsdngAndUnvstdMember2025-01-012025-12-310001974640ecd:NonPeoNeoMemberecd:ChngInFrValAsOfVstngDtOfPrrYrEqtyAwrdsVstdInCvrdYrMember2025-01-012025-12-310001974640ecd:EqtyAwrdsInSummryCompstnTblForAplblYrMemberecd:PeoMember2025-01-012025-12-310001974640apge:JanePritchettHendersonMember2026-01-02utr:Ratexbrli:sharesiso4217:USD
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
SCHEDULE 14A
Proxy Statement Pursuant to Section 14(a) of the
Securities Exchange Act of 1934
(Amendment No. )
Filed by the Registrant ☒
Filed by a Party other than the Registrant ☐
Check the appropriate box:
|
|
☐ |
Preliminary Proxy Statement |
☐ |
Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) |
☒ |
Definitive Proxy Statement |
☐ |
Definitive Additional Materials |
☐ |
Soliciting Material under §240.14a-12 |
Apogee Therapeutics, Inc.
(Name of Registrant as Specified In Its Charter)
(Name of Person(s) Filing Proxy Statement if other than the Registrant)
Payment of Filing Fee (Check all boxes that apply):
|
|
☒ |
No fee required. |
☐ |
Fee paid previously with preliminary materials. |
☐ |
Fee computed on table in exhibit required by Item 25(b) per Exchange Act Rules 14a-6(i)(1) and 0-11. |

221 Crescent St., Building 17, Suite 102b, Waltham, MA 02453
NOTICE OF THE 2026 ANNUAL MEETING OF STOCKHOLDERS
TO BE HELD ON JUNE 9, 2026
To the Stockholders of Apogee Therapeutics:
Apogee Therapeutics, Inc. (the “Company”) will hold its 2026 Annual Meeting of Stockholders (the “Annual Meeting”) on Tuesday, June 9, 2026, at 11:00 a.m. Eastern Time. The Annual Meeting will be a virtual meeting conducted exclusively online via live audio webcast at www.virtualshareholdermeeting.com/APGE2026. The Annual Meeting will be held for the following purposes, as more fully described in the accompanying proxy statement (the “Proxy Statement”):
(1)To elect the three Class III director nominees named in the Proxy Statement to serve until the 2029 Annual Meeting of Stockholders and until their successors are duly elected and qualified;
(2)To ratify the appointment of Ernst & Young LLP as the Company’s independent registered public accounting firm for the year ending December 31, 2026;
(3)To approve on a non-binding, advisory basis, the compensation of our named executive officers as disclosed in the Proxy Statement; and
(4)To transact any other matters that may properly come before the Annual Meeting or any adjournments or postponements thereof.
The Board of Directors has fixed April 15, 2026 as the Record Date. Only stockholders of record at the close of business on that date will be entitled to notice of, and to vote at, the Annual Meeting or any adjournment or postponement thereof.
Instructions for accessing the virtual Annual Meeting are provided in the Proxy Statement. Unless otherwise announced differently at the meeting or on the meeting website, in the event of a technical malfunction or other situation that the meeting chair determines may affect the ability of the Annual Meeting to satisfy the requirements for a meeting of stockholders to be held by means of remote communication under the Delaware General Corporation Law, or that otherwise makes it advisable to adjourn the Annual Meeting, the meeting chair or secretary will convene the meeting at 12:00 p.m. Eastern Time on the date specified above and at the Company’s address specified above solely for the purpose of adjourning the meeting to reconvene at a date, time and physical or virtual location announced by the meeting chair or secretary. Under either of the foregoing circumstances, we will post information regarding the announcement on the Investors page of the Company’s website at https://investors.apogeetherapeutics.com/.
By Order of the Board of Directors,
|
/s/ Matt Batters |
|
Matt Batters |
Corporate Secretary |
|
Waltham, Massachusetts |
April 24, 2026 |
Whether or not you expect to participate in the virtual Annual Meeting, please vote as promptly as possible in order to ensure your representation at the Annual Meeting. You may vote online or, if you requested printed copies of the proxy materials, by telephone or by using the proxy card or voting instruction form provided with the printed proxy materials.
LEGAL MATTERS
Reorganization. Apogee Therapeutics, LLC was formed as a limited liability company under the laws of the State of Delaware in February 2022. Apogee Therapeutics, Inc. was incorporated in June 2023, in connection with our initial public offering (“IPO”) to serve as a holding company that would wholly own the assets of Apogee Therapeutics, LLC. Prior to July 13, 2023, our business was conducted by Apogee Therapeutics, LLC and its subsidiary, Apogee Biologics, Inc. In July 2023, in connection with our IPO, we completed a series of transactions which are referred to, collectively, as the “Reorganization,” and pursuant to which Apogee Therapeutics, Inc., became the parent and holding company that wholly owns the assets of Apogee Therapeutics, LLC, including stock of its subsidiary, Apogee Biologics, Inc. Effective December 31, 2024, Apogee Biologics, Inc. merged with and into Apogee Therapeutics, Inc. with Apogee Therapeutics, Inc. surviving the merger. As used in this Proxy Statement, unless the context otherwise requires, references to “we,” “us,” “our,” the “Company,” “Apogee” and similar references refer: (1) following the consummation of our Reorganization, to Apogee Therapeutics, Inc. and our subsidiary, and (2) prior to the completion of our Reorganization, to Apogee Therapeutics, LLC and its subsidiary.
Important Notice Regarding the Availability of Proxy Materials for the 2026 Annual Meeting of Stockholders to Be Held on June 9, 2026. The Proxy Statement and Annual Report for the year ended December 31, 2025 are available at www.proxyvote.com.
Forward-Looking Statements. The Proxy Statement may contain “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, which statements are subject to substantial risks and uncertainties and are based on estimates and assumptions. All statements other than statements of historical fact included in the Proxy Statement, including statements about the Company’s Board of Directors, corporate governance practices and executive compensation program and equity compensation utilization, are forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “might,” “will,” “objective,” “intend,” “should,” “could,” “can,” “would,” “expect,” “believe,” “design,” “estimate,” “predict,” “potential,” “plan” or the negative of these terms, and similar expressions intended to identify forward-looking statements. These statements involve known and unknown risks, uncertainties and other factors that could cause our actual results or outcomes to differ materially from the forward-looking statements expressed or implied in the Proxy Statement. Such risks, uncertainties and other factors include those risks described in “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in the Company’s most recent Annual Report on Form 10-K filed with the U.S. Securities and Exchange Commission (“SEC”) and other subsequent documents we have filed with the SEC. The Company expressly disclaims any obligation to update or alter any statements whether as a result of new information, future events or otherwise, except as required by law.
Website References. Website references throughout this document are inactive textual references and provided for convenience only, and the content on the referenced websites is not incorporated herein by reference and does not constitute a part of the Proxy Statement.
Use of Trademarks. Apogee Therapeutics is the registered trademark of Apogee Therapeutics, Inc. Other names and brands may be claimed as the property of others.

221 Crescent St., Building 17, Suite 102b, Waltham, MA 02453
PROXY STATEMENT
FOR THE 2026 ANNUAL MEETING OF STOCKHOLDERS
QUESTIONS AND ANSWERS ABOUT THE PROXY MATERIALS AND VOTING
What Is the Purpose of These Proxy Materials?
We are making these proxy materials available to you in connection with the solicitation of proxies by the Board of Directors (the “Board”) of Apogee Therapeutics, Inc. (“we,” “us,” “our” or the “Company”) for use at the 2026 Annual Meeting of Stockholders (the “Annual Meeting”) to be held virtually on June 9, 2026 at 11:00 a.m. Eastern Time, or at any other time following adjournment or postponement thereof.
You are invited to participate in the Annual Meeting and to vote on the proposals described in this proxy statement (the “Proxy Statement”). The proxy materials are first being made available to our stockholders on or about April 24, 2026.
Why Did I Receive a Notice of Internet Availability?
Pursuant to SEC rules, we are furnishing the proxy materials to our stockholders primarily via the Internet instead of mailing printed copies. This process allows us to expedite our stockholders’ receipt of proxy materials, lower the costs of printing and mailing the proxy materials and reduce the environmental impact of our Annual Meeting. If you received a Notice of Internet Availability of Proxy Materials (the “Notice”), you will not receive a printed copy of the proxy materials unless you request one. The Notice provides instructions on how to access the proxy materials for the Annual Meeting via the Internet, how to request a printed set of proxy materials and how to vote your shares.
Why Are We Holding a Virtual Annual Meeting?
We have adopted a virtual meeting format for the Annual Meeting to provide a consistent experience to all stockholders regardless of geographic location. We believe this expands stockholder access, improves communications and lowers our costs while reducing the environmental impact of the meeting. In structuring our virtual Annual Meeting, our goal is to enhance rather than constrain stockholder participation in the meeting, and we have designed the meeting to provide stockholders with the same rights and opportunities to participate as they would have at an in-person meeting.
Who Can Vote?
Only stockholders of record at the close of business on April 15, 2026 (the “Record Date”) are entitled to notice of the Annual Meeting and to vote on the proposals described in this Proxy Statement. At the close of business on the Record Date, 61,853,254 shares of our voting common stock were issued and outstanding. Shares of our non-voting common stock generally do not have voting rights and holders of our non-voting common stock are therefore not entitled to vote at the Annual Meeting. Unless otherwise noted, all references to our “common stock” herein refers to our voting common stock.
Why Does the Company have Non-Voting Common Stock?
We created a class of non-voting common stock solely to facilitate certain of our pre-IPO holders of Series A preferred units and Series B preferred units of Apogee Therapeutics, LLC being able to beneficially own our common stock without exceeding certain beneficial ownership thresholds. Specifically, as part of the Reorganization, pursuant to a contribution and exchange agreement, effective July 13, 2023, holders of Series A preferred units and Series B preferred units of Apogee Therapeutics, LLC contributed their Series A preferred units and Series B preferred stock units to Apogee Therapeutics, Inc. in exchange for shares of non-voting common stock of Apogee Therapeutics, Inc. Holders of our non-voting common stock shall have the right to convert each share of our non-voting common stock into one share of common stock at such holder’s election, provided that as a result of such conversion, such holder, together with its affiliates and any members of a Schedule 13(d) group with such holder, would not beneficially own in excess of 9.99% of our common stock immediately prior to and following such conversion (the “Beneficial Ownership Limitation”), unless otherwise as expressly provided for in our amended and restated certificate of incorporation (“Certificate of Incorporation”). However, the Beneficial Ownership Limitation may be increased or decreased to any other percentage (not to exceed 19.99%) designated by such holder of non-voting common stock upon 61 days’ notice to us.
What Is the Difference between Holding Shares as a Registered Stockholder and as a Beneficial Owner?
Registered Stockholder: Shares Registered in Your Name
If your shares of common stock are registered directly in your name with our transfer agent, Computershare Trust Company, N.A., you are considered to be, with respect to those shares of common stock, the registered stockholder, and these proxy materials are being sent directly to you by us.
Beneficial Owner: Shares Registered in the Name of a Broker, Fiduciary or Custodian
If your shares of common stock are held by a broker, fiduciary or custodian, you are considered the beneficial owner of shares of common stock held in “street name,” and these proxy materials are being forwarded to you from that broker, fiduciary or custodian.
How Can I Participate in the Virtual Annual Meeting?
Stockholders of record as of the close of business on the Record Date are entitled to participate in and vote at the Annual Meeting. To participate in the Annual Meeting, including to vote and ask questions, stockholders of record should go to the meeting website at www.virtualshareholdermeeting.com/APGE2026, enter the 16-digit control number found on your proxy card or Notice, and follow the instructions on the website. If your shares are held in street name and your voting instruction form or Notice indicates that you may vote those shares through www.proxyvote.com, then you may access, participate in and vote at the Annual Meeting with the 16-digit access code indicated on that voting instruction form or Notice. Otherwise, stockholders who hold their shares in street name should contact their bank, broker or other nominee (preferably at least five days before the Annual Meeting) and obtain a “legal proxy” in order to be able to attend, participate in or vote at the Annual Meeting.
We will endeavor to answer as many stockholder-submitted questions as time permits that comply with the Annual Meeting rules of conduct. We reserve the right to edit profanity or other inappropriate language and to exclude questions regarding topics that are not pertinent to meeting matters or Company business. If we receive substantially similar questions, we may group such questions together and provide a single response to avoid repetition.
The meeting webcast will begin promptly at 11:00 a.m. Eastern Time. Online check-in will begin approximately 15 minutes before then, and we encourage you to allow ample time for check-in procedures. If you experience technical difficulties during the check-in process or during the meeting, please call the number listed on the meeting website for technical support. Additional information regarding the rules and procedures for participating in the Annual Meeting will be set forth in our meeting rules of conduct, which stockholders can view during the meeting at the meeting website.
What Am I Voting on?
The proposals to be voted on at the Annual Meeting are as follows:
(1)Election of three Class III director nominees to serve until the 2029 Annual Meeting of Stockholders (“Proposal 1”);
(2)Ratification of the appointment of Ernst & Young LLP (“EY”) as the Company’s independent auditor for 2026 (“Proposal 2”); and
(3)Approval, on a non-binding, advisory basis, of the compensation of our named executive officers (each, an “NEO”) as disclosed in this Proxy Statement (“Proposal 3”).
How Does the Board Recommend That I Vote?
The Board recommends that you vote your shares “FOR” each director nominee in Proposal 1, “FOR” Proposal 2 and “FOR” for Proposal 3.
What If Another Matter Is Properly Brought before the Annual Meeting?
As of the date of filing this Proxy Statement, the Board knows of no other matters that will be presented for consideration at the Annual Meeting. If any other matters are properly brought before the Annual Meeting, it is the intention of the persons named as proxies in the proxy card to vote on such matters in accordance with their best judgment.
How Many Votes Do I Have?
Each share of voting common stock is entitled to one vote on each proposal to be voted on at the Annual Meeting.
What Does It Mean If I Receive More Than One Set of Proxy Materials?
If you receive more than one set of proxy materials, your shares may be registered in more than one name or held in different accounts. Please cast your vote with respect to each set of proxy materials that you receive to ensure that all of your shares are voted.
How Do I Vote?
Even if you plan to attend the Annual Meeting, we recommend that you also submit your vote as early as possible in advance so that your vote will be counted if you later decide not to, or are unable to, virtually attend the Annual Meeting.
Registered Stockholder: Shares Registered in Your Name
If you are the registered stockholder, you may vote your shares online during the virtual Annual Meeting (see “How Can I Participate in the Virtual Annual Meeting?” above) or by proxy in advance of the Annual Meeting by Internet (at www.proxyvote.com) or, if you requested paper copies of the proxy materials, by completing and mailing a proxy card or by telephone (at 800-690-6903).
Beneficial Owner: Shares Registered in the Name of a Broker, Fiduciary or Custodian
If you are the beneficial owner, you may vote your shares online during the virtual Annual Meeting (see “How Can I Participate in the Virtual Annual Meeting?” above) or you may direct your broker, fiduciary or custodian how to vote in advance of the Annual Meeting by following the instructions they provide.
What Happens If I Do Not Vote?
Registered Stockholder: Shares Registered in Your Name
If you are the registered stockholder and do not vote in one of the ways described above, your shares will not be voted at the Annual Meeting and will not be counted toward the quorum requirement.
Beneficial Owner: Shares Registered in the Name of a Broker, Fiduciary or Custodian
If you are the beneficial owner and do not direct your broker, fiduciary or custodian on how to vote your shares, your broker, fiduciary or custodian will only be able to vote your shares with respect to proposals considered to be “routine.” Your broker, fiduciary or custodian is not entitled to vote your shares with respect to “non-routine” proposals, which we refer to as a “broker non-vote.” Whether a proposal is considered routine or non-routine is subject to stock exchange rules and final determination by the stock exchange. Even with respect to routine matters, some brokers are choosing not to exercise discretionary voting authority. As a result, we urge you to direct your broker, fiduciary or custodian on how to vote your shares on all proposals to ensure that your vote is counted.
What If I Sign and Return a Proxy Card or Otherwise Vote but Do Not Indicate Specific Choices?
Registered Stockholder: Shares Registered in Your Name
The shares represented by each signed and returned proxy will be voted at the Annual Meeting by the persons named as proxies in the proxy card in accordance with the instructions indicated on the proxy card. However, if you are the registered stockholder and sign and return your proxy card without giving specific instructions, the persons named as proxies in the proxy card will vote your shares in accordance with the recommendations of the Board. Your shares will be counted toward the quorum requirement.
Beneficial Owner: Shares Registered in the Name of a Broker, Fiduciary or Custodian
If you are the beneficial owner and do not direct your broker, fiduciary or custodian on how to vote your shares, your broker, fiduciary or custodian will only be able to vote your shares with respect to proposals considered to be “routine.” Your broker, fiduciary or custodian is not entitled to vote your shares with respect to “non-routine” proposals, resulting in a broker non-vote with respect to such proposals.
Can I Change My Vote after I Submit My Proxy?
Registered Stockholder: Shares Registered in Your Name
If you are the registered stockholder, you may revoke your proxy at any time before the final vote at the Annual Meeting in any one of the following ways:
(1)You may complete and submit a new proxy card, but it must bear a later date than the original proxy card;
(2)You may submit new proxy instructions via telephone or the Internet;
(3)You may send a timely written notice that you are revoking your proxy to our Corporate Secretary at the address set forth on the first page of this Proxy Statement; or
(4)You may vote by attending the Annual Meeting virtually. However, your virtual attendance at the Annual Meeting will not, by itself, revoke your proxy.
Your last submitted vote is the one that will be counted.
Beneficial Owner: Shares Registered in the Name of a Broker, Fiduciary or Custodian
If you are the beneficial owner, you must follow the instructions you receive from your broker, fiduciary or custodian with respect to changing your vote.
What Is the Quorum Requirement?
The holders of a majority of the shares of common stock outstanding and entitled to vote at the Annual Meeting, either virtually or represented by proxy, must be present at the Annual Meeting to constitute a quorum. A quorum is required to transact business at the Annual Meeting.
Your shares will be counted toward the quorum only if you submit a valid proxy (or a valid proxy is submitted on your behalf by your broker, fiduciary or custodian) or if you attend the Annual Meeting virtually and vote. Abstentions and broker non-votes, if any, will be counted toward the quorum requirement. If there is no quorum, the chair of the Annual Meeting or the holders of a majority of shares of common stock present at the Annual Meeting, either virtually or represented by proxy, may adjourn the Annual Meeting to another time or date.
How Many Votes Are Required to Approve Each Proposal and How Are Votes Counted?
Votes will be counted by Broadridge Financial Solutions, the Inspector of Elections appointed for the Annual Meeting.
Proposal 1: Election of Directors
A nominee will be elected as a director at the Annual Meeting if the nominee receives a plurality of the votes cast “FOR” his or her election. “Plurality” means that the individuals who receive the highest number of votes cast “FOR” are elected as directors. Broker non-votes, if any, and votes that are withheld will not be counted as votes cast on the matter and will have no effect on the outcome of the election. Stockholders do not have cumulative voting rights for the election of directors.
Proposal 2: Ratification of Independent Auditor Appointment
The affirmative vote of the holders of at least a majority of shares of common stock present or represented at the Annual Meeting and entitled to vote on the matter is required to approve this proposal. Abstentions will have the same effect as a vote “AGAINST” the matter. Broker non-votes, if any, will have no effect on the outcome of the matter.
Proposal 3: Advisory Vote on our NEOs’ Compensation
The advisory vote on the compensation of our NEOs requires the affirmative vote of the holders of at least a majority of shares of common stock present or represented at the Annual Meeting and entitled to vote on the matter. Abstentions will have the same effect as a vote “AGAINST” the matter. Broker non-votes, if any, will have no effect on the outcome of the matter.
Who Is Paying for This Proxy Solicitation?
We will pay the costs associated with the solicitation of proxies, including the preparation, assembly, printing and mailing of the proxy materials. We may also reimburse brokers, fiduciaries or custodians for the cost of forwarding proxy materials to beneficial owners of shares of common stock held in “street name.”
Our employees, officers and directors may solicit proxies in person or via telephone or the Internet. We will not pay additional compensation for any of these services.
How Can I Find out the Voting Results?
We expect to announce preliminary voting results at the Annual Meeting. Final voting results will be published in a Current Report on Form 8-K to be filed with the SEC within four business days after the Annual Meeting.
PROPOSAL 1: ELECTION OF DIRECTORS
In accordance with our Amended and Restated Certificate of Incorporation (the “Certificate of Incorporation”) and Amended and Restated Bylaws (the “Bylaws”), the Board has fixed the number of directors constituting the Board at seven, effective as of May 11, 2026. At the Annual Meeting, the stockholders will vote to elect the three Class III director nominees named in this Proxy Statement to serve until the 2029 Annual Meeting of Stockholders and until their successors are duly elected and qualified or until their earlier resignation or removal. Our Board has nominated Mark C. McKenna, Jennifer Fox and William (BJ) Jones, Jr. for election to our Board. Mr. McKenna was appointed to our Board in August 2023, and Ms. Fox and Mr. Jones have served on our Board since prior to our IPO in July 2023. Mr. McKenna was recommended to the Nominating and Corporate Governance Committee (the “Nominating Committee”) by a third-party search firm.
Our director nominees have indicated that they are willing and able to serve as directors. However, if any of them becomes unable or, for good cause, unwilling to serve, proxies may be voted for the election of such other person as shall be designated by our Board, or the Board may decrease the size of the Board.
Information Regarding Director Nominees and Continuing Directors
Our Board is divided into three classes, with members of each class holding office for staggered three-year terms. On April 21, 2026, Class I directors, Andrew Gottesdiener, M.D. and Peter Harwin, each notified the Board of his intention to resign from the Board effective May 11, 2026. The resignations of Dr. Gottesdiener and Mr. Harwin are not the result of any disagreement with the Company on any matter relating to the Company’s operations, policies or practices. The size of the Board will be reduced from nine directors to seven directors effective May 11, 2026.
There are currently three Class III directors, who are up for election at this meeting for a term expiring at the 2029 Annual Meeting of Stockholders; effective as of May 11, 2026, one Class I director, whose term expires at the 2027 Annual Meeting of Stockholders; and currently three Class II directors, whose terms expire at the 2028 Annual Meeting of Stockholders.
Biographical and other information regarding our director nominees and directors continuing in office, including the primary skills and experiences considered by our Nominating Committee in determining to recommend them as nominees, is set forth below. The Board committee composition set forth below reflects updated committee membership effective as of May 11, 2026.
|
|
|
|
|
|
|
Name |
|
Class |
|
Age
(as of April 24) |
|
Position |
Michael Henderson, M.D. |
|
I |
|
36 |
|
Chief Executive Officer and Director |
Mark C. McKenna(1)(2) |
|
III |
|
46 |
|
Independent Chair of the Board |
Lisa Bollinger, M.D.(3) |
|
II |
|
61 |
|
Independent Director |
Jennifer Fox (1) (2) |
|
III |
|
54 |
|
Independent Director |
William (BJ) Jones, Jr. (2)(3) |
|
III |
|
62 |
|
Independent Director |
Tomas Kiselak |
|
II |
|
39 |
|
Independent Director |
Nimish Shah(1) |
|
II |
|
48 |
|
Independent Director |
(1)Member of the Audit Committee
(2)Member of the Compensation Committee
(3)Member of the Nominating Committee
Class III Director Nominees
Mark C. McKenna. Mr. McKenna has served as a member of our Board since August 2023. Mr. McKenna has served as Chief Executive Officer and chair of the board of directors of Mirador Therapeutics, Inc., a biotechnology company, since March 2024, and as a venture partner at ARCH Venture Partners, an investment firm, since February 2024. Prior to Mirador, Mr. McKenna served as the President and Chief Executive Officer and as a member of the board of directors of Prometheus Biosciences, Inc., a clinical stage biotechnology company, from September 2019 to June 2023, when Prometheus was acquired by Merck & Co., Inc., and as chairman of the board of Prometheus from August 2021 to June 2023. Prior to Prometheus, he served as President of Salix Pharmaceuticals, Inc., a pharmaceutical company and wholly-owned subsidiary of Bausch Health Companies, Inc., from March 2016 through August 2019. Prior to Salix, Mr. McKenna spent more than a decade in various roles with Bausch + Lomb, also a division of Bausch Health Companies, Inc., most recently as Senior Vice President and General Manager of its U.S. Vision Care business. Before joining Bausch + Lomb, he held several positions with Johnson & Johnson. Mr. McKenna has served on the board of directors of Spyre Therapeutics, Inc. (Nasdaq: SYRE), a biotechnology company, since February 2024 and NewAmsterdam Pharma Company N.V. (Nasdaq: NAMS), a clinical biopharmaceutical company, since July 2024. Mr. McKenna received a B.S. in Marketing from Arizona State University and an M.B.A. from Azusa Pacific University.
We believe Mr. McKenna is qualified to serve on our Board because of his extensive business leadership experience as a CEO of various biopharmaceutical companies, including of companies in the inflammatory and immunology space, as well as his experience serving as a director of other companies in the biopharmaceutical industry.
Jennifer Fox. Ms. Fox has served as a member of our Board since June 2023 and as a member of the board of managers of Apogee Therapeutics, LLC from May 2023 to July 2023. Ms. Fox has served as the Chief Business Officer and Chief Financial Officer of Zenas BioPharma, Inc., (Nasdaq: ZBIO), a biopharmaceutical company, since December 2023. Prior to joining Zenas BioPharma, Ms. Fox served as Chief Financial Officer of Nuvation Bio Inc. (NYSE: NUVB), a biopharmaceutical company, from October 2020 to November 2023. Prior to Nuvation Bio, Ms. Fox served as Managing Director, Co-Head of North America Healthcare Corporate and Investment Banking Group at Citigroup (NYSE: C), a global investment bank, from June 2015 to October 2020. From February 2006 to June 2015, Ms. Fox served as Managing Director at Deutsche Bank (NYSE: DB), a global investment bank, and most recently, as Co-Head of Life Sciences Investment Banking Group. Ms. Fox has served on the board of directors of ProKidney Corp. (Nasdaq: PROK), a biotechnology company, since July 2022. Ms. Fox received B.S. degrees in finance and marketing from Manhattan College.
We believe Ms. Fox is qualified to serve on our Board because of her finance expertise, leadership experience as an officer of other biopharmaceutical companies and extensive experience in the healthcare investment banking industry, including as a lead advisor to life sciences companies on financing and strategic transactions.
William (BJ) Jones, Jr. Mr. Jones has served as a member of our Board since June 2023 and as a member of the board of managers of Apogee Therapeutics, LLC from May 2023 to July 2023. Mr. Jones has served as Chief Commercial Officer at NewAmsterdam Pharma (Nasdaq: NAMS), a clinical stage biopharmaceutical company, since August 2023. Mr. Jones previously served as Chief Commercial Officer, Migraine and Common Diseases, of Biohaven Pharmaceuticals Holding Company Ltd., a biopharmaceutical company and subsidiary of Pfizer Inc. (NYSE: PFE), where he was responsible for building the company’s commercial capability and launching its first FDA-approved product (Nurtec ODT), from April 2019 to December 2022. Prior to Biohaven Pharmaceuticals, Mr. Jones served as Vice President, Head of Sales and Commercial Operations for the general medicine business unit of Takeda Pharmaceutical Company Limited (NYSE: TAK), a pharmaceutical company, from January 2016 to March 2019. Mr. Jones has served on the board of directors of Annexon Biosciences (Nasdaq: ANNX), a clinical stage biopharmaceutical company since January 2025 and served on the board of directors of Akili, Inc. (Nasdaq: AKLI), a digital medicine company, from August 2022 to July 2024. Mr. Jones received a B.S. in human factors engineering from the U.S. Air Force Academy, an M.S. in industrial engineering from Texas A&M University and an M.B.A. from the Stanford University Graduate School of Business.
We believe Mr. Jones is qualified to serve on our Board because of his experience in drug development and commercial strategy in the pharmaceutical industry and leadership experience as an officer and director of other publicly traded biopharmaceutical companies.
Class I Director Continuing in Office
Michael Henderson, M.D. Dr. Henderson has served as a member of our Board since June 2023, as a member of the board of managers of Apogee Therapeutics, LLC from 2022 to July 2023 and as our Chief Executive Officer (“CEO”) since September 2022. Dr. Henderson is an experienced biotechnology executive with expertise in business leadership, drug development, and commercial strategy. He has overseen the creation of multiple companies, launched a significant number of drug development programs, and led teams to two FDA approvals, to date. Prior to joining Apogee, Dr. Henderson served as Chief Business Officer of BridgeBio Pharma, Inc. (Nasdaq: BBIO), a commercial-stage biopharmaceutical company, from January 2020 to September 2022, where he was responsible for furthering the overarching strategy of BridgeBio, identifying and investing in new technologies and running business development and operations. Prior to holding that position, he spent two years serving as BridgeBio’s Senior Vice President, Asset Acquisition, Strategy and Operations, where he was responsible for business development, strategy and operations. Dr. Henderson joined BridgeBio as Vice President of Asset Acquisition, Strategy and Operations in April 2016. Dr. Henderson also served as the Chief Executive Officer of a number of BridgeBio’s subsidiaries. Prior to BridgeBio, Dr. Henderson worked at McKinsey & Company, a global management consulting firm, from January 2015 to April 2016 and prior to that, he co-founded PellePharm, Inc., a biotechnology company, in August 2011. Dr. Henderson has served on the board of directors of Spyre Therapeutics (Nasdaq: SYRE), a biotechnology company, since June 2023 and served on the board of directors of ARYA Sciences Acquisition Corp IV (Nasdaq: ARYD), a special purpose acquisition company focused on the healthcare industry, from February 2021 to July 2024. Dr. Henderson received a B.A. in global health from Harvard University and an M.D. from Stanford University.
We believe Dr. Henderson is qualified to serve on our Board because of his experience in business leadership, drug development, and commercial strategy in the life sciences area, including his leadership in launching multiple drug development programs and obtaining FDA approvals.
Class II Directors Continuing in Office
Lisa Bollinger, M.D. Dr. Bollinger has served as a member of our Board since May 2024. Dr. Bollinger has served as the Chief Medical Officer of Polaryx Therapeutics, Inc. (Nasdaq: PLYX), a clinical-stage biotechnology company, since October 2025. Dr. Bollinger is a pediatric trained physician with over 30 years of experience in drug development, including senior level positions at the U.S. Food and Drug Administration (the “FDA”) and multinational biotechnology and pharmaceutical companies in both regulatory affairs and pharmacovigilance. She most recently served as the Vice President, Regulatory Affairs, Global Regulatory Affairs and Clinical Safety of Merck & Co., Inc. (NYSE: MRK), a multinational biopharmaceutical company, from March 2021 to May 2024. Before joining Merck, Dr. Bollinger held various positions of increasing responsibility at Amgen Inc. (Nasdaq: AMGN), a multinational biotechnology company, from September 2012 to March 2021, most recently serving as its Vice President of Global Patient Safety & Pediatrics, and Labeling Global Regulatory Affairs & Safety, Research and Development, from September 2018 to March 2021. Prior to Amgen, Dr. Bollinger held various positions of increasing responsibility at the FDA from September 1998 to July 2012, most recently serving as Associate Director, Office of New Drugs, Center for Drug Evaluation and Research. Dr. Bollinger received a B.S. in Physiology from the University of California, Davis, an M.D. from Uniformed Services University of the Health Sciences F. Edward Hebert School of Medicine and completed her residency in pediatrics at the University of California Davis Medical Center.
We believe Dr. Bollinger is qualified to serve on our Board because of her vast drug development and pharmacovigilance experience, including her regulatory experience at the FDA and her leadership experience as an officer at a number of multinational biotechnology and pharmaceutical companies.
Tomas Kiselak. Mr. Kiselak has served as a member of our Board since June 2023 and as a member of the board of managers of Apogee Therapeutics, LLC from 2022 to July 2023. Mr. Kiselak is a Managing Member at Fairmount Funds Management LLC, a healthcare investment firm he co-founded in April 2016. Prior to Fairmount, Mr. Kiselak was a managing director at RA Capital Management, LLC, a healthcare and life science investment firm.
Mr. Kiselak currently serves as the chairman of the board of directors of Viridian Therapeutics, Inc. (Nasdaq: VRDN) and as a director for Jade Biosciences, Inc. (Nasdaq: JBIO) since April 2025, Spyre Therapeutics, Inc. (Nasdaq: SYRE), Zenas BioPharma, Inc. (Nasdaq: ZBIO), and serves as chair of Korsana Biosciences and as a director of several other private companies. Mr. Kiselak previously served as a director of Dianthus Therapeutics, Inc. (Nasdaq: DNTH) from September 2023 until March 2025. Mr. Kiselak received a B.S. in neuroscience and economics from Amherst College.
We believe Mr. Kiselak is qualified to serve on our Board because of his extensive leadership, executive and managerial experience in the biotechnology industry, including his experience advising biotechnology companies and as a manager of funds specializing in the life sciences area.
Nimish Shah. Mr. Shah co-founded the Company and has served as a member of our Board since June 2023, and as a member of the board of managers of Apogee Therapeutics, LLC from 2022 to July 2023. He is a partner at Venrock Healthcare Capital Partners, an investment firm, where he primarily focuses on Venrock’s public and crossover biotech funds. Mr. Shah originally joined Venrock in 2013 and has been investing in public and private healthcare companies since 2010. He previously served as a director for Instil Bio, Inc. (Nasdaq: TIL) until December 2021 and as a board observer for LianBio (Nasdaq: LIAN), Biohaven Ltd. (NYSE: BHVN), Viridian Therapeutics, Inc. (Nasdaq: VRDN) and Dianthus Therapeutics, Inc. (Nasdaq: DNTH). Mr. Shah co-founded Korsana Biosciences, a pre-clinical biotechnology company, and has served as a member of its board since November 2024. Mr. Shah received a B.S. in pharmacy from Rutgers College of Pharmacy, an M.P.H. from the Mailman School of Public Health at Columbia University, and an M.B.A. from Columbia Business School. He is also a member of the Columbia Business School Healthcare and Pharmaceutical Management Advisory Board.
We believe Mr. Shah is qualified to serve on our Board because of his extensive investment management and finance experience in the healthcare sector, as well as his experience serving on the boards of directors of numerous other biotechnology companies.
Board Recommendation
The Board recommends a vote “FOR” the election of each of the Class III director nominees set forth above.
PROPOSAL 2: RATIFICATION OF INDEPENDENT AUDITOR APPOINTMENT
Our Audit Committee has appointed EY as the Company’s independent registered public accounting firm for the year ending December 31, 2026. In this Proposal 2, we are asking stockholders to vote to ratify this appointment. Representatives of EY are expected to be present at the Annual Meeting. They will have the opportunity to make a statement, if they desire to do so, and are expected to be available to respond to appropriate questions from stockholders.
Stockholder ratification of the appointment of EY as the Company’s independent auditor is not required by law or our Bylaws. However, we are seeking stockholder ratification as a matter of good corporate governance. If our stockholders fail to ratify the appointment, the committee will reconsider its appointment. Even if the appointment is ratified, the committee, in its discretion, may direct the appointment of a different independent auditor at any time during the year if it determines that such a change would be in the best interests of the Company and our stockholders.
EY has served as our independent auditor since 2023. The following table summarizes the audit fees billed and expected to be billed by EY for the indicated fiscal years and the fees billed by EY for all other services rendered during the indicated fiscal years. All services associated with such fees and provided in 2025 were pre-approved by our Audit Committee in accordance with the “Pre-Approval Policies and Procedures” described below.
|
|
|
|
|
|
|
|
|
|
|
Year Ended
December 31, |
|
Fee Category |
|
2025 |
|
|
2024 |
|
Audit Fees(1) |
|
$ |
1,190,000 |
|
|
$ |
1,257,500 |
|
Audit-Related Fees(2) |
|
|
— |
|
|
|
— |
|
Tax Fees(3) |
|
|
— |
|
|
|
— |
|
All Other Fees(4) |
|
|
— |
|
|
|
— |
|
Total Fees |
|
$ |
1,190,000 |
|
|
$ |
1,257,500 |
|
(1)Consists of aggregate fees for professional services provided in connection with the annual audit of our consolidated financial statements, the review of our quarterly condensed consolidated financial statements and comfort letters, consents and review of documents filed with the SEC. Fees for fiscal year 2025 and 2024 include services provided for the audit of the effectiveness of our internal control over financial reporting.
(2)Consists of fees for assurance and related services associated with consultations on matters directly related to the audit.
(3)Consists of fees for tax compliance, advice and tax services.
(4)Consists of fees for all other services.
Pre-Approval Policies and Procedures
Our Audit Committee has adopted procedures requiring the pre-approval of all audit and non-audit services performed by our independent auditor in order to assure that these services do not impair the auditor’s independence. These procedures generally approve the performance of specific services subject to a cost limit for all such services. This general approval is reviewed, and if necessary modified, at least annually. Management must obtain the specific prior approval of the committee for each engagement of our auditor to perform other audit-related or non-audit services. The committee does not delegate its responsibility to pre-approve services performed by our auditor to any member of management. The committee has delegated authority to the committee chair to pre-approve audit and non-audit services to be provided to us by our auditor provided that the fees for such services do not exceed $200,000. Any pre-approval of services by the committee chair pursuant to this delegated authority must be reported to the committee at its next regularly scheduled meeting.
Report of the Audit Committee
The Audit Committee has reviewed and discussed the audited financial statements for the year ended December 31, 2025 with the Company’s management and with EY, the Company’s independent registered public accounting firm. The Audit Committee has discussed with EY the matters required to be discussed by the applicable standards of the Public Company Accounting Oversight Board (“PCAOB”) and the SEC. The Audit Committee has also received the written disclosures and the letter from EY pursuant to applicable PCAOB requirements regarding its communications with the Audit Committee concerning independence, and the Audit Committee has discussed with EY its independence. Based on the foregoing, the Audit Committee recommended to the Board that the audited financial statements be included in the Company’s Annual Report on Form 10-K for the year ended December 31, 2025 for filing with the SEC.
This report is provided by the following directors, who serve on the Audit Committee:
Jennifer Fox (Chair)
Andrew Gottesdiener, M.D.
Peter Harwin
Board Recommendation
The Board recommends a vote “FOR” this proposal.
PROPOSAL 3: ADVISORY VOTE ON THE EXECUTIVE COMPENSATION OF
OUR NEOS
In accordance with the rules of the SEC and pursuant to the Dodd-Frank Act, we are asking stockholders to cast a non-binding, advisory vote on the compensation of our NEOs (as disclosed under the section titled “Executive Compensation,” including the compensation tables included therein and the accompanying narrative disclosure) (the “Say-on-Pay Vote”).
As an advisory vote, this proposal is not binding. However, our Board and Compensation Committee, which is responsible for designing and administering our executive compensation program, value the opinions expressed by stockholders in their vote on this proposal and will consider the outcome of the vote when making future compensation decisions for our NEOs.
We are required to hold a Say-on-Pay Vote at least once every three years, and we have determined to hold this vote every year. Unless the Board modifies its policy on the frequency of holding Say-on-Pay Votes, the next such vote is expected to occur in 2027.
The vote on this resolution is not intended to address any specific element of compensation but rather relates to the overall compensation of our NEOs, as described in this Proxy Statement. As discussed under the section titled “Compensation Discussion and Analysis,” our Board believes that its compensation philosophy and decisions support our key business objectives of creating value for, and promoting the interests of, our stockholders. Compensation of our NEOs is designed to enable us to attract and retain talented and experienced executives to lead us successfully in a competitive environment.
Board Recommendation
The Board recommends a vote “FOR” the compensation of our NEOs.
CORPORATE GOVERNANCE
Our business affairs are managed under the direction of our Board. Our Board has adopted the Principles of Corporate Governance (the “Corporate Governance Guidelines”) as a framework for the governance of the Company, which is posted on our website located at https://investors.apogeetherapeutics.com under “Corporate Governance.”
Our Governance Structure and Philosophy
Our governance practices reflect the environment in which we operate and are designed to support our mission to reshape the current standard of care for inflammatory and immune diseases. We are a clinical stage biotechnology company in an evolving industry, with a focus on developing our candidate pipeline through both business development and internal research efforts, and, like other companies in the biotechnology industry, face extreme stock price and volume fluctuations that are often unrelated or disproportionate to our operating performance. With these business environment considerations in mind, the Board believes our current governance structure enables the management team to act with deliberation and to focus on delivering long-term value to stockholders and protect minority investors from the interests of potentially short-sighted investors who may seek to act opportunistically and not in the best interests of the Company or stockholders generally. This structure includes the following elements:
•Classified board: our directors serve three-year terms, with approximately 1/3 of the Board (instead of the entire Board) elected at each annual meeting. This helps to provide stability and continuity, permitting directors to develop and share institutional knowledge and focus on the long term, and encourages stockholders to engage directly with the Board and management team regarding significant corporation transactions.
•Supermajority voting: the voting standard for most items is a majority of shares present, but 66 2/3% of the outstanding shares of our common stock are needed to amend certain provisions of our Certificate of Incorporation and Bylaws and remove directors. This helps protect against a small group of stockholders acting to amend our governing documents or to remove directors for reasons that may not be in the best interests of all stockholders.
•Plurality voting for directors: our directors are elected by a plurality of votes cast (instead of a majority of votes cast), meaning the nominees with the most votes are elected. This helps avoid potential disruption to the Board and management team as a result of a “failed election.”
•Stockholders cannot call special meetings or act by written consent: stockholders can propose business at each annual meeting (in accordance with our advance notice bylaws and Rule 14a-8) but cannot call a stockholder vote in between annual meetings or act by written consent. This helps avoid unnecessary diversion of Board and management time (potentially at the request of a limited number of stockholders acting to further short-term special interests) from executing on our long-term strategy.
Recognizing that the Company’s operating environment continues to evolve and that governance practices should not be static as a matter of course, the Board annually evaluates our governance structure to confirm it remains in the best interests of the Company and stockholders and values input from our stockholders on this topic.
Board Composition
Director Nomination Process
The Nominating Committee is responsible for, among other things, engaging in succession planning for directors and identifying qualified individuals to become members of the Board to oversee management’s execution of the Company’s strategy and safeguard the long-term interests of stockholders. In this regard, the committee is charged with developing and recommending Board membership criteria to the Board for approval, evaluating the composition of the Board annually to assess the skills and experience that are currently represented on the Board and to assess the criteria that may be needed in the future, and identifying and reviewing the qualifications of and recommending potential director candidates.
In identifying potential candidates for Board membership, the Nominating Committee considers recommendations from directors, stockholders, management and others, including, from time to time, third-party search firms to assist it in locating qualified candidates. Once potential director candidates are identified, the committee, with the assistance of management, undertakes a vetting process that considers each candidate’s background, independence and fit with the Board’s priorities. As part of this vetting process, the committee, as well as other members of the Board and our CEO, may conduct interviews with the candidates. If the committee determines that a potential candidate meets the needs of the Board and has the desired qualifications, it recommends the candidate to the full Board for appointment or nomination and to the stockholders for election at the annual meeting.
Criteria for Board Membership
In assessing potential candidates for Board membership and in assessing Board composition, the Nominating Committee considers a wide range of factors, including directors’ experience, knowledge, understanding of our business environment and specific skills they may possess that are helpful to the Company (including leadership experience, financial expertise and industry knowledge). The committee generally seeks to balance the experiences, skills and characteristics represented on the Board and does not assign specific weight to any of these factors. In addition, the Nominating Committee generally believes it is important for all Board members to possess the highest personal and professional ethics, integrity and values, an inquisitive and objective perspective, a sense for priorities and balance, the ability and willingness to devote sufficient time and attention to Board matters, and a willingness to represent the long-term interests of all our stockholders.
Board Diversity
In addition to the factors discussed above, the Board and the Nominating Committee actively seek to achieve a diversity of perspectives, skills and occupational and personal backgrounds on the Board. The Nominating Committee considers a potential director candidate’s ability to contribute to the diversity of personal backgrounds on the Board, including with respect to gender, race, ethnic and national background, geography, age and sexual orientation. As part of the search process for new directors, the Nominating Committee endeavors to include, and instructs any search firm it engages to endeavor to include women and minority candidates in the broader pool of candidates from which the Committee considers director candidates, with the ultimate selection of nominees determined based on merit.
The Nominating Committee assesses its effectiveness in balancing these considerations in connection with its annual evaluation of the composition of the Board. In this regard, our Board of seven directors, as of May 11, 2026, will include two directors (29%) who self-identify as female and three directors (43%) who self-identify as racially/ethnically diverse.
Stockholder Recommendations for Directors
It is the Nominating Committee’s policy to consider written recommendations from stockholders for director candidates. The committee considers candidates recommended by our stockholders in the same manner as a candidate recommended by other sources. Any such recommendations should be submitted to the committee as described under “Stockholder Communications” and should include the same information required under our Bylaws for nominating a director, as described under “Stockholder Proposals and Director Nominations for Next Year’s Annual Meeting.”
Director Time Commitments
While Board members benefit from service on the boards of other companies and such service is encouraged, pursuant to the Corporate Governance Guidelines, directors are expected to limit the number of other boards on which they serve so as not to interfere with their service as a director of the Company. As part of the annual director nomination process, the Nominating Committee considers directors’ adherence to these expectations.
Board Leadership Structure
Currently, the roles of Chair of the Board and the CEO are separate, and the Chair is an independent director - Mr. McKenna serves as our independent Chair of the Board while Dr. Henderson serves as our CEO. The Board believes that this structure is appropriate for the Company as the independent Chair’s leadership of the Board and oversight of corporate governance matters enables our CEO to focus on leading the Company’s business and developing our drug candidate pipeline.
We do not have a policy regarding whether the roles of the Chair and the CEO should be separate or combined, and our Board believes that we should maintain our flexibility to select the Chair and CEO and determine the appropriate leadership structure, from time to time, based on criteria that are in our best interests and the best interests of our stockholders. Our Board periodically reviews its leadership structure to evaluate whether the structure remains appropriate for the Company. At any time when the Chair is not independent or there is not a Chair, the independent directors of the Board will designate an independent director to serve as lead independent director for a period of at least one year.
The independent directors have the opportunity to meet in executive sessions without management present at every regular Board meeting. The purpose of these executive sessions is to encourage and enhance communication among the independent directors.
The Board believes that its programs for overseeing risk, as described under “Board Risk Oversight,” would be effective under a variety of leadership frameworks. Accordingly, the Board’s risk oversight function did not significantly impact its selection of the current leadership structure.
Director Independence
Nasdaq listing rules require a majority of a listed company’s board of directors to be comprised of independent directors who, in the opinion of the board of directors, do not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. Subject to specified exceptions, each member of a listed company’s audit, compensation and nominating committees must be independent, and audit and compensation committee members must satisfy additional independence criteria under the Securities Exchange Act of 1934, as amended (the “Exchange Act”).
Our Board undertook a review of its composition and the independence of each director. Based upon information requested from and provided by each director concerning his or her background, employment and affiliations, our Board has determined that Lisa Bollinger, M.D., Jennifer Fox, Andrew Gottesdiener, M.D., Peter Harwin, William (BJ) Jones, Jr., Tomas Kiselak, Mark C. McKenna and Nimish Shah qualify as “independent directors” as defined by the Nasdaq listing rules. Michael Henderson, M.D., is not an independent director because he is our CEO. In making such determinations, our Board considered the relationships that each such non-employee director has with our Company and all other facts and circumstances our Board deemed relevant in determining independence, including any business, consulting or employment relationships with our significant investors, and the transactions described under “Certain Relationships and Transactions” in this Proxy Statement. Our Board also determined that each of the directors currently serving on the Audit Committee and the Compensation Committee satisfy the additional independence criteria applicable to directors on such committees under Nasdaq listing rules and the rules and regulations established by the SEC.
Board Committees
Our Board has a separately designated Audit Committee, Compensation Committee and Nominating Committee, each of which has the composition and responsibilities described below. Members serve on these committees until their resignation or until otherwise determined by our Board. Each of these committees is empowered to retain outside advisors as it deems appropriate, regularly reports its activities to the full Board and has a written charter, which is posted on our website located at https://investors.apogeetherapeutics.com under “Corporate Governance.” The following table provides membership information for each of the Board committees as of the date of this Proxy Statement.
|
|
|
|
|
|
|
|
|
|
Name |
|
|
Audit
Committee |
|
|
Compensation
Committee |
|
|
Nominating
Committee |
Lisa Bollinger, M.D. |
|
|
|
|
|
|
|
|
X |
Jennifer Fox |
|
|
Chair |
|
|
X |
|
|
|
Andrew Gottesdiener, M.D.(1) |
|
|
X |
|
|
|
|
|
|
Peter Harwin(1) |
|
|
X |
|
|
|
|
|
|
Michael Henderson, M.D. |
|
|
|
|
|
|
|
|
|
William (BJ) Jones, Jr. |
|
|
|
|
|
X |
|
|
Chair |
Tomas Kiselak |
|
|
|
|
|
|
|
|
|
Mark C. McKenna(2) |
|
|
|
|
|
Chair |
|
|
X |
Nimish Shah(3) |
|
|
|
|
|
|
|
|
X |
# of Meetings in 2025 |
|
|
4 |
|
|
5 |
|
|
3 |
(1)Effective as of May 11, 2026, Dr. Gottesdiener and Mr. Harwin will no longer be members of the Board.
(2)Effective as of May 11, 2026, Mr. McKenna will no longer be a member of the Nominating Committee and will serve as a member of the Audit Committee.
(3)Effective as of May 11, 2026, Mr. Shah will no longer be a member of the Nominating Committee and will serve as a member of the Audit Committee.
Audit Committee. The primary responsibilities of our Audit Committee are to oversee the accounting and financial reporting processes of the Company, including the audits of the Company’s financial statements, the integrity of the financial statements and the annual review of the performance, qualifications and independence of the outside auditor. This includes reviewing the financial information provided to stockholders and others and the adequacy and effectiveness of the Company’s internal controls. The committee also makes recommendations to the Board as to whether financial statements should be included in the Company’s Annual Report on Form 10-K.
Ms. Fox qualifies as an “audit committee financial expert,” as that term is defined in the rules and regulations established by the SEC, and all members of the Audit Committee are “financially literate” under Nasdaq listing rules.
Compensation Committee. The primary responsibilities of our Compensation Committee are to periodically review and approve the compensation and other benefits for our senior officers and directors. This includes reviewing and approving corporate goals and objectives relevant to the compensation of our CEO, evaluating the performance of our CEO in light of the goals and objectives and setting or recommending to the Board the CEO’s compensation based on the committee’s evaluation. The committee also oversees the evaluation of other senior officers and sets or recommends to the Board the compensation of such senior officers based upon the recommendation of the CEO. The committee also administers and makes recommendations to the Board regarding equity incentive plans that are subject to the Board’s approval and approves the grant of equity awards under the plans.
The Compensation Committee may delegate its duties and responsibilities to one or more subcommittees. The committee may also delegate authority to review and approve the compensation of our employees to certain of our executive officers. Even where the committee does not delegate authority, our executive officers will typically make recommendations to the committee regarding compensation to be paid to our employees and the size of equity awards under our equity incentive plans but will not be present during voting or deliberations on their own compensation. The committee has the authority to engage outside advisors, such as compensation consultants, to assist it in carrying out its responsibilities. The committee has engaged Alpine Rewards, LLC (“Alpine”) since 2023 to provide advice regarding the amount and form of executive and director compensation. The committee assessed Alpine’s independence and has determined that (1) Alpine satisfies applicable independence criteria, and (2) Alpine’s work with the Company does not raise any conflicts of interest, in each case under applicable Nasdaq listing rules and the rules and regulations established by the SEC.
Nominating Committee. The primary responsibilities of our Nominating Committee are to identify individuals qualified to become members of the Board, recommend director candidates to the Board, including for election or reelection to the Board at each annual stockholders’ meeting, and perform a leadership role in shaping the Company’s corporate governance. In addition, the committee is responsible for developing and recommending to the Board criteria for identifying and evaluating qualified director candidates and developing and recommending to the Board a set of corporate governance principles. The committee is also responsible for making recommendations to the Board concerning the size, structure, composition and functioning of the Board and its committees, engaging in succession planning for the Board and key leadership roles on the Board and its committees and overseeing succession planning for positions held by executive officers and other critical leadership roles.
Board Risk Oversight
We believe that risk management is an important part of establishing and executing on the Company’s business strategy. Our Board, as a whole and at the committee level, focuses its oversight on the most significant risks facing the Company and on the Company’s processes to identify, prioritize, assess, manage and mitigate those risks. The committees oversee specific risks within their purview, as follows:
•The Audit Committee has overall responsibility for overseeing the Company’s practices with respect to risk assessment and management, including cybersecurity risks. Additionally, the committee is responsible for overseeing management of risks related to our accounting and financial reporting processes.
•The Compensation Committee is responsible for overseeing management of risks related to our compensation policies and programs.
•The Nominating Committee is responsible for overseeing management of risks related to director succession planning and corporate governance practices.
Our Board and its committees receive regular reports from members of the Company’s senior management on areas of material risk to the Company, including strategic, operational, financial, legal and regulatory risks. While our Board has an oversight role, management is principally tasked with direct responsibility for assessing and managing risks, including implementing processes and controls to mitigate their effects on the Company.
Other Corporate Governance Practices and Policies
Director Attendance
The Board met six times during the year ended December 31, 2025. During 2025, each member of the Board attended at least 75% of the aggregate number of meetings of the Board and the committees on which he or she served during the period in which he or she was on the Board or committee.
Directors are encouraged to attend the annual meeting of stockholders. All of our directors attended the 2025 annual meeting of stockholders.
Stockholder Communications
Stockholders and other interested parties may communicate with our Board or a particular director by sending a letter addressed to the Board or a particular director to our Corporate Secretary at the address set forth on the first page of this Proxy Statement. These communications will be compiled and reviewed by our Corporate Secretary, who will determine whether the communication is appropriate for presentation to the Board or the particular director. The purpose of this screening is to allow the Board to avoid having to consider irrelevant or inappropriate communications (such as advertisements, solicitations and hostile communications).
To enable the Company to speak with a single voice, as a general matter, senior management serves as the primary spokesperson for the Company and is responsible for communicating with various constituencies, including stockholders, on behalf of the Company. Directors may participate in discussions with stockholders and other constituencies on issues where Board-level involvement is appropriate. In addition, the Board oversees the Company’s stockholder engagement efforts.
Code of Business Conduct and Ethics
Our Board has adopted a Code of Business Conduct and Ethics (“the Code of Conduct”) that establishes the standards of ethical conduct applicable to all our directors, officers and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions. The full text of our Code of Conduct is posted on our website at https://investors.apogeetherapeutics.com/corporate-governance/governance-overview. It addresses, among other matters, compliance with laws and policies, conflicts of interest, corporate opportunities, regulatory reporting, external communications, confidentiality requirements, insider trading, proper use of assets and how to report compliance concerns. We intend to disclose future amendments to certain provisions of the Code of Conduct and waivers of the Code of Conduct granted to executive officers and directors on the website within four business days following the date of the amendment or waiver. The Audit Committee is responsible for applying and interpreting our Code of Conduct in situations where questions are presented to it.
Insider Trading Policy and Anti-Hedging Policy
We have adopted insider trading policies and procedures governing the purchase, sale and other transactions in Company securities by our directors, officers and employees, and other covered persons, as well as the Company itself, that we believe are reasonably designed to promote compliance with insider trading laws, rules and regulations, and Nasdaq listing rules, as applicable. As part of these policies and procedures, we prohibit our directors, officers, employees and consultants from engaging in (a) short sales; (b) transactions involving publicly traded options or other derivatives, such as trading in puts or calls with respect to Company securities; and (c) hedging or monetization transactions.
Director Compensation
The following table presents all of the compensation awarded to, earned by, or paid to our non-employee directors during the year ended December 31, 2025. We have reimbursed and will continue to reimburse all of our non-employee directors for their reasonable out-of-pocket expenses incurred in attending Board and committee meetings. Dr. Henderson, our CEO, does not receive any additional compensation for his service on the Board. The compensation received by Dr. Henderson for his service as our CEO is presented in the 2025 Summary Compensation Table below.
|
|
|
|
|
|
|
|
|
|
|
|
|
Name |
|
Option
Awards
($)(1)(2) |
|
|
Fees Earned
or Paid in
Cash
($) |
|
|
Total
($) |
|
Lisa Bollinger, M.D.(3) |
|
$ |
399,988 |
|
|
$ |
44,538 |
|
|
$ |
444,526 |
|
Jennifer Fox(4) |
|
$ |
399,988 |
|
|
$ |
64,038 |
|
|
$ |
464,026 |
|
Andrew Gottesdiener, M.D.(5)(11) |
|
$ |
399,988 |
|
|
$ |
48,846 |
|
|
$ |
448,834 |
|
Peter Harwin(6)(11) |
|
$ |
399,988 |
|
|
$ |
48,846 |
|
|
$ |
448,834 |
|
William (BJ) Jones, Jr.(7) |
|
$ |
399,988 |
|
|
$ |
55,423 |
|
|
$ |
455,411 |
|
Tomas Kiselak(8) |
|
$ |
399,988 |
|
|
$ |
40,000 |
|
|
$ |
439,988 |
|
Mark McKenna(9) |
|
$ |
399,988 |
|
|
$ |
87,231 |
|
|
$ |
487,219 |
|
Nimish Shah(10) |
|
$ |
399,988 |
|
|
$ |
44,538 |
|
|
$ |
444,526 |
|
(1)The amounts disclosed represent the aggregate grant date fair value of the stock options and awards granted to our non-employee directors during 2025 under our 2023 Equity Incentive Plan (the “2023 Plan”). The amounts were computed in accordance with ASC Topic 718. The assumptions used in calculating the grant date fair value of the stock options are set forth in Note 2 to our audited consolidated financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2025. These amounts do not reflect the actual economic value that may be realized by the director.
(2)In 2025, Dr. Bollinger, Ms. Fox, Dr. Gottesdiener and Messrs. Harwin, Jones, Kiselak, McKenna and Shah were each awarded 14,461 options to purchase shares of our common stock, as described further below.
(3)As of December 31, 2025, Dr. Bollinger held vested options to purchase 14,295 shares of our common stock.
(4)As of December 31, 2025, Ms. Fox held vested options to purchase 10,370 shares of our common stock.
(5)As of December 31, 2025, Dr. Gottesdiener held vested options to purchase 42,208 shares of our common stock.
(6)As of December 31, 2025, Mr. Harwin held vested options to purchase 42,208 shares of our common stock.
(7)As of December 31, 2025, Mr. Jones held vested options to purchase 10,370 shares of our common stock.
(8)As of December 31, 2025, Mr. Kiselak held vested options to purchase 42,208 shares of our common stock.
(9)As of December 31, 2025, Mr. McKenna held vested options to purchase 127,042 shares of our common stock.
(10)As of December 31, 2025, Mr. Shah held vested options to purchase 42,208 shares of our common stock.
(11)On April 21, 2026, each of Dr. Gottesdiener and Mr. Harwin notified the Board of his intention to resign from the Board effective May 11, 2026.
Director Compensation Policy
Our Board approved our amended director compensation policy for our non-employee directors which went into effect in February 2024 and which consisted of the following:
•an annual cash retainer of $40,000 for service on the Board (other than as Chair);
•an annual cash retainer of $70,000 for service as Chair of the Board;
•an annual cash retainer of $15,000 for service as chairperson of the Audit Committee of the Board;
•an annual cash retainer of $10,000 for service as chairperson of the Compensation Committee of the Board;
•an annual cash retainer of $8,000 for service as chairperson of the Nominating Committee of the Board;
•an annual cash retainer of $7,500 for service on the Audit Committee of the Board (other than as chairperson);
•an annual cash retainer of $5,000 for service on the Compensation Committee of the Board (other than as chairperson);
•an annual cash retainer of $4,000 for service on the Nominating Committee of the Board (other than as a chairperson);
•an initial one-time equity grant of stock options with a fair value of $700,000 (valued based on the grant date fair value and subject to a limit of 60,000 stock options) under the 2023 Plan, subject to annual vesting over three years following the date of grant;
•an annual equity grant of stock options with a fair value of $350,000 (valued based on the grant date fair value and subject to a limit of 30,000 stock options) under the 2023 Plan, subject to vesting on the one year anniversary of the date of grant; and
•a director compensation limit of $1,000,000 during the year such director is appointed and $750,000 annually thereafter.
In March 2025, the Board approved, upon the recommendation of the Compensation Committee, an amended and restated director compensation policy, which became effective as of the date of the 2025 Annual Meeting of Stockholders and which consists of the following:
•an annual cash retainer of $40,000 for service on the Board (other than as Chair);
•an annual cash retainer of $70,000 for service as Chair of the Board;
•an annual cash retainer of $20,000 for service as chairperson of the Audit Committee of the Board;
•an annual cash retainer of $15,000 for service as chairperson of the Compensation Committee of the Board;
•an annual cash retainer of $10,000 for service as chairperson of the Nominating Committee of the Board;
•an annual cash retainer of $10,000 for service on the Audit Committee of the Board (other than as chairperson);
•an annual cash retainer of $7,500 for service on the Compensation Committee of the Board (other than as chairperson);
•an annual cash retainer of $5,000 for service on the Nominating Committee of the Board (other than as a chairperson);
•an initial one-time equity grant of stock options with a fair value of $800,000 (valued based on the grant date fair value and subject to a limit of 35,000 stock options) under the 2023 Plan, subject to annual vesting over three years following the date of grant;
•an annual equity grant of stock options with a fair value of $400,000 (valued based on the grant date fair value and subject to a limit of 17,500 stock options) under the 2023 Plan, subject to vesting on the one year anniversary of the date of grant; and
•a director compensation limit of $1,000,000 during the year such director is appointed and $750,000 annually thereafter.
EXECUTIVE OFFICERS
Biographical and other information regarding our executive officers, who are appointed by the Board and serve at the Board’s discretion, is set forth below. There are no family relationships among any of our directors or executive officers.
|
|
|
|
|
|
|
|
|
|
|
Name |
|
|
Age
(as of April 24) |
|
|
Position |
|
Michael Henderson, M.D.(1) |
|
|
|
|
36 |
|
|
|
Chief Executive Officer and Director |
|
Carl Dambkowski, M.D. |
|
|
|
|
41 |
|
|
|
Chief Medical Officer |
|
Jane Pritchett Henderson |
|
|
|
|
60 |
|
|
|
Chief Financial Officer |
|
(1)For Dr. Henderson’s biographical information, see “Information Regarding Director Nominees and Continuing Directors” above.
Carl Dambkowski, M.D. Dr. Dambkowski has served as our Chief Medical Officer since September 2022. Prior to joining Apogee, Dr. Dambkowski served as a strategic and clinical leader for a variety of companies, including as Chief Medical Officer of QED Therapeutics, Inc., a private biotechnology company, from July 2021 to September 2022; Chief Strategy Officer and EVP of Operations of Origin Biosciences, Inc., a private bioecology company, from March 2018 to June 2021; and Chief Medical Officer of Navire Pharma, Inc., a private biotechnology company, from January 2020 to September 2022, where he served as the clinical lead starting prior to IND for BBP-398 through the out licensing of the compound to Bristol-Myers Squibb based on initial clinical data and for low-dose infigratinib in achondroplasia through initial proof-of-concept data. He was part of the core team that brought TRUSELTIQ® (infigratinib) and NULIBRY® (fosdenopterin) through regulatory review and FDA approval at QED Therapeutics and Origin Biosciences, respectively. From July 2016 to March 2018, Dr. Dambkowski was an associate at McKinsey & Company, a global management consulting firm, where he advised biotech and pharmaceutical companies across the world on a range of research and development activities. Dr. Dambkowski co-founded Novonate, Inc., a private medical device company focused on building life-saving devices for neonates, in January 2015. Dr. Dambkowski has served on the board of directors of Oruka Therapeutics, Inc. (Nasdaq: ORKA), a publicly traded biotechnology company, since February 2024. Dr. Dambkowski has coauthored numerous peer-reviewed publications and scientific abstracts and is a named inventor on multiple published and granted patents. Dr. Dambkowski was trained as a physician at Stanford University, where he also received his M.D. with a concentration in bioengineering. He also received a B.A. (with honors) from Stanford University and an M.A. from Columbia University.
Jane Pritchett Henderson. Ms. Henderson has served as our Chief Financial Officer since January 2023. Prior to joining Apogee, Ms. Henderson served as the Chief Financial Officer and Chief Business Officer of Adagio Therapeutics, Inc. (now Invivyd, Inc.) (Nasdaq: IVVD), a biotechnology company developing antibody therapeutics for coronaviruses, from December 2020 to November 2022. Prior to joining Adagio Therapeutics, Ms. Henderson served as Chief Financial Officer of Turnstone Biologics Corp., a private viral immuno-oncology company, from June 2018 to December 2020, as Chief Financial Officer and Senior Vice President of Corporate Development of Voyager Therapeutics, Inc. (Nasdaq: VYGR), a gene therapy company, from January 2017 to June 2018, and as the Senior Vice President, Chief Financial and Business Officer of Kolltan Pharmaceuticals, Inc., a private oncology biopharmaceutical company, from February 2013 until November 2016, when Kolltan Pharmaceuticals was acquired by Celldex Therapeutics, Inc. Prior to Kolltan Pharmaceuticals, Ms. Henderson served in various financial and business development executive roles at biopharmaceutical companies after spending almost 20 years in health care investment banking. Ms. Henderson serves on the board of directors of Ventus Therapeutics, Inc., a private biopharmaceutical company. She also served on the board of directors of Akero Therapeutics, Inc. (Nasdaq: AKRO), a biotechnology company, from April 2019 until its acquisition by Novo Nordisk in December 2025, Cargo Therapeutics, Inc. (Nasdaq: CRGX), a biotechnology company, from June 2024 until its acquisition by Concentra Biosciences in August 2025, IVERIC Bio, Inc. (Nasdaq: ISEE), a biopharmaceutical company, from January 2018 until its acquisition by Astellas Pharma Inc. in July 2023, and Sesen Bio Inc. (Nasdaq: SESN), a biopharmaceutical company, from October 2018 to November 2021. Ms. Henderson also serves on the Dedman College Executive Board of Southern Methodist University. Ms. Henderson received a B.S. in psychology from Duke University.
EXECUTIVE COMPENSATION
COMPENSATION DISCUSSION & ANALYSIS
In this Compensation Discussion and Analysis (“CD&A”) we provide an overview of our compensation philosophy and each element of our executive compensation program with regard to the compensation awarded to, earned by, or paid to our NEOs during our fiscal year ended December 31, 2025.
For the fiscal year ended December 31, 2025, our NEOs were:
|
|
|
Name |
|
Position |
Michael Henderson, M.D. |
|
Chief Executive Officer and Director |
Carl Dambkowski, M.D. |
|
Chief Medical Officer |
Jane Pritchett Henderson |
|
Chief Financial Officer |
2025 Business Highlights
2025 was a significant year for the Company as we advanced clinical development of our lead product candidate, zumilokibart (APG777), while also continuing to advance our other pipeline programs. The following Company performance highlights from the year directly contributed to achievement under our 2025 annual incentive program.
Zumilokibart (APG777) Program Highlights
•In July 2025, announced positive 16-week data from our APEX Phase 2 Part A clinical trial of zumilokibart in moderate-to-severe atopic dermatitis (“AD”);
•Accelerated the readout, completed enrollment ahead of schedule and exceeded the enrollment target for our APEX Phase 2 Part B clinical trial of zumilokibart (collectively, the “Zumilokibart Program Achievements”); and
•In 2025, completed enrollment of our Phase 1b trial of zumilokibart in patients with mild-to-moderate asthma.
APG279 Program Highlights
•In March 2025, announced positive interim safety and pharmacokinetic (“PK”) data from the APG990 Phase 1 clinical trial for healthy volunteers; and
•In July 2025, dosed the first patient in our Phase 1b head-to-head clinical trial of APG279 (zumilokibart + APG990) vs. DUPIXENT in AD (collectively, the “APG279 Program Achievements”).
APG273 Program Highlights
•In November 2025, announced positive interim safety, PK and pharmacodynamic results from our healthy volunteer trial of APG333 (the “APG273 Program Achievement”).
APG808 Program Highlights
•In May 2025, reported positive interim Phase 1b results of APG808 in patients with mild-to-moderate asthma (the “APG808 Program Achievement”).
Finance Highlights
•In October 2025, completed an underwritten public offering, including the full exercise of the underwriter’s option to purchase additional shares, for aggregate gross proceeds of $345.1 million before deducting underwriting discounts and commissions and other expenses;
•Raised gross proceeds of $67.6 million, less commissions and other offering expenses from the Company’s at-the-market offering program during the year ended December 31, 2025; and
•In 2025, extended projected cash runway into the second half of 2028 and through estimated Phase 3 topline data for zumilokibart in moderate to severe AD (collectively, the “Finance Achievements”).
2025 Compensation Highlights
Our Compensation Committee believes that executive compensation should be directly linked to short-term and long-term performance. The following compensation features are designed to align our NEOs’ interests with stockholder interests and market best practices:
•Modest base salary adjustments in 2025: Base salary increases for our NEOs ranged from 3% to 6%. Salary increases were generally based on competitive market positioning and took into account individual responsibilities, performance and experience.
•Annual bonuses linked to pre-determined milestone performance goals: Annual bonuses for our NEOs were paid out at 130% of target, based on the achievement of the 2025 corporate performance goals. See “—2025 Business Highlights” discussed above and “—Key Elements of Compensation—Annual Cash Bonus.”
•Annual stock option grants aligned to performance results: NEOs received stock option grants in December 2024, based on 2024 performance and to incentivize future performance, and in January 2026, based on 2025 performance and to incentivize future performance.
•Pay mix is highly “at risk”: The 2025 percentage of pay that is “at risk” for our CEO and other NEOs based on 2025 base salary, 2025 annual bonus payout and December 2024 equity grants is 95% and 89%, respectively, helping us align pay with performance.
Compensation Components:
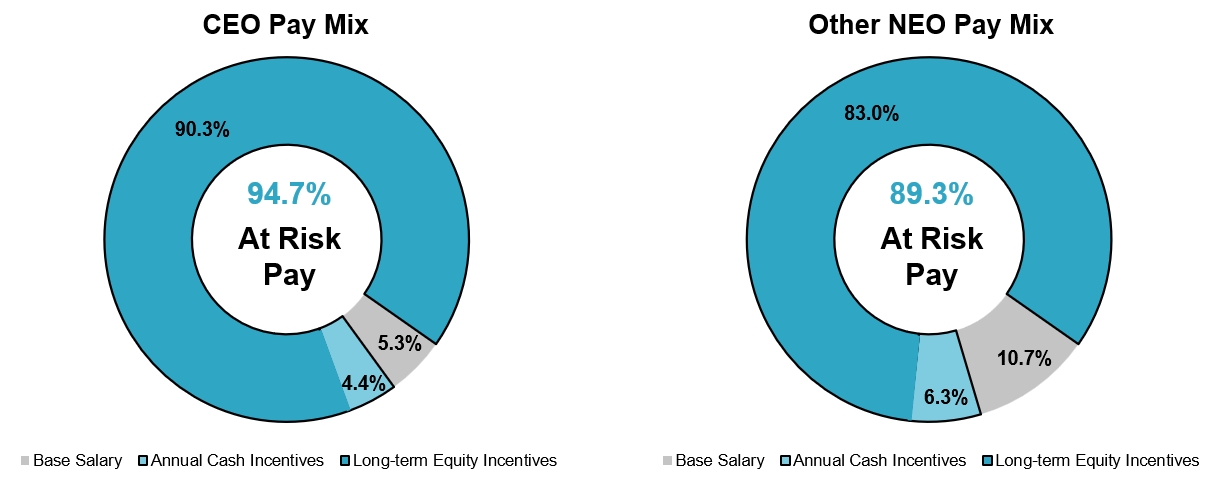
*Equity awards based upon grant date value.
Responsible Executive Compensation Policies and Practices
The following table summarizes our executive compensation policies and practices, including responsible policies and practices we have implemented and the policies and practices we have avoided to better serve our stockholders’ long-term interests.
|
|
|
What we do: |
|
What we do not do: |
☑ Performance metrics tied to the Company performance. The performance metrics used to determine payouts under our annual incentive plan are based on Company performance, aligning the interests of our NEOs and our stockholders. ☑ At risk compensation. We emphasize long-term incentive grants to align with stockholder value and balance short- and long-term strategic objectives. ☑ Multi-year vesting requirements. The time-based equity awards granted to our executive officers vest over multi-year periods, consistent with current market practice and our retention objectives. ☑ Independent compensation consultant. Our Compensation Committee engages an independent compensation consultant to provide information, analysis, and other advice on executive compensation independent of management. ☑ Compensation recovery (“clawback”) policy. We maintain a clawback policy that complies with the requirements of Exchange Act Rule 10D-1 and applicable Nasdaq listing standards for all incentive payments and performance-based equity awards granted to executive officers. ☑ Annual compensation reviews and votes. The Compensation Committee evaluates our executive compensation program at least annually, and we are conducting a “say-on-pay” vote on an annual basis |
|
☒ No excise tax “gross-ups”. None of our employment-related agreements provide for excise tax “gross-ups”. ☒ No single-trigger change in control arrangements. Our compensation arrangements do not provide for single-trigger payments or benefits upon the occurrence of a change in control and instead also require a termination of employment. ☒ No retirement plans other than Section 401(k) plan. Our NEOs are eligible to participate in our Section 401(k) retirement savings plan available to our other full-time, salaried employees. We do not provide any pension or other retirement benefits to our NEOs. ☒ No hedging, short selling or other inherently speculative transactions. Our insider trading policy prohibits our executive officers and directors from engaging in hedging, short sales or other inherently speculative transactions. ☒ No stock option repricing. We do not reprice any stock options, including underwater stock options, without stockholder approval. |
Consideration of Stockholder Advisory Vote on Executive Compensation
At the 2025 Annual Meeting of Stockholders, consistent with the recommendation of our Board, our stockholders voted to hold non-binding, advisory votes on the compensation of our NEOs (each, a “Say-on-Pay Vote”) every year. Beginning at our 2026 Annual Meeting of Stockholders, and each year thereafter until our next vote on the frequency of Say-on-Pay Votes, which will occur at the 2031 Annual Meeting of Stockholders, our stockholders will be provided the opportunity to cast a Say-on-Pay Vote. Because we value the opinions of our stockholders, the Board and the Compensation Committee will consider the outcome of the Say-on-Pay Vote, as well as any feedback received throughout the year, when making compensation decisions for our NEOs and our executive compensation program in the future.
Compensation Philosophy and Objectives
Our current executive compensation program is intended to align executive compensation with our business objectives and to enable us to attract, retain and reward executive officers who contribute to our long-term success. In furtherance of these goals, the Compensation Committee adheres to the following guidelines when making determinations regarding compensation:
•provide a competitive total compensation package that enables us to attract and retain highly qualified executives with the skills and experience required for the achievement of business goals;
•align compensation elements with the Company’s corporate strategies, business objectives, and the long-term interests of our stockholders;
•promote the achievement of key clinical development goals and strategic and financial performance measures by linking short-term and long-term cash and equity incentives to the achievement of measurable corporate and individual performance goals and objectives;
•reward executives for actions that create short- and long-term sustainable stockholder value;
•reinforce a performance driven orientation and Company culture; and
•enable differentiation in the industry.
The compensation paid or awarded to our executive officers is generally based on a qualitative assessment of each individual’s performance compared against the business objectives established for the fiscal year as well as market peer group data and our historical compensation practices. In the case of new hire executive officers, their compensation is primarily determined based on the negotiations of the parties, as well as our historical compensation practices and market peer group data. For 2025, 2024, and 2023, the material elements of our executive compensation program were base salary, annual cash bonus awards and long-term equity incentives in the form of incentive units prior to the IPO and stock options following the IPO. The Compensation Committee believes that cash compensation in the form of base salary and an annual bonus opportunity provides our executive officers with short-term rewards for success in achieving annual goals and objectives, and that long-term compensation through the award of stock options aligns the objectives of management with those of our stockholders with respect to long-term performance and success of the Company.
In setting compensation levels for our executive officers, the Compensation Committee considers a variety of factors, including peer group survey data, experience, tenure, role, responsibilities, performance, the relative importance of the executive’s role within the organization and competitive market practices. Compensation paid to our NEOs is delivered primarily through at-risk pay, based on both short-term and long-term incentives.
In addition to our compensation elements, the following compensation program features are designed to align our executive officers’ interests with stockholder interests and market best practices.
Process for Setting Executive Compensation
Our Compensation Committee reviews compensation practices and philosophy annually for all employees, including our executives. In setting executive officer base salaries and bonuses and granting executive officer equity incentive awards, they consider compensation for comparable positions in the market, the historical compensation levels of our executives, individual performance as compared to our expectations and objectives, our desire to motivate our executive officers to achieve short-and long-term results that are in the best interests of our stockholders and our desire to incentivize a long-term commitment to our Company. While we do not establish compensation levels based solely on benchmarking, pay practices at other companies are an important factor that the Compensation Committee considers in assessing the reasonableness of compensation and ensuring that our compensation practices are competitive in the marketplace.
Our Compensation Committee is responsible for approving all executive compensation matters, and in the case of our CEO, makes recommendations to the Board for approval, as appropriate. Our Compensation Committee typically reviews and discusses management’s proposed compensation with the CEO for all NEOs other than the CEO. Based on those discussions and its discretion, taking into account the factors noted above, the Compensation Committee then determines the compensation for each NEO, and in the case of the CEO, makes recommendations to the Board for approval, as appropriate. While the CEO discusses his recommendations for the other NEOs with the Compensation Committee, he does not participate in the deliberations concerning, or the determination of, his own compensation. Since 2023, our Compensation Committee has engaged Alpine Rewards (“Alpine”) as its independent executive compensation consultant and the Board and the Compensation Committee considered Alpine’s input on certain compensation matters as they deemed appropriate. Pursuant to the factors set forth in Item 407 of Regulation S-K of the Exchange Act, the Compensation Committee has reviewed the independence of Alpine and conducted a conflicts of interest assessment (taking into consideration factors specified in the Nasdaq listing standards) and has concluded that Alpine is independent and its work for the Compensation Committee has not raised any conflicts of interest. No other consulting services were provided by Alpine or its affiliates to the Company in 2025.
Peer Group and the Use of Market Data
While we do not establish compensation levels based solely on benchmarking, pay practices at other companies are an important factor that the Compensation Committee considers in assessing the reasonableness of compensation and ensuring that our compensation practices are competitive in the marketplace. The Compensation Committee reviews all market percentiles of relevant market data as one frame of reference in making its executive compensation decisions.
In order to evaluate the level of compensation for our NEOs for 2025, our Compensation Committee, using information provided by Alpine, established a peer group of publicly traded companies in the biopharmaceutical space (“Peer Group”) based on a balance of the following criteria:
•companies with comparable market capitalizations (i.e., in the range of $1.5 billion to $6.5 billion);
•U.S. based pre-commercial biopharmaceutical companies; and
•companies with headcount between 100 to 500 employees.
|
|
Akero Therapeutics |
Neumora Therapeutics |
Arcellx |
Nuvalent |
Arrowhead Pharmaceuticals |
Revolution Medicines |
Avidity Biosciences |
Spyre Therapeutics |
Celldex Therapeutics |
Structure Therapeutics |
Dyne Therapeutics |
Vaxcyte |
IDEAYA Biosciences |
Viking Therapeutics |
Immunovant |
Vir Biotechnology |
Kymera Therapeutics |
|
Key Elements of Compensation
Base Salary
Each NEO’s base salary is a fixed component of annual compensation for performing specific duties and functions, and has been established by our Board taking into account each individual’s experience, skills, knowledge and responsibilities, as well as market peer group data. Base salaries for our NEOs are reviewed annually by our Compensation Committee, typically in connection with our annual performance review process, and adjusted from time to time, based on the recommendation of the Compensation Committee, to realign salaries with market levels after taking into account individual responsibilities, performance and experiences. For 2025, base salary increases for the NEOs ranged from 3% to 6% after consideration of the foregoing factors. The 2025 base salaries of our NEOs are set forth in the table below:
|
|
|
|
|
|
|
|
|
|
|
Name |
|
2025 Base Salary |
|
|
2024 Base Salary |
|
|
Percentage Change |
Michael Henderson, M.D. |
|
$ |
700,000 |
|
|
$ |
661,500 |
|
|
6% |
Carl Dambkowski, M.D. |
|
$ |
540,000 |
|
|
$ |
525,000 |
|
|
3% |
Jane Pritchett Henderson |
|
$ |
540,000 |
|
|
$ |
525,000 |
|
|
3% |
Annual Cash Bonus
Our Compensation Committee approves annual cash bonuses for our NEOs (other than our CEO, whose annual cash bonus is determined by the Board) based on Company performance as compared to the corporate performance goals and objectives established by the Board at the beginning of each year or as otherwise determined appropriate. This annual cash incentive plan was established to motivate our executive officers to achieve short-term financial and business objectives, supporting our “pay-for-performance” culture and tying a significant portion of NEO compensation directly to Company achievements.
All NEOs are assigned annual bonus targets, expressed as a percentage of base salary, based on each NEO’s accountability, scope of responsibilities, and potential impact on performance, as well as peer group competitive data for similarly situated positions. Based on this review process, the Compensation Committee increased the annual bonus target for Dr. Henderson from 55% to 65% for the 2025 fiscal year. The target bonus percentage for Dr. Dambkowski and Ms. Henderson did not change between 2024 and 2025. The table below sets forth the target bonus for each NEO:
|
|
|
Name |
|
2025 Target Bonus
(% of Base Salary) |
Michael Henderson, M.D. |
|
65% |
Carl Dambkowski, M.D. |
|
45% |
Jane Pritchett Henderson |
|
45% |
The overall amount of the annual cash incentive plan pool each performance year is determined by the Compensation Committee and is generally based on (i) the individual’s target bonus, as a percentage of base salary and (ii) the percentage attainment of our corporate goals as established by the Compensation Committee, subject to the Compensation Committee’s discretion. Payouts under the annual cash incentive plan are not guaranteed, and the Compensation Committee may decide that payouts are as low as zero for a year of poor Company performance or may decide that payouts exceed target performance for a year of exceptional Company performance. For 2026, we have shifted the bonus weighting from 100% corporate to 80% corporate and 20% individual for the NEOs other than the CEO.
At the time the 2025 corporate objectives were determined, the Compensation Committee believed that payout at the target performance level was challenging but achievable and that payout above target represented true “stretch” performance goals. All awards are reviewed and approved by the Compensation Committee.
The pre-established corporate objectives for 2025 and their bonus payout are described in the table below. This table does not include the target levels with respect to the specific quantitative and qualitative objectives, as we believe disclosure of such information would result in competitive harm to the Company.
|
|
|
|
|
|
|
Category |
|
Goal(1) |
|
Weight |
|
Bonus
Payout |
Pipeline |
|
Advance programs fully into clinic, including zumilokibart (APG777) through critical first proof of concept trial and AD combination initiation |
|
70% |
|
90% |
Team |
|
Build organization embodying C.O.R.E values and attract/retain top talent |
|
10% |
|
12% |
Finance/Operations |
|
Manage resources and build capabilities to become leader in I&I space with line of sight to first launch, including through the maintenance of cash runway and strengthening of balance sheet |
|
20% |
|
28% |
(1)The annual cash bonuses relating to 2025 performance were paid in January 2026.
In reviewing the Company’s performance against the pre-established 2025 corporate objectives, in December 2025, the Compensation Committee approved a corporate goal achievement of 130%, which was based on: the Zumilokibart Program Achievements, the APG279 Program Achievements, the APG273 Program Achievement, the APG808 Program Achievement and the Finance Achievements.
The Board approved Dr. Henderson’s payout under the annual cash incentive plan at 130% of target. As a result, the annual cash incentive plan payouts approved by the Compensation Committee and the Board for 2025 were as follows:
|
|
|
|
|
Name |
|
2025 Annual
Cash Bonus |
|
Michael Henderson, M.D. |
|
$ |
591,500 |
|
Carl Dambkowski, M.D. |
|
$ |
315,900 |
|
Jane Pritchett Henderson |
|
$ |
315,900 |
|
Long-Term Incentive Compensation
Our equity grant program is intended to recognize the contributions of our NEOs to the achievement of corporate objectives, to create a greater sense of employee ownership, to align their interests with those of our stockholders by creating value tied to the performance of our stock price, and for retention purposes. In determining the form and value of an annual grant, the Compensation Committee considers the contributions and responsibilities of each NEO, appropriate incentives for the achievement of our long-term growth, the size and value of grants made to other executives at peer companies holding comparable positions, individual achievement of designated performance goals, and our overall performance relative to corporate objectives. The Compensation Committee also may grant equity awards to new executive officer hires or special awards upon promotion or for retention. We generally approve annual equity awards in the final month of each calendar year. In December 2024, we approved annual equity awards, which grants became effective at the time of grant in December 2024. In December 2025, we approved annual equity awards, which grants became effective in January 2026, which we expect to be the new grant schedule going forward.
Stock Options
For 2025, the Compensation Committee further determined that, after evaluating current compensation trends, reviewing the intended purpose of long-term equity incentive compensation, and considering the Company’s stage of development as a growing biopharmaceutical company and related market practice, the long-term equity incentive awards for NEOs should continue to consist solely of stock options. Stock options are inherently performance based, as they only provide value to the participant when the stock price appreciates over time, and typically have a four-year vesting schedule, which provides a retention component. The Compensation Committee continues to evaluate the appropriate mix of equity awards in light of the Company’s stage of development, business strategy, operational and
clinical development results, competitive market conditions in the industry, and Peer Group practices. In 2024, in consideration of the foregoing factors, and in recognition of 2024 performance, the Compensation Committee granted options to the NEOs in December 2024. And, in January 2026, the Compensation Committee granted options to the NEOs based on 2025 performance. As a result, the Company did not grant any stock options to the NEOs, and the Summary Compensation Table does not reflect stock option grants for 2025. The Company anticipates granting NEO equity awards at the beginning of 2027, in recognition of prior year 2026 performance and to incentivize future performance.
The table below sets forth the options granted to NEOs in 2024.
|
|
|
|
|
Name |
|
Options
(#) |
|
Michael Henderson, M.D. |
|
|
357,036 |
|
Carl Dambkowski, M.D. |
|
|
124,962 |
|
Jane Pritchett Henderson |
|
|
124,962 |
|
The table below sets forth the options granted to NEOs in 2026.
|
|
|
|
|
Name |
|
Options
(#) |
|
Michael Henderson, M.D. |
|
|
253,016 |
|
Carl Dambkowski, M.D. |
|
|
83,690 |
|
Jane Pritchett Henderson |
|
|
83,690 |
|
Each stock option represents the right to purchase one share of Common Stock at the option exercise price (once the option is vested). The stock options granted to the NEOs in 2024 based on 2024 performance and in 2026 based on 2025 performance vest over four years on a monthly basis, until fully vested on the fourth anniversary. The stock options expire 10 years after the date of the grant. This provides a reasonable timeframe during which the NEOs can benefit from the appreciation of the Company’s shares. The exercise price of the stock options was equal to the fair market value of the underlying stock on the date of grant.
Other Elements of Compensation
Benefits and perquisites
We offer participation in broad-based retirement, health and welfare plans to all of our employees. We pay the premiums for basic life and disability insurance for all of our employees, including our NEOs. All employees, including NEOs, are eligible to participate in a variety of ancillary benefits, including dental, vision, flexible spending accounts, health savings accounts, paid time off benefits, and an employee stock purchase plan. We currently maintain a retirement plan intended to provide benefits under section 401(k) of the Internal Revenue Code in which employees, including the NEOs, are allowed to contribute portions of their eligible compensation to a tax-qualified retirement account. Effective January 1, 2024, we established a 401(k) matching program which enables our employees, including the NEOs, to receive a dollar-for-dollar Company match of up to 4% of his or her compensation to the 401(k) fund, subject to limitations under applicable law. All of our employees are eligible to participate in the 401(k) plan on the same terms as our NEOs. We do not sponsor any qualified or non-qualified defined benefit plans for any of our employees or NEOs.
Employment Agreements
Each of the Company’s NEOs is covered by an employment agreement providing for a minimum annual level of salary, target incentives, and benefit eligibility. The agreements also provide for a severance benefit upon certain terminations of employment pursuant to the terms of the Executive Severance Policy, as described below under the subsection titled “Potential Payments Upon Termination or Change in Control.” It is the Compensation Committee’s belief that the employment agreements are necessary from a competitive perspective and contribute to the stability of the management team.
Other Policies
Clawback Policy
In July 2023, we adopted a clawback policy intended to comply with the requirements of Nasdaq Listing Standard 5608 implementing Rule 10D-1 under the Exchange Act. In the event the Company is required to prepare an accounting restatement of the Company’s financial statements due to material non-compliance with any financial reporting requirement under the federal securities laws, the Company will seek to recover, on a reasonably prompt basis, the excess incentive-based compensation received by any covered executive, including our NEOs, during the prior three fiscal years that exceeds the amount that the executive otherwise would have received had the incentive-based compensation been determined based on the restated financial statements.
In addition, in any instance in which, in the view of the Compensation Committee, a covered executive engaged in an act of fraud or misconduct that contributed to the need for a financial restatement, the Compensation Committee may, in its discretion, recover and the covered executive shall forfeit or repay, all of the covered executive’s incentive-based compensation for the relevant period, plus a reasonable rate of interest.
Insider Trading, Anti-Hedging, and Anti-Pledging Policy
We have adopted an insider trading policy governing the purchase, sale and other dispositions of our securities that applies our executive officers, directors, employees and certain designated consultants and contractors, as well as to the Company. We believe that our insider trading policy and repurchase procedures are reasonably designed to promote compliance with insider trading laws, rules and regulations, and listing standards applicable to us.
Certain transactions in our securities (such as purchases and sales of publicly traded put and call options, and short sales) create a heightened compliance risk or could create the appearance of misalignment between management and stockholders. In addition, securities held in a margin account or pledged as collateral may be sold without consent if the owner fails to meet a margin call or defaults on the loan, thus creating the risk that a sale may occur at a time when an officer or director is aware of material, non-public information or otherwise is not permitted to trade in Company securities. Our insider trading policy expressly prohibits covered persons from engaging in any short sales of our stock, any transactions involving publicly traded options or other derivatives and any hedging transactions. Our insider trading policy also expressly prohibits our officers and directors from pledging Company securities as collateral for a loan.
Equity Grant Policies and Practices
Stock option awards are approved by the Board or Compensation Committee each year in December, and, beginning with the December 2025 compensation decision cycle, are granted in January of the following year. Approval dates are normally set one year in advance based on the date of the fourth quarter Board meeting. Stock option awards may also be made to new executive officers upon hire or promotion, generally coincident with the date of hire for new executive officers or the last business day of the month in which the executive officer was promoted. While we do not maintain written policies on the timing of stock option awards, we have established processes to ensure that the timing of any stock option grants to the NEOs is not influenced by material non-public information (“MNPI”) about the Company, either positive or negative, and that all grant decisions are based on a predetermined schedule. The Compensation Committee considers factors like Company and employee performance and market conditions, regardless of any upcoming announcements or events that could impact our stock price. The Company has not timed the disclosure of MNPI for the purpose of affecting the value of executive compensation. The exercise price of options granted is equal to the closing market price of the Company’s common stock on the effective date of the grant.
The following table provides information regarding stock options granted to our NEOs in 2026 within four business days before the filing of a periodic report or current report disclosing MNPI or within one business day after the filing of such information.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Name |
|
Grant Date |
|
Number of securities underlying
the award |
|
|
Exercise price of
the award
($) |
|
|
Grant date fair value of the award
($) |
|
|
Percentage change in the closing market price of the securities underlying the award between the trading day ending immediately prior to the disclosure of material nonpublic information and the trading day beginning immediately following the disclosure of material nonpublic information |
Michael Henderson, M.D. |
|
January 2, 2026 |
|
|
253,016 |
|
|
$ |
75.78 |
|
|
$ |
12,999,229 |
|
|
1.86% |
Carl Dambkowski, M.D. |
|
January 2, 2026 |
|
|
83,690 |
|
|
$ |
75.78 |
|
|
$ |
4,299,750 |
|
|
1.86% |
Jane Pritchett Henderson |
|
January 2, 2026 |
|
|
83,690 |
|
|
$ |
75.78 |
|
|
$ |
4,299,750 |
|
|
1.86% |
(1)On January 6, 2026, we filed a Current Report on Form 8-K announcing positive interim data from the Phase 1b clinical trial of zumilokibart (APG777) for the treatment of patients with mild-to-moderate asthma and highlighting upcoming 2026 milestones.
Risk Management
During fiscal year 2025, our Compensation Committee reviewed our compensation policies in order to determine whether any such programs were likely to present a material risk to the Company. As part of its assessment, the Compensation Committee considered, among other things, our general compensation design philosophy, the total compensation levels and market data benchmarking, allocation of compensation among base salary and short- and long-term compensation, our approach to establishing Company-wide and individual performance targets, our equity plan design, including the emphasis on stock options as a significant compensation delivery vehicle, and our risk mitigation strategies. As a result of this review and analysis, the Compensation Committee determined that our policies and programs do not encourage excessive or inappropriate risk taking, and that they do not create risks that are reasonably likely to have a material adverse effect on the Company.
Compensation Committee Interlocks
None of the members of our Compensation Committee has at any time since our inception been one of our officers or employees. None of our executive officers currently serves, or in the past fiscal year has served, as a member of the board of directors or compensation committee of any entity that has one or more executive officers serving on our Board or Compensation Committee.
Compensation Committee Report
The Compensation Committee has reviewed and discussed with management the Compensation Discussion and Analysis required by Item 402(b) of Regulation S-K. Based on this review and discussion, the Compensation Committee recommended to the Board of Directors that the foregoing Compensation Discussion and Analysis be included in this Proxy Statement and incorporated by reference in our Annual Report on Form 10-K for the fiscal year ended December 31, 2025.
Submitted by the Compensation Committee of the Board of Directors
Mark C. McKenna, Chair
Jennifer Fox
William (BJ) Jones, Jr.
EXECUTIVE COMPENSATION TABLES
2025 Summary Compensation Table
The following table sets forth the total compensation that was awarded to, earned by or paid to our NEOs for services rendered during the years ended December 31, 2023, 2024 and 2025 (respectively, the “2023 Fiscal Year”, “2024 Fiscal Year” and the “2025 Fiscal Year”).
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Name and Principal Position |
|
Year |
|
Salary
($) |
|
|
Bonus
($)(1) |
|
|
Non-Equity Incentive Plan Compensation
($)(2) |
|
|
Options
($)(3) |
|
|
Stock
Awards
($)(4) |
|
|
Total
($) |
|
Michael Henderson, M.D. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chief Executive Officer |
|
2025 |
|
$ |
700,000 |
|
|
$ |
— |
|
|
$ |
591,500 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
1,291,500 |
|
|
|
2024 |
|
$ |
661,500 |
|
|
$ |
— |
|
|
$ |
545,738 |
|
|
$ |
12,000,444 |
|
|
$ |
— |
|
|
$ |
13,207,682 |
|
|
|
2023 |
|
$ |
564,833 |
|
|
$ |
606,375 |
|
|
$ |
— |
|
|
$ |
7,500,000 |
|
|
$ |
— |
|
|
$ |
8,671,208 |
|
Carl Dambkowski, M.D. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chief Medical Officer |
|
2025 |
|
$ |
540,000 |
|
|
$ |
— |
|
|
$ |
315,900 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
855,900 |
|
|
|
2024 |
|
$ |
525,000 |
|
|
$ |
— |
|
|
$ |
354,375 |
|
|
$ |
4,200,135 |
|
|
$ |
— |
|
|
$ |
5,079,510 |
|
|
|
2023 |
|
$ |
477,083 |
|
|
$ |
393,750 |
|
|
$ |
— |
|
|
$ |
3,300,000 |
|
|
$ |
— |
|
|
$ |
4,170,833 |
|
Jane Pritchett Henderson |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chief Financial Officer |
|
2025 |
|
$ |
540,000 |
|
|
$ |
— |
|
|
$ |
315,900 |
|
|
$ |
— |
|
|
$ |
— |
|
|
$ |
855,900 |
|
|
|
2024 |
|
$ |
525,000 |
|
|
$ |
— |
|
|
$ |
354,375 |
|
|
$ |
4,200,135 |
|
|
$ |
— |
|
|
$ |
5,079,510 |
|
|
|
2023(5) |
|
$ |
469,311 |
|
|
$ |
374,332 |
|
|
$ |
— |
|
|
$ |
3,300,000 |
|
|
$ |
881,270 |
|
|
$ |
5,024,913 |
|
(1)Amounts in this column include, for each NEO, discretionary annual bonuses with respect to the 2023 Fiscal Year, as determined in accordance with the terms of their respective employment agreements and based on the Board’s assessment of each NEO’s individual performance as well as the Board’s assessment of overall Company performance.
(2)Amounts in this column represent cash bonus awards earned by our NEOs pursuant to an annual cash incentive plan, based on pre-determined corporate performance goals.
(3)Amounts in this column represent the aggregate grant date fair value of stock options, computed in accordance with Financial Accounting Standards Board (FASB) Accounting Standards Codification Topic 718 (ASC Topic 718), rather than the amounts paid to or realized by the named individual. We provide information regarding the assumptions used to calculate the value of these awards in Note 2 to our audited consolidated financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2025. These amounts do not reflect the actual economic value that may be realized by the NEOs.
(4)Prior to our IPO in July 2023, we granted Ms. Henderson incentive units under the Second Amended and Restated Limited Liability Company Agreement of Apogee Therapeutics, LLC (the “LLC Agreement”), the economics of which are similar to stock options. The amounts disclosed represent the aggregate grant date fair value of incentive units granted under the LLC Agreement during the indicated fiscal year computed in accordance with ASC Topic 718. The assumptions used in calculating the grant date fair value of the incentive units are set forth in the notes to our audited consolidated financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2023. These amounts do not reflect the actual economic value that may be realized by the NEOs.
(5)Ms. Henderson was appointed as our Chief Financial Officer in January 2023. As such, the amounts reported for Ms. Henderson are pro-rated to reflect her commencement date as the Chief Financial Officer of the Company.
2025 Grants of Plan-Based Awards Table
The following table sets forth certain information with respect to 2025 plan-based awards granted to the NEOs during the year ended December 31, 2025. Due to the timing of our long-term incentive grants, no options were granted to our NEOs in 2025.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Estimated Future Payments Under Non-Equity Incentive
Plan Awards (1) |
|
Name |
|
Threshold
($) |
|
|
Target
($) |
|
|
Maximum
($) |
|
Michael Henderson, M.D. |
|
$ |
— |
|
|
$ |
455,000 |
|
|
$ |
— |
|
Carl Dambkowski, M.D. |
|
$ |
— |
|
|
$ |
243,000 |
|
|
$ |
— |
|
Jane Pritchett Henderson |
|
$ |
— |
|
|
$ |
243,000 |
|
|
$ |
— |
|
(1)Represents the target annual cash bonus payouts pursuant to our annual cash incentive plan and based on pre-determined corporate performance goals. There are no threshold or maximum levels set under the program. Please see the description of the annual cash incentive plan under “Compensation Discussion and Analysis - Elements of Executive Compensation - Annual Cash Bonus”.
Outstanding Equity Awards at 2025 Fiscal Year-End Table
The following table summarizes equity awards held by our NEOs as of the end of the 2025 Fiscal Year. Prior to the IPO, our NEOs each held incentive units pursuant to the LLC Agreement, which were exchanged for shares of common stock (with respect to vested incentive units) or restricted common stock awards (with respect to unvested incentive units) in connection with our IPO. Each of the equity awards in the table below may be subject to accelerated vesting, as described below under the subsection titled “Additional Narrative Disclosure - Potential Payments Upon Termination or Change in Control.”
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock Options |
|
Stock Awards |
|
Name |
|
Grant
Date |
|
Number of
Securities
Underlying
Unexercised
Options
Exercisable
(#) |
|
|
Number of
Securities
Underlying
Unexercised
Options
Unexercisable
(#) |
|
Option
Exercise
Price |
|
|
Option
Expiration
Date |
|
Number of
Shares
That Have
Not
Vested |
|
Market
Value of
Shares
That Have
Not Vested
($) |
|
Michael Henderson, M.D. |
|
12/18/2023 |
|
|
190,756 |
|
|
199,256(1) |
|
$ |
22.86 |
|
|
12/18/2033 |
|
|
|
|
|
|
|
12/9/2024 |
|
|
89,259 |
|
|
267,777(2) |
|
$ |
49.07 |
|
|
12/9/2034 |
|
|
|
|
|
|
|
10/3/2022 |
|
|
|
|
|
|
|
|
|
|
|
60,497(3) |
|
$ |
4,566,314 |
|
|
|
10/17/2022 |
|
|
|
|
|
|
|
|
|
|
|
64,724(3) |
|
$ |
4,885,368 |
|
|
|
12/21/2022 |
|
|
|
|
|
|
|
|
|
|
|
71,845(4) |
|
$ |
5,422,861 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Carl Dambkowski, M.D. |
|
12/18/2023 |
|
|
65,892 |
|
|
87,673(1) |
|
$ |
22.86 |
|
|
12/18/2033 |
|
|
|
|
|
|
|
12/9/2024 |
|
|
31,240 |
|
|
93,722(2) |
|
$ |
49.07 |
|
|
12/9/2034 |
|
|
|
|
|
|
|
10/3/2022 |
|
|
|
|
|
|
|
|
|
|
|
24,749(5) |
|
$ |
1,868,055 |
|
|
|
12/21/2022 |
|
|
|
|
|
|
|
|
|
|
|
42,199(6) |
|
$ |
3,185,181 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Jane Pritchett Henderson |
|
12/18/2023 |
|
|
87,672 |
|
|
87,673(1) |
|
$ |
22.86 |
|
|
12/18/2033 |
|
|
|
|
|
|
|
12/9/2024 |
|
|
31,240 |
|
|
93,722(2) |
|
$ |
49.07 |
|
|
12/9/2034 |
|
|
|
|
|
|
|
2/1/2023 |
|
|
|
|
|
|
|
|
|
|
|
60,338(7) |
|
$ |
4,554,312 |
|
(1)Shares subject to the option vest in 48 monthly installments beginning on December 18, 2023, subject to the recipient’s continued service to us through each applicable vesting date.
(2)Shares subject to the option vest in 48 monthly installments beginning on December 9, 2024, subject to the recipient’s continued service to us through each applicable vesting date.
(3)25% of the awards vested on May 2, 2023, with the remainder vesting in equal monthly installments until May 2026, subject to Dr. Henderson’s continued service to us through each applicable vesting date.
(4)25% of the awards vested on December 14, 2023, with the remainder vesting in equal monthly installments until December 2026, subject to Dr. Henderson’s continued service to us through each applicable vesting date.
(5)25% of the awards vested on September 16, 2023, with the remainder vesting in equal monthly installments until September 2026, subject to Dr. Dambkowski’s continued service to us through each applicable vesting date.
(6)25% of the awards vested on December 14, 2023, with the remainder vesting in equal monthly installments until December 2026, subject to Dr. Dambkowski’s continued service to us through each applicable vesting date.
(7)25% of the awards vested on February 1, 2024, with the remainder vesting in equal monthly installments until February 2027, subject to Ms. Henderson’s continued service to us through each applicable vesting date.
Option Exercises and Stock Vested Table
The following table sets forth information regarding the option awards exercised and stock awards that vested for our NEOs during fiscal 2025.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Option Awards |
|
|
Stock Awards |
|
Name |
|
Number of Shares Acquired on Exercise
(#)(1) |
|
|
Value Realized on Exercise
($)(2) |
|
|
Number of Shares Acquired on Vesting
(#) |
|
|
Value Realized on Vesting
($)(3) |
|
Michael Henderson, M.D. |
|
|
8,500 |
|
|
$ |
157,250 |
|
|
|
372,372 |
|
|
$ |
16,256,623 |
|
Carl Dambkowski, M.D. |
|
|
19,965 |
|
|
$ |
509,916 |
|
|
|
75,197 |
|
|
$ |
3,377,066 |
|
Jane Pritchett Henderson |
|
|
— |
|
|
$ |
— |
|
|
|
51,718 |
|
|
$ |
2,210,359 |
|
(1)This column reflects the number of shares acquired on exercise of stock options during the fiscal year ended December 31, 2025.
(2)Value realized does not represent proceeds from any sale of any common stock acquired upon exercise, but is determined by multiplying the number of shares acquired upon exercise by the difference between the exercise price of the option and the closing price of our common stock on each exercise date.
(3)Value realized does not represent proceeds from any sale of commons stock, but is determined by multiplying the number of shares subject to restricted stock awards that vested by the closing price of our common stock on the vesting date.
Potential Payments Upon Termination or Change in Control
The table below reflects the amount of compensation that would become payable to each of the NEOs under existing plans and arrangements if that NEO’s employment had terminated on December 31, 2025 (pursuant to the executive’s employment agreement then in effect) and/or a change of control had occurred on such date, given the NEO’s compensation levels as of such date and, if applicable, based on the Company’s closing stock price on that date of $75.48. These benefits are in addition to benefits available prior to the occurrence of any termination of employment, including benefits generally available to salaried employees, such as distributions under the Company’s 401(k) plan. The actual amounts that would be paid upon an NEO’s termination of employment can be determined only at the time of such NEO’s separation from the Company. Due to the number of factors that affect the nature and amount of any benefits provided upon the events discussed below, any actual amounts paid or distributed may be higher or lower than reported below.
Executive Severance Policy
We maintain an executive severance policy that covers Dr. Henderson, Dr. Dambkowski, and Ms. Henderson (as amended, the “Executive Severance Policy”). Potential payments in connection with a change in control to each of Dr. Henderson, Dr. Dambkowski, and Ms. Henderson are governed by the Executive Severance Policy.
Under the Executive Severance Policy, Dr. Henderson is eligible to receive upon a termination by us without cause (as defined below) or a resignation for good reason (as defined below) that is not within the change in control period (as defined below): (i) 1.5 times his annual base salary, (ii) payment of any bonus amount earned but unpaid for the year prior to the year of termination, (iii) payment of a pro-rata portion of the target bonus that he would have earned for the year in which the termination occurs, (iv) subsidized continued health coverage for up to 18 months, and (v) the immediate acceleration of 30% of his equity-based awards. In addition, upon a termination by us without cause or a resignation for good reason, occurring within the change in control period, Dr. Henderson will be eligible to receive: (i) 1.5 times his annual base salary, (ii) payment of any bonus amount earned but unpaid for the year prior to the year of termination, (iii) payment of the full target bonus he would have earned for the year in which the termination occurs, (iv) subsidized continued health coverage for up to 18 months, and (v) the immediate acceleration of 100% of his equity-based awards.
Under the Executive Severance Policy, Dr. Dambkowski and Ms. Henderson are eligible to receive upon a termination by us without cause or a resignation for good reason (as defined below) that is not within the change in control period: (i) 1.0 times annual base salary, (ii) payment of any bonus amount earned but unpaid for the year prior to the year of termination, (iii) payment of a pro-rata portion of the target bonus that he or she would have earned for the year in which the termination occurs, and (iv) subsidized continued health coverage for up to 12 months. In addition, upon a termination by us without cause or a resignation for good reason, occurring within the change in control period, Dr. Dambkowski and Ms. Henderson will be eligible to receive the benefits listed in items (i), (ii), and (iv) and the payment of the full target bonus he or she would have earned for the year in which the termination occurs and the immediate acceleration of 100% of his or her equity-based awards.
For purposes of the Executive Severance Policy, the following definitions apply:
•“Cause” means the NEO’s (i) dishonest statements or acts with respect to the Company or any affiliate of the Company, or any current or prospective customers, suppliers, vendors or other third parties with which such entity does business that results in or is reasonably anticipated to result in material harm to the Company; (ii) commission of (A) a felony or (B) any misdemeanor involving moral turpitude, deceit, dishonesty or fraud; (iii) failure to perform in all material respects his or her assigned duties and responsibilities to the reasonable satisfaction of the Board, which failure continues, in the reasonable judgment of the Board, for thirty (30) days after written notice given to the NEO describing such failure; (iv) gross negligence, willful misconduct that results in or is reasonably anticipated to result in harm to the Company; or (v) violation of any material provision of any agreement(s) between the NEO and the Company or any Company policies including, without limitation, agreements relating to noncompetition, non-solicitation, nondisclosure and/or assignment of inventions or policies related to ethics or workplace conduct and such violation, if curable, is not cured within thirty (30) days after the Company provides written notice to the NEO of such violation.
•“Change in Control” means (i) any person is or becomes the beneficial owner, directly or indirectly, of securities of the Company (not including the securities beneficially owned by such person or any securities acquired directly from the Company or its affiliates) representing 50% or more of the combined voting power of the Company’s then outstanding securities, excluding any person who becomes such a beneficial owner in connection with a transaction described in (iii) below; (ii) the following individuals cease for any reason to constitute a majority of the number of directors then serving: (A) individuals who, on the effective date of the 2023 Plan, constitute the Board and (B) any new director (other than a director whose initial assumption of office is in connection with an actual or threatened election contest, including a consent solicitation, relating to the election of directors of the Company) whose appointment or election by the Board or nomination for election by the Company’s stockholders was approved or recommended by a vote of at least a majority of the directors then still in office who were either directors on the effective date of the 2023 Plan or whose appointment, election or nomination for election was previously so approved or recommended; (iii) there is consummated a merger or consolidation of the Company or any
direct or indirect subsidiary of the Company with any other entity, other than a merger or consolidation which would result in the holders of the voting securities of the Company outstanding immediately prior to such merger or consolidation continuing to represent (either by remaining outstanding or by being converted into voting securities of the surviving entity or any parent thereof) at least 50% of the combined voting power of the securities of the Company or such surviving entity or any parent thereof outstanding immediately after such merger or consolidation; or (iv) the implementation of a plan of complete liquidation or dissolution of the Company; or (v) there is consummated a sale or disposition by the Company of all or substantially all of the Company’s assets, other than a sale or disposition by the Company of all or substantially all of the Company’s assets to an entity, at least 50% of the combined voting power of the voting securities of which is owned by stockholders of the Company in substantially the same proportions as their ownership of the Company immediately prior to such sale.
•“Change in Control Period” means the three (3) month period immediately before and the twelve (12) month period that immediately follows the first event constituting a Change in Control.
•For Dr. Henderson only, “Good Reason” means the occurrence of any of the following (both within and outside of the Change in Control Period): (i) a material diminution in Dr. Henderson’s base salary or target bonus except for across-the-board salary and target bonus reductions based on the Company’s financial performance similarly affecting all or substantially all senior management employees of the Company; or (ii) a material change in the geographic location at which Dr. Henderson provides services to the Company; or (iii) a material reduction in Dr. Henderson’s duties, authority or responsibilities, but excluding any change in title that does not represent a material reduction in Dr. Henderson’s duties, authority or responsibilities; or (iv) the failure of the Company to obtain the assumption of Dr. Henderson’s amended and restated employment agreement by a successor; or (v) the material breach of Dr. Henderson’s amended and restated employment agreement by the Company; or (vi) a requirement by the Company that Dr. Henderson’s primary work location shall be in-office when remote work is feasible and does not impair Dr. Henderson’s ability to perform his duties.
•For Dr. Dambkowski and Ms. Henderson only, “Good Reason” means (A) with respect to a resignation by the NEO outside of the Change in Control Period: (i) the material breach of such NEO’s written employment or services agreement by the Company or (ii) a requirement by the Company that such NEO’s primary work location shall be in-office when remote work is feasible and does not impair such NEO’s ability to perform such NEO’s duties or (B) with respect to a resignation by the NEO within the Change in Control Period: (i) a material diminution in such NEO’s base salary or target bonus except for across-the-board salary and target bonus reductions based on the Company’s financial performance similarly affecting all or substantially all senior management employees of the Company; or (ii) a material change in the geographic location at which such NEO provides services to the Company; or (iii) a material reduction in such NEO’s duties, authority or responsibilities, but excluding any change in title that does not represent a material reduction in such NEO’s duties, authority or responsibilities; or (iv) the failure of the Company to obtain the assumption of such NEO’s written employment or services agreement by a successor; or (v) the material breach of such NEO’s written employment or services agreement by the Company; or (vi) a requirement by the Company that such NEO’s primary work location shall be in-office when remote work is feasible and does not impair such NEO’s ability to perform such NEO’s duties.
|
|
|
|
|
|
|
|
|
|
|
Involuntary Termination (Without Cause or for Good Reason) not in Connection with a Change in Control |
|
|
Involuntary Termination (Without Cause or for Good Reason) in Connection with a Change in Control |
|
Name |
|
($) |
|
|
($) |
|
Michael Henderson, M.D. |
|
|
|
|
|
|
Base Salary |
|
$ |
1,050,000 |
|
|
$ |
1,050,000 |
|
Continuation of Health Coverage |
|
|
45,706 |
|
|
|
45,706 |
|
Acceleration of RSUs/RSAs (1) |
|
|
4,462,363 |
|
|
|
14,874,542 |
|
Acceleration of Option Awards(1) |
|
|
5,267,052 |
|
|
|
17,556,841 |
|
Total |
|
$ |
10,825,121 |
|
|
$ |
33,527,089 |
|
Carl Dambkowski, M.D. |
|
|
|
|
|
|
Base Salary |
|
$ |
540,000 |
|
|
$ |
540,000 |
|
Continuation of Health Coverage |
|
|
24,739 |
|
|
|
24,739 |
|
Acceleration of RSUs/RSAs (1) |
|
|
— |
|
|
|
5,053,235 |
|
Acceleration of Option Awards(1) |
|
|
— |
|
|
|
7,088,551 |
|
Total |
|
$ |
564,739 |
|
|
$ |
12,706,525 |
|
Jane Pritchett Henderson |
|
|
|
|
|
|
Base Salary |
|
$ |
540,000 |
|
|
$ |
540,000 |
|
Continuation of Health Coverage |
|
|
11,545 |
|
|
|
11,545 |
|
Acceleration of RSUs/RSAs (1) |
|
|
— |
|
|
|
4,554,312 |
|
Acceleration of Option Awards(1) |
|
|
— |
|
|
|
7,088,551 |
|
Total |
|
$ |
551,545 |
|
|
$ |
12,194,408 |
|
(1)The value of the acceleration of equity awards is calculated based on the closing price of our common stock on December 31, 2025, which was $75.48 per share, as reported on Nasdaq. The value of the RSU awards with accelerated vesting is calculated based on that closing market price for unvested RSUs on December 31, 2025. The value of the option awards with accelerated vesting is calculated as the amount by which that closing market price exceeds the exercise price for unvested stock options on December 31, 2025.
CEO PAY RATIO
Pursuant to Item 402(u) of Regulation S-K, we are required to calculate and disclose the annual total compensation of our median employee (excluding our CEO, Dr. Henderson), the annual total compensation of Dr. Henderson, and the ratio of these two amounts.
Our median employee was identified using the entire population of our employees as of December 31, 2025 based on a consistently applied compensation measure (“CACM”), that reasonably reflects the annual compensation of our employees. The CACM selected by us for our disclosure included the total cash compensation and the grant-date fair value for equity awards granted in fiscal 2025.
Based on the CACM methodology described above, we identified the median employee and calculated the fiscal 2025 compensation for this selected employee in the same manner we determine the annual total compensation of our NEOs for purposes of the Summary Compensation Table. The median employee’s fiscal 2025 annual total compensation was $283,326. Dr. Henderson’s fiscal 2025 annual total compensation as disclosed in the 2025 Summary Compensation Table was $1,291,500. As a result, our CEO to median employee pay ratio for fiscal 2025 is 5:1.
This pay ratio is a reasonable estimate calculated in a manner consistent with the SEC requirements, described above, and based on our payroll and employment records. As a result of a variety of factors, including employee populations, potential differences in the components used for the CACM, compensation philosophies and certain assumptions, pay ratios reported by other companies may not be comparable to our pay ratio. The pay ratio is not utilized by our management or our Compensation Committee for compensation-related decisions.
PAY FOR PERFORMANCE
Our Compensation Committee approves and administers our executive compensation program to align executive compensation with stockholder interests by linking pay to performance. Our overall compensation program includes a mix of short-term and long-term components through our annual cash incentive plan and our equity incentive awards. As required by Item 402(v) of Regulation S-K, we are providing the following information about the relationship between the compensation actually paid to our NEOs and certain aspects of our financial performance. For purposes of this discussion, our CEO is also referred to as our principal executive officer or “PEO” and our other NEOs are referred to as our “Non-PEO NEOs”.
As described in more detail in “Executive Compensation—Compensation Discussion & Analysis”, the Company’s annual incentive program does not utilize a quantifiable financial performance metric that is directly linked to compensation; therefore, no Company-Selected Measure is presented and no tabular list of financial performance measures is presented.
During our fiscal year ending December 31, 2024, we were a smaller reporting company pursuant to Rule 405 of the Securities Act. For the current fiscal year, we are filing as a Large Accelerated Filer and have included the required disclosures accordingly:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Fiscal Year |
|
Summary Compensation Table Total for PEO(1),(2) |
|
Compensation Actually Paid to PEO(1),(3) |
|
Average Summary Compensation Table Total for Non-PEO NEOs(1),(2) |
|
Average Compensation Actually Paid to Non-PEO NEOs(1),(3) |
|
Total Shareholder Return Value of an initial $100 Investment(4) |
|
Peer Group Total Shareholder Return(5) |
|
Net Income (thousands)(6) |
|
2025 |
|
$ |
1,291,500 |
|
$ |
15,951,124 |
|
$ |
855,900 |
|
$ |
6,424,794 |
|
$ |
356 |
|
$ |
142 |
|
$ |
(255,843 |
) |
2024 |
|
$ |
13,207,682 |
|
$ |
35,067,552 |
|
$ |
5,079,510 |
|
$ |
10,862,926 |
|
$ |
213 |
|
$ |
106 |
|
$ |
(182,146 |
) |
2023 |
|
$ |
8,671,208 |
|
$ |
35,328,071 |
|
$ |
4,597,874 |
|
$ |
10,813,019 |
|
$ |
132 |
|
$ |
107 |
|
$ |
(83,985 |
) |
(1)NEOs included in these columns reflect the following individuals:
|
|
|
|
|
Fiscal Year |
|
PEO |
|
Non-PEO NEOs |
2025 |
|
Michael Henderson, M.D. |
|
Carl Dambkowski, M.D.,
Jane Pritchett Henderson |
2024 |
|
Michael Henderson, M.D. |
|
Carl Dambkowski, M.D.,
Jane Pritchett Henderson |
2023 |
|
Michael Henderson, M.D. |
|
Carl Dambkowski, M.D.,
Jane Pritchett Henderson |
(2)Amounts reflect Summary Compensation Table Total Pay for our NEOs for each corresponding year. Please refer to “Executive Compensation – Summary Compensation Table” above.
(3)Compensation Actually Paid (CAP) has been calculated based on the requirements and methodology set forth in the applicable SEC rules (Item 402(v) of Regulation S-K). The CAP calculation includes the end-of-year value of awards granted within the fiscal year, the change in fair value from prior year end of vested awards and the change in the fair value of unvested awards granted in prior years, regardless of if, when, or at which intrinsic value they will actually vest. To calculate CAP the following amounts were deducted from or added to the total compensation number shown in the Summary Compensation Table (SCT):
|
|
|
|
|
Reconciliation of Summary Compensation Table Total to Compensation Actually Paid for PEO |
|
2025 |
|
Summary Compensation Table Total |
|
$ |
1,291,500 |
|
(Minus): Grant Date Fair Value of Equity Awards Granted in the Fiscal Year |
|
|
— |
|
Plus: Fair Value at Fiscal Year End of Outstanding and Unvested Equity Awards Granted in the Fiscal Year |
|
|
— |
|
Plus/(Minus): Change in Fair Value From the end of the Prior Fiscal Year to the End of the Fiscal Year of Outstanding and Unvested Equity Awards Granted in Prior Fiscal Years |
|
|
15,520,579 |
|
Plus/(Minus): Change in Fair Value as of the Vesting Date of Equity Awards Granted in Prior Fiscal Years that Vested in the Fiscal Year |
|
|
(860,955 |
) |
Compensation Actually Paid |
|
$ |
15,951,124 |
|
|
|
|
|
|
Reconciliation of Average Summary Compensation Table Total to Average Compensation Actually Paid for Non-PEO NEOs |
|
2025 |
|
Summary Compensation Table Total |
|
$ |
855,900 |
|
(Minus): Grant Date Fair Value of Equity Awards Granted in the Fiscal Year |
|
|
— |
|
Plus: Fair Value at Fiscal Year End of Outstanding and Unvested Equity Awards Granted in the Fiscal Year |
|
|
— |
|
Plus/(Minus): Change in Fair Value From the end of the Prior Fiscal Year to the End of the Fiscal Year of Outstanding and Unvested Equity Awards Granted in Prior Fiscal Years |
|
|
5,740,621 |
|
Plus/(Minus): Change in Fair Value as of the Vesting Date of Equity Awards Granted in Prior Fiscal Years that Vested in the Fiscal Year |
|
|
(171,727 |
) |
Compensation Actually Paid |
|
$ |
6,424,794 |
|
For purposes of the above adjustments, the fair value of equity awards on the applicable date were determined in accordance with FASB’s ASC Topic 718, using valuation methodologies that are generally consistent with those used to determine the grant-date fair value for accounting purposes.
(4)Total Shareholder Return (TSR) represents the cumulative return on a fixed investment of $100 in the Company’s common stock, for the period beginning on July 14, 2023, the date our common stock commenced trading on the Nasdaq Global Market, through the end of the applicable fiscal year, assuming reinvestment of dividends.
(5)Peer Group Total Shareholder Return represents the cumulative return on a fixed investment of $100 in the Nasdaq Biotechnology Index for the period beginning on July 14, 2023, the date our common stock commenced trading on the Nasdaq Global Market, assuming reinvestment of dividends.
(6)The dollar amounts reported represent the net income reflected in the Company’s audited financial statements for the applicable year and calculated in accordance with GAAP.
Relationship between CAP vs. Cumulative TSR of Company
In accordance with Item 402(v) of Regulation S-K, the following chart illustrates the relationship between CAP for our PEO and the average CAP for our Non-PEO NEOs against the Company’s TSR:
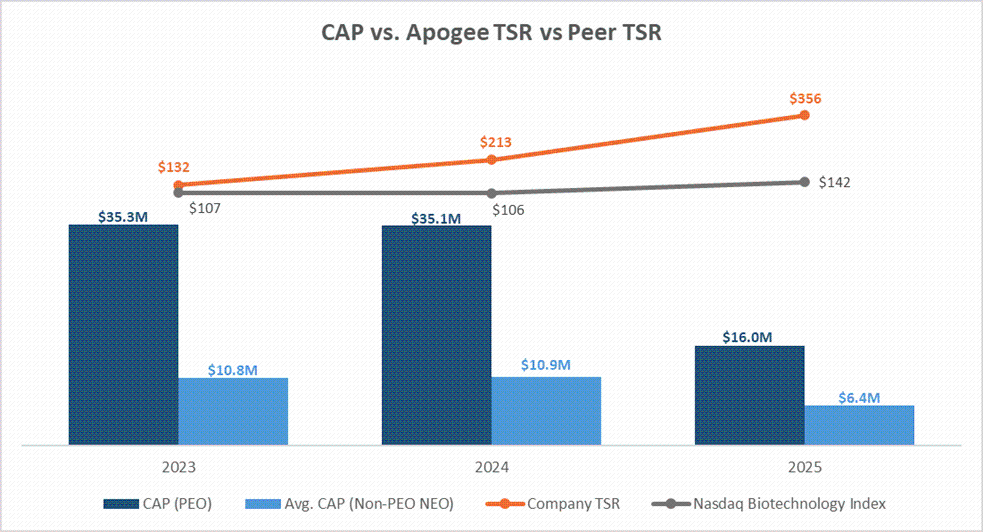
Relationship between CAP vs. Net Income
In accordance with Item 402(v) of Regulation S-K, the following chart illustrates the relationship between CAP for our PEO and the average CAP for our Non-PEO NEOs against the Company’s net income:
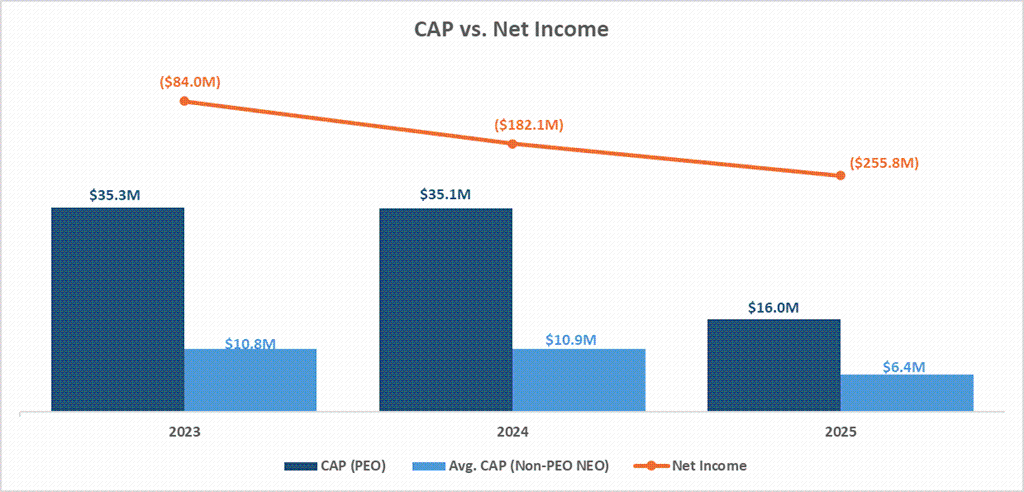
CERTAIN INFORMATION ABOUT OUR COMMON STOCK
Security Ownership of Certain Beneficial Owners and Management
The following table presents information regarding beneficial ownership of our equity interests as of April 1, 2026 by:
•each stockholder or group of stockholders known by us to be the beneficial owner of more than 5% of our common stock and non-voting common stock;
•each of our directors and nominees;
•all of our current directors and executive officers as a group.
Beneficial ownership is determined in accordance with the rules of the SEC, and thus represents voting or investment power with respect to our securities as of April 1, 2026. Under such rules, beneficial ownership includes any shares of common stock over which the individual or entity has sole or shared voting power or investment power as well as any shares of common stock that the individual or entity has the right to acquire within 60 days after the date of this table through the exercise of any stock option, warrant or other right. To our knowledge and subject to applicable community property rules, and except as otherwise indicated below, the persons and entities named in the table have sole voting and sole investment power with respect to all equity interests beneficially owned.
The percentage ownership information shown in the column titled “Total Percentage Ownership” in the table below is based on 75,327,868 shares of our common stock outstanding, including 433,457 unvested shares of restricted common stock and 13,486,642 shares of non-voting common stock as of the date of this table (plus, as to any particular beneficial owner, any shares as to which such person has the right to acquire beneficial ownership within 60 days thereafter). The percentage ownership information shown in the column titled “Voting Power” in the table below is based on 61,841,226 shares of our voting common stock outstanding. Unless otherwise indicated, the address of each individual listed in this table is the Company’s address set forth on the first page of this Proxy Statement.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Name of Beneficial Owner |
|
Number of
Shares of Voting
Common Stock
Owned |
|
|
Number of
Shares of
Non-Voting
Common
Stock Owned |
|
|
Total
Percentage
Ownership(1) |
|
|
Voting
Power(2) |
|
Greater than 5% Stockholders: |
|
|
|
|
|
|
|
|
|
|
|
|
T. Rowe Price Investments Management, Inc.(3) |
|
|
10,291,220 |
|
|
|
— |
|
|
|
13.7 |
% |
|
|
16.6 |
% |
Entities affiliated with FMR LLC(4) |
|
|
6,907,100 |
|
|
|
— |
|
|
|
9.2 |
% |
|
|
11.2 |
% |
Entities affiliated with Wellington Management Group LLP(5) |
|
|
4,746,585 |
|
|
|
— |
|
|
|
6.3 |
% |
|
|
7.7 |
% |
Entities affiliated with Venrock Healthcare Capital Partners III, L.P.(6) |
|
|
2,115,853 |
|
|
|
6,743,321 |
|
|
|
11.7 |
% |
|
3.4%^ |
|
Entities affiliated with Fairmount Funds Management LLC(7) |
|
|
298,647 |
|
|
|
6,743,321 |
|
|
|
9.3 |
% |
|
*^ |
|
Named Executive Officers, Directors and Nominees: |
|
|
|
|
|
|
|
|
|
|
|
|
Michael Henderson, M.D.(8) |
|
|
1,532,788 |
|
|
|
— |
|
|
|
2.0 |
% |
|
|
2.5 |
% |
Carl Dambkowski, M.D.(9) |
|
|
317,386 |
|
|
|
— |
|
|
* |
|
|
* |
|
Jane Pritchett Henderson(10) |
|
|
334,539 |
|
|
|
— |
|
|
* |
|
|
* |
|
Mark C. McKenna(11) |
|
|
167,872 |
|
|
|
— |
|
|
* |
|
|
* |
|
Lisa Bollinger, M.D.(12) |
|
|
20,478 |
|
|
|
— |
|
|
* |
|
|
* |
|
Jennifer Fox(13) |
|
|
45,194 |
|
|
|
— |
|
|
* |
|
|
* |
|
Andrew Gottesdiener, M.D.(14) |
|
|
42,208 |
|
|
|
— |
|
|
* |
|
|
* |
|
Peter Harwin(7)(15) |
|
|
392,021 |
|
|
|
6,743,321 |
|
|
|
9.5 |
% |
|
*^ |
|
William (BJ) Jones, Jr.(16) |
|
|
45,194 |
|
|
|
— |
|
|
* |
|
|
* |
|
Tomas Kiselak(7)(17) |
|
|
392,021 |
|
|
|
6,743,321 |
|
|
|
9.5 |
% |
|
*^ |
|
Nimish Shah(6)(18) |
|
|
2,158,061 |
|
|
|
6,743,321 |
|
|
|
11.8 |
% |
|
3.5%^ |
|
All current executive officers and directors as a group (11 persons)(19) |
|
|
5,149,115 |
|
|
|
13,486,642 |
|
|
|
24.3 |
% |
|
|
8.1 |
% |
* Represents beneficial ownership of less than one percent.
^ Entities affiliated with Fairmount Funds Management LLC and entities affiliated with Venrock Healthcare Capital Partners III, L.P. each beneficially own the shares of common stock underlying their non-voting common stock, subject to an ownership limitation of 9.99% of outstanding common stock. Accordingly, such entities have the ability to convert their shares of non-voting common stock into common stock, and thereby increase their voting power, subject to such ownership limitation.
(1)Calculated based on the sum of “Number of Shares of Voting Common Stock Owned” and “Number of Shares of Non-Voting Common Stock Owned,” divided by the sum of (1) the number of shares of voting common stock and non-voting common stock outstanding as of the date of this table, and (2) the number of shares of common stock that a person has the right to acquire within 60 days after the date of this table (which includes the number of shares of non-voting common stock owned by such person to the extent they can be converted to common stock within 60 days after the date of this table).
(2)Calculated based on “Number of Shares of Voting Common Stock Owned” divided by the number of shares of voting common stock outstanding as of the date of this table, excluding unvested shares of restricted common stock units.
(3)Based on a Schedule 13G filed on April 8, 2026. Consists of 10,291,220 shares of common stock held in the aggregate T. Rowe Price Capital Appreciation Fund, of which T. Rowe Price Investment Management, Inc. is the investment adviser, holds the securities represented in the table above in their investment portfolio managed by T. Rowe Price Investment Management, Inc. and such funds have the right to receive or the power to direct the receipt of dividends from, or the proceeds from the sale of, the securities that they hold. T. Rowe Price Capital Appreciation Fund has an interest in 5,295,932 of the shares of common stock held. The address of T. Rowe Price Investment Management, Inc. is 1307 Point Street, Baltimore, MD 21231.
(4)Based on a Schedule 13G/A filed on August 6, 2025. The securities represented in the table above are owned by funds or accounts managed by direct or indirect subsidiaries of FMR LLC and are beneficially owned, or may be deemed to be beneficially owned, by FMR LLC. Abigail P. Johnson is a Director, the Chairman and the Chief Executive Officer of FMR LLC. The address of FMR LLC is 245 Summer Street, Boston, MA 02210.
(5)Based on a Schedule 13G/A filed on February 10, 2026. Consists of 4,746,585 shares of common stock owned by clients of investment advisers directly or indirectly owned by Wellington Management Group LLP. The address of the entities is c/o Wellington Management Company LLP, 280 Congress Street, Boston, MA 02210.
(6)Based on a Schedule 13D filed on July 28, 2023 and Company records. Consists of (i) 460,075 shares of common stock and 2,495,319 shares of non-voting common stock held by Venrock Healthcare Capital Partners III, L.P. (“VHCP III”); (ii) 46,025 shares of common stock and 249,522 shares of non-voting common stock held by VHCP Co-Investment Holdings III, LLC (“VHCP Co-III”); (iii) 1,243,900 shares of common stock and 3,998,480 shares of non-voting common stock held by Venrock Healthcare Capital Partners EG, L.P (“VHCP EG”); (iv) and pre-funded warrants to purchase up to 365,853 shares of common stock held by VHCP EG. VHCP Management III, LLC (“VHCPM III”) is the sole general partner of VHCP III and the sole manager of VHCP Co-III. VHCP Management EG, LLC (“VHCPM EG”) is the sole general partner of VHCP EG. Dr. Bong Koh and Nimish Shah are the voting members of VHCPM III and VHCPM EG. Dr. Koh, Mr. Shah, VHCPM III and VHCPM EG disclaim beneficial ownership over all shares held by VHCP III, VHCP Co-III, and VHCP EG, except to the extent of their respective indirect pecuniary interests therein. The address for the entities listed is 3340 Hillview Avenue, Palo Alto, CA 94304.
(7)Based on a Schedule 13D/A filed on March 27, 2026 and Company records. Consists of (i) 298,647 shares of common stock and 6,743,321 shares of non-voting common stock held by Fairmount Healthcare Fund II L.P. (“Fairmount Fund II”). Fairmount Funds Management LLC (“Fairmount”) is the investment manager for Fairmount Fund II. Peter Harwin and Tomas Kiselak are founding partners of Fairmount. Fairmount, Peter Harwin and Tomas Kiselak may be deemed to have voting and investment power over the shares held by Fairmount Fund II. Fairmount, Peter Harwin and Tomas Kiselak disclaim beneficial ownership of such shares, except to the extent of any pecuniary interest therein. The address for the entities listed is 200 Barr Harbor Drive, Suite 400, West Conshohocken, PA 19428.
(8)Includes 1,049,014 shares of common stock, 103,973 shares of restricted voting common stock and 379,801 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(9)Includes 160,249 shares of common stock, 48,149 shares of restricted voting common stock and 108,988 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(10)Includes 134,272 shares of common stock, 43,099 shares of restricted voting common stock and 157,168 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(11)Includes 20,000 shares of common stock, and 147,872 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(12)Includes 20,478 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(13)Includes 24,667 shares of common stock, 10,157 shares of restricted voting common stock and 10,370 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(14)Includes 42,208 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(15)Includes 51,166 shares of common stock and 42,208 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(16)Includes 24,667 shares of common stock, 10,157 shares of restricted voting common stock and 10,370 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(17)Includes 51,166 shares of common stock and 42,208 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(18)Includes 42,208 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026.
(19)Includes 215,535 shares of restricted voting common stock, 1,003,879 shares of common stock underlying options that have vested or will vest within 60 days of April 1, 2026 and 365,853 pre-funded warrants that are exercisable immediately. Excludes 298,647 shares of voting common stock and 6,743,321 shares of non-voting common stock that are beneficially owned by two directors to avoid double counting.
Securities Authorized for Issuance Under Equity Compensation Plans
The following table provides information as of December 31, 2025 with respect to the shares of our common stock that may be issued under our existing equity compensation plans.
|
|
|
|
|
|
|
|
|
|
|
|
|
Plan Category |
|
Number of
Securities to be
Issued Upon
Exercise of
Outstanding
Options,
Warrants and
rights |
|
|
Weighted
average
Exercise Price
of Outstanding
Options,
Warrants and
Rights(1) |
|
|
Number of
Securities
Remaining
Available for
Future Issuance
Under Equity
Compensation
Plans
(Excluding
Securities
Reflected in the
First Column) |
|
Equity compensation plans approved by stockholders(2) |
|
|
5,788,059 |
|
|
$ |
36.95 |
|
|
|
7,438,162 |
|
Total |
|
|
5,788,059 |
|
|
$ |
36.95 |
|
|
|
7,438,162 |
|
(1)The weighted-average exercise price does not take into account shares issuable upon vesting of any outstanding restricted stock units and restricted stock awards, which have no exercise price.
(2)Includes the following plans: our 2023 Plan, our ESPP, including 1,473,613 shares subject to purchase thereunder during the purchase periods in effect as of December 31, 2025, and a stock option grant to Mr. McKenna outside of the 2023 Plan (the “Non-Plan Option Grant”). Excludes 3,420,067 and 684,013 shares that were added to our 2023 Plan and our ESPP, respectively, on January 1, 2026 pursuant to the evergreen provisions thereunder that provide for automatic annual increases on January 1 of each year during the term of the respective plan equal to 5% of our outstanding shares as of the preceding December 31 (or such lesser amount as approved by the Compensation Committee), in the case of the 2023 Plan, or 1% of our outstanding shares as of the preceding December 31 (or such lesser amount as approved by the Compensation Committee), in the case of the ESPP. The Non-Plan Option Grant relates to a grant of 100,000 stock options to Mr. McKenna outside of our 2023 Plan, which was approved by stockholders in June 2024.
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
The following is a summary of each transaction or series of similar transactions since January 1, 2025 or any currently proposed transaction, to which we were or are a party in which:
•the amount involved exceeds $120,000; and
•any related person (including our directors, executive officers, beneficial owners of more than 5% of our voting capital stock and any affiliates or members of their immediate family) had or will have a direct or indirect material interest, other than compensation and other arrangements that are described under the sections titled “Executive Compensation” and “Director Compensation” or that were approved by our Compensation Committee.
Beneficial ownership of securities is determined in accordance with the rules of the SEC.
Related Party Transactions
Our Relationship with Paragon
We are party to a number of agreements with Paragon. Paragon beneficially owns more than 5% of our capital stock through its holdings of incentive units and common units. Fairmount Funds Management LLC beneficially owns more than 5% of our capital stock, has two seats on our Board, and, as of May 11, 2026, will have one seat on our Board, and beneficially owns more than 5% of Paragon, which is a joint venture between Fairmount Funds Management LLC and Fair Journey Biologics. Fairmount Funds Management LLC has appointed the sole director on Paragon’s board of directors and has the contractual right to approve the appointment of any executive officers.
In February 2022, we entered into an antibody discovery and option agreement with Paragon, which was subsequently amended in November 2022 (as amended, the “2022 Option Agreement”). In consideration for the exclusive options granted under the 2022 Option Agreement, we paid an upfront cash amount of $1.3 million and issued 1,250,000 common units to Paragon. Paragon was also entitled to up to an additional 3,750,000 of common units in exchange for the rights granted under the 2022 Option Agreement, which were issued in connection with the closings of the additional tranches of the Series A Preferred Unit financing. Pursuant to the terms of the 2022 Option Agreement, the parties will initiate certain research programs that will generally be focused on a particular target (each, a “Research Program”). Each Research Program is aimed at discovering, generating, identifying and/or characterizing antibodies directed to the respective target. For each Research Program, the parties established a research plan that sets forth the activities that will be conducted, and the associated research budget (each, a “Research Plan”). On a Research Program-by-Research Program basis following the finalization of the Research Plan for each respective Research Program, we are required to pay Paragon a nonrefundable fee in cash of $0.5 million under the 2022 Option Agreement. We are also obligated to compensate Paragon on a quarterly basis for its services performed under each Research Program based on the actual costs incurred.
In November 2023, we entered into an additional antibody discovery and option agreement with Paragon (the “2023 Option Agreement” and, together with the 2022 Option Agreement, the “Option Agreements”). On a Research Program-by-Research Program basis following the finalization of the Research Plan for each respective Research Program, we are required to pay Paragon a nonrefundable fee in cash of $2.0 million under the 2023 Option Agreement. We are also obligated to compensate Paragon on a quarterly basis for its services performed under each Research Program based on the actual costs incurred. In January 2024, we finalized the Research Plan with Paragon related to the TSLP target. As such, we made a one-time non-refundable payment of $2.0 million to Paragon in the first quarter of 2024.
In November 2022, we exercised our option available under the 2022 Option Agreement with respect to the IL-13 Research Program and entered into an associated license agreement with Paragon (the “IL-13 License Agreement”). In April 2023, we exercised our option available under the 2022 Option Agreement with respect to the IL-4Rα Research Program and the OX40L Research Program and entered into associated license agreements with Paragon (respectively, the “IL-4Rα License Agreement” and the “OX40L License Agreement”). In connection with each such exercise, we paid Paragon a nonrefundable fee in cash of $0.5 million per Research Program. In August 2024, we exercised our option available under the 2023 Option Agreement with respect to the TSLP Research Program and
entered into the associated license agreement (the “TSLP License Agreement” and together with the IL-13 License Agreement, the IL-4Rα License Agreement, and the OX40L License Agreement, the “License Agreements”). We are also obligated to pay Paragon up to $3.0 million upon the achievement of specific development and clinical milestones for the first product under each of the IL-13 License Agreement, IL-4Rα License Agreement and OX40L License Agreement that achieves such specified milestones. Under the TSLP License Agreement, we are obligated to pay Paragon up to $28.0 million upon the achievement of specific development and clinical milestones for the first product, including a payment of $3.0 million upon the nomination of a development candidate and $5.0 million upon the first dosing of a human patient in a Phase 1 trial. Upon execution of the IL-13 License Agreement, we paid Paragon a $1.0 million fee for nomination of a development candidate. In August 2023, we announced the dosing of our first participant in the Phase 1 trial of APG777 and made a milestone payment of $2.0 million to Paragon in the fourth quarter of 2023. In November 2023, we finalized the nomination of a development candidate under the IL-4Rα License Agreement and made a milestone payment of $1.0 million to Paragon in the fourth quarter of 2023. In March 2024, we announced the dosing of our first participants in a Phase 1 trial of APG808 and made a milestone payment of $2.0 million to Paragon in the first quarter of 2024. In May 2024, we finalized the nomination of a development candidate under the OX40L License Agreement and made a milestone payment of $1.0 million to Paragon in the second quarter of 2024. In August 2024, we announced the dosing of our first participants in the Phase 1 trial of APG990 and made a milestone payment of $2.0 million in the third quarter of 2024. In October 2024, we finalized the nomination of a development candidate under the TSLP License Agreement and made a milestone payment of $3.0 million to Paragon in the fourth quarter of 2024. In December 2024, we announced the dosing of our first participant in the Phase 1 trial of APG333 and made a milestone payment of $5.0 million in the fourth quarter of 2024. The Option Agreements and License Agreements were negotiated on an arm’s-length basis and are market rate transactions on terms that we believe are no less favorable than would have been reached with an unrelated third party. For additional detail regarding our arrangements with Paragon, see Part I, Item 1, “Business - Our Collaboration, License and Services Agreements” of our Annual Report on Form 10-K for the year ended December 31, 2025.
For the twelve months ended December 31, 2025, we recognized $0.1 of expense in connection with the services provided by Paragon under the Option Agreements and License Agreements and $2.1 million of research and development expense related to an undisclosed target. For the period from January 1, 2026 through March 31, 2026, we recognized approximately $0.6 million of research and development expense related to an undisclosed target,
In connection with our IPO, we entered into agreements to indemnify our directors and executive officers. These agreements, among other things, require us to indemnify these individuals for certain expenses (including attorneys’ fees), judgments, fines and settlement amounts reasonably incurred by such person in any action or proceeding, including any action by or in our right, on account of any services undertaken by such person on behalf of our company or that person’s status as a director or officer, as applicable, to the maximum extent allowed under Delaware law.
Reorganization
In connection with our IPO, the members of Apogee Therapeutics, LLC contributed their units in Apogee Therapeutics, LLC to Apogee Therapeutics, Inc. in exchange for shares of common stock and/or non-voting common stock of Apogee Therapeutics, Inc, as applicable.
Registration Rights Agreement
We have entered into a registration rights agreement with the holders of 24,987,750 shares of our common stock (including shares of common stock issuable upon conversion of our non-voting common stock). The registration of shares of our common stock pursuant to the exercise of registration rights described below would enable holders to sell these shares without restriction under the Securities Act of 1933, as amended (the “Securities Act”), when the registration statement is declared effective. We will pay all expenses related to any demand, piggyback or Form S-3 registration described below, with the exception of underwriting discounts, selling commissions, and stock transfer taxes.
The registration rights described below will expire upon the earliest to occur of: (i) three years after the completion of our IPO; (ii) the closing of a merger or consolidation in which (A) we are constituent party or (B) a subsidiary of ours is a constituent party and we issue shares of our capital stock pursuant to such merger or consolidation; or (iii), with respect to any particular holder, at such time that such holder can sell its shares, under Rule 144 or another similar exemption under the Securities Act, during any three-month period without registration.
Form S-1 Demand Registration Rights
The holders of registrable securities who are party to the registration rights agreement (the “Registration Rights Holders”) are entitled to certain demand registration rights. At any time after January 9, 2024, Registration Rights Holders who hold a majority of the registrable securities then outstanding may request that we file a Form S-1 registration statement for which the anticipated aggregate offering price would exceed $20,000,000.
Form S-3 Demand Registration Rights
At any time after we are qualified to file a registration statement on Form S-3, and subject to limitations and conditions, Registration Rights Holders who hold at least 30% of the registrable securities then outstanding may make a written request that we prepare and file a registration statement on Form S-3 under the Securities Act covering their shares, so long as the aggregate price to the public, net of the underwriters’ discounts and commissions, is at least $5,000,000. We will prepare and file the Form S-3 registration statement as requested, unless, in the good faith judgment of our Board, such registration would be materially detrimental to the Company and its stockholders and filing should be deferred. We may defer only once in any 12-month period, and such deferral shall not exceed 90 days after receipt of the request. In addition, we are not obligated to prepare or file any of these registration statements (i) during the period that is 30 or 60 days, as the case may be, before our good faith estimate of the date of filing of, and ending on a date that is 180 days after the effective date of, a Company-initiated registration or (ii) if two of these registrations have been completed within any 12-month period.
Piggyback Registration Rights
Subject to certain specified exceptions, if we propose to register any of our securities under the Securities Act either for our own account or for the account of other stockholders, the Registration Rights Holders are entitled to notice and certain “piggyback” registration rights allowing them to include their shares in our registration statement. These registration rights are subject to specified conditions and limitations, including the right of the underwriters, in their sole discretion, to limit the number of shares included in any such offering under certain circumstances, but not below 30% of the total amount of securities included in such offering.
Related Party Transaction Policy
Our Board has a written policy regarding the review and approval or ratification by our Audit Committee of related person transactions. For purposes of our policy only, a related person transaction is a transaction, arrangement or relationship or any series of similar transactions, arrangements or relationships between us or any of our subsidiaries and any related person in which the aggregate amount involved since the beginning of our last completed fiscal year exceeds or is expected to exceed $120,000 and such related person has or will have a direct or indirect interest. A related person is defined to include any executive officers, directors or director nominees or beneficial owner of more than 5% of our common stock and any immediate family member of any of the foregoing persons. In determining to approve or ratify any such transaction, our Audit Committee is expected to take into account, among other factors it deems appropriate, whether the transaction is on terms no less favorable than terms generally available to an unaffiliated third-party under the same or similar circumstances and the extent of the related person’s interest in the transaction. Transactions involving compensation for services provided to us as an employee or director, among other limited exceptions, are deemed under the terms of the policy to have standing pre-approval by the Audit Committee but may be specifically reviewed if appropriate in light of the facts and circumstances. Any director who is a related person with respect to a transaction under review is not permitted to participate in the deliberations (other than to provide information concerning the transaction to the Audit Committee) or vote on approval of the transaction.
The 2022 Option Agreement described above was consummated prior to our adoption of the formal, written policy described above, and, accordingly, the foregoing policies and procedures were not followed with respect to this transaction.
OTHER MATTERS
Stockholder Proposals and Director Nominations for Next Year’s Annual Meeting
Pursuant to Rule 14a-8 of the Exchange Act, stockholders who wish to submit proposals for inclusion in the proxy statement for the 2027 Annual Meeting of Stockholders must send such proposals to our Corporate Secretary at the address set forth on the first page of this Proxy Statement. Such proposals must be received by us as of the close of business (6:00 p.m. Eastern Time) on December 25, 2026 and must comply with Rule 14a-8 of the Exchange Act. The submission of a stockholder proposal does not guarantee that it will be included in the proxy statement.
As set forth in our Bylaws, if a stockholder intends to make a nomination for director election or present a proposal for other business (other than pursuant to Rule 14a-8 of the Exchange Act) at the 2027 Annual Meeting of Stockholders, the stockholder’s notice must be received by our Corporate Secretary at the address set forth on the first page of this Proxy Statement no earlier than the 120th day and no later than the close of business (6:00 p.m. Eastern Time) on the 90th day before the anniversary of the last annual meeting; provided, however, that if the date of the annual meeting is more than 30 days before or more than 60 days after such anniversary date, the stockholder’s notice must be delivered not earlier than the 120th day prior to such annual meeting and not later than the close of business on the later of the 90th day prior to such annual meeting or the 10th day following the date on which the first public announcement of the date of such annual meeting is made by the Company. Therefore, unless the 2027 Annual Meeting of Stockholders is more than 30 days before or more than 60 days after the anniversary of the Annual Meeting, notice of proposed nominations or proposals (other than pursuant to Rule 14a-8 of the Exchange Act) must be received by our Corporate Secretary no earlier than February 9, 2027 and no later than the close of business on March 11, 2027. Any such director nomination or stockholder proposal must be a proper matter for stockholder action and must comply with the terms and conditions set forth in our Bylaws (which includes the timing and information required under Rule 14a-19 of the Exchange Act). If a stockholder fails to meet these deadlines or fails to satisfy the requirements of Rule 14a-4 of the Exchange Act, we may exercise discretionary voting authority under proxies we solicit to vote on any such proposal as we determine appropriate. We reserve the right to reject, rule out of order or take other appropriate action with respect to any nomination or proposal that does not comply with these and other applicable requirements.
Delivery of Documents to Stockholders Sharing an Address
A number of brokerage firms have adopted a procedure approved by the SEC called “householding”. Under this procedure, certain stockholders who have the same address and do not participate in electronic delivery of proxy materials will receive only one copy of the proxy materials, including this Proxy Statement, the Notice and our Annual Report on Form 10-K for the year ended December 31, 2025, until such time as one or more of these stockholders notifies us that they wish to receive individual copies. This procedure helps to reduce duplicate mailings and save printing costs and postage fees, as well as natural resources. If you received a “householding” mailing this year and would like to have additional copies of the proxy materials mailed to you, please send a written request to our Corporate Secretary at the address set forth on the first page of this Proxy Statement, or call (650) 394-5230, and we will promptly deliver the proxy materials to you. Please contact your broker if you received multiple copies of the proxy materials and would prefer to receive a single copy in the future, or if you would like to opt out of “householding” for future mailings.
Availability of Additional Information
We will provide, free of charge, a copy of our Annual Report on Form 10-K for the year ended December 31, 2025, including exhibits, upon the written or oral request of any stockholder of the Company. Please send a written request to our Corporate Secretary at the address set forth on the first page of this Proxy Statement, or call the number above.

SCAN TO VIEW MATERIALS & VOTE APOGEE THERAPEUTICS, INC. 221 CRESCENT STREET BUILDING 17, SUITE 102B WALTHAM, MA 02453 VOTE BY INTERNET Before The Meeting - Go to www.proxyvote.com or scan the QR Barcode above Use the Internet to transmit your voting instructions up until 11:59 p.m. Eastern Time on June 8, 2026. Have your proxy card in hand when you access the website and follow the instructions to obtain your records and to create an electronic voting instruction form. During The Meeting - Go to www.virtualshareholdermeeting.com/APGE2026 You may attend the meeting via the Internet and vote during the meeting. Have the information that is printed in the box marked by the arrow available and follow the instructions. VOTE BY PHONE - 1-800-690-6903 Use any touch-tone telephone to transmit your voting instructions up until 11:59 p.m. Eastern Time on June 8, 2026. Have your proxy card in hand when you call and then follow the instructions. VOTE BY MAIL Mark, sign and date your proxy card and return it in the postage-paid envelope we have provided or return it to Vote Processing, c/o Broadridge, 51 Mercedes Way, Edgewood, NY 11717. V94482-P47026 APOGEE THERAPEUTICS, INC. For Withhold For All To withhold authority to vote for any individual All All Except nominee(s), mark "For All Except" and write the The Board recommends that you vote these shares "FOR" number(s) of the nominee(s) on the line below. each director nominee in Proposal 1, "FOR" Proposal 2 and "FOR" Proposal 3. !!! 1.Election of Class III Directors for terms expiring in 2029. Nominees: 01) Mark C. McKenna 02) Jennifer Fox 03) William (BJ) Jones, Jr. For Against Abstain 2. Ratification of the appointment of Ernst & Young LLP as the Company’s independent registered public accounting firm for the year ending December 31, 2026. 3. Approval, on a non-binding advisory basis, of the compensation of our named executive officers as disclosed in the proxy statement NOTE: Such other business as may properly come before the meeting or any adjournment or postponement thereof. Please sign exactly as your name(s) appear(s) hereon. When signing as attorney, executor, administrator, or other fiduciary, please give full title as such. Joint owners should each sign personally. All holders must sign. If a corporation or partnership, please sign in full corporate or partnership name by authorized officer.

Important Notice Regarding the Availability of Proxy Materials for the Annual Meeting: The Notice, Proxy Statement and Annual Report are available at www.proxyvote.com. V94483-P47026 APOGEE THERAPEUTICS, INC. ANNUAL MEETING OF STOCKHOLDERS JUNE 9, 2026 11:00 AM EASTERN TIME THIS PROXY IS SOLICITED ON BEHALF OF THE BOARD OF DIRECTORS The stockholder(s) hereby appoint(s) Michael Henderson, M.D., Jane Pritchett Henderson, and Matt Batters, or any of them, as proxies and attorneys-in-fact, each with the power to act without the others and with the power to appoint their substitute, and hereby authorize(s) them to represent and to vote, as designated on the reverse side of this form, all of the shares of Common Stock of Apogee Therapeutics, Inc. that the stockholder(s) is/are entitled to vote at the Annual Meeting of Stockholders to be held at 11:00 AM, Eastern Time on June 9, 2026, live via the Internet at www.virtualshareholdermeeting.com/APGE2026 or at any adjournment or postponement thereof. This proxy, when properly executed, will be voted in the manner directed herein. If no such direction is made but the card is signed, this proxy will be voted in accordance with the Board of Directors’ recommendations and in the discretion of the proxies with respect to such other business as may properly come before the meeting or any adjournment or postponement thereof. In the event that any of the director nominees named on the reverse side of this form are unavailable for election or unable to serve, the shares represented by the proxy may be voted for a substitute director nominee selected by the Board of Directors. Continued and to be signed on reverse side





